Collaboration to Advance Molecular Imaging Technology
|
By HospiMedica staff writers Posted on 20 Oct 2003 |
A collaboration to develop a cost-effective, readily accessible molecular imaging technology that can help more clinics and hospitals diagnose cancer has been announced by Philips Medical Systems (Best, The Netherlands) and CellPoint, L.L.C. (Englewood, CO, USA).
The collaboration will combine Cellpoint's novel diagnostic imaging agent and the nuclear medicine camera of Philips to make functional imaging readily available. The agreement will utilize CellPoint's ethylenedicysteine drug conjugate technology (EC technology), a delivery system that links tissue-specific ligands (hormones, proteins, peptides) or pharmaceutical compounds to radioisotopes for cancer diagnosis and treatment. Using clinical trial data, Philips will further develop and refine imaging techniques for SKYlight, its gantry-free nuclear camera, to determine requirements for the highest possible image quality and quantitative information about patients' disease.
The companies will collaborate on CellPoint's first imaging agent, Tc-99m-EC-deoxyglucose. EC technology allows the deoxyglucose to be labeled with the radioisotope technetium-99m (Tc-99m). The agent will target tumors, while single positron emission computed tomography (SPECT) cameras will image them, including Philips' nuclear medicine cameras. Tumors absorb more glucose than surrounding tissue, so when a cancer patient is injected with this chemically linked agent, active tumors will absorb both the glucose and the radioisotope.
"Philips has outstanding SPECT technology, and we're thrilled to strategically align ourselves with them to help advance molecular imaging agents, enabling more facilities to conduct cancer screening,” said Greg Colip, CEO of CellPoint. The company obtained the worldwide license to EC technology from the University of Texas M.D. Anderson Cancer Center (Houston, USA).
Related Links:
CellPoint
Philips Medical
The collaboration will combine Cellpoint's novel diagnostic imaging agent and the nuclear medicine camera of Philips to make functional imaging readily available. The agreement will utilize CellPoint's ethylenedicysteine drug conjugate technology (EC technology), a delivery system that links tissue-specific ligands (hormones, proteins, peptides) or pharmaceutical compounds to radioisotopes for cancer diagnosis and treatment. Using clinical trial data, Philips will further develop and refine imaging techniques for SKYlight, its gantry-free nuclear camera, to determine requirements for the highest possible image quality and quantitative information about patients' disease.
The companies will collaborate on CellPoint's first imaging agent, Tc-99m-EC-deoxyglucose. EC technology allows the deoxyglucose to be labeled with the radioisotope technetium-99m (Tc-99m). The agent will target tumors, while single positron emission computed tomography (SPECT) cameras will image them, including Philips' nuclear medicine cameras. Tumors absorb more glucose than surrounding tissue, so when a cancer patient is injected with this chemically linked agent, active tumors will absorb both the glucose and the radioisotope.
"Philips has outstanding SPECT technology, and we're thrilled to strategically align ourselves with them to help advance molecular imaging agents, enabling more facilities to conduct cancer screening,” said Greg Colip, CEO of CellPoint. The company obtained the worldwide license to EC technology from the University of Texas M.D. Anderson Cancer Center (Houston, USA).
Related Links:
CellPoint
Philips Medical
Latest Business News
- Olympus Partnership Aims to Expand Access to Robot-Assisted Endoscopic Therapy
- Johnson & Johnson Launches AI-Driven Cardiac Mapping System
- Proximie Advances AI-Driven Intelligent Operating Rooms with NVIDIA Collaboration
- GE HealthCare, DeepHealth Expand AI Breast Imaging Collaboration
- Sinocare Presents AI-Driven Integrated Digital Health Solutions at CMEF
- GE HealthCare and Medtronic Expand Alliance with Intraoperative Imaging Integration
- New Partnership Advances Physical AI into Perioperative Workflows
- External Liver Assist System Receives FDA RMAT Designation
- New Partnership Expands Access to Predictive Tool for Patient Monitoring
- GE HealthCare Leads Major European Initiative to Advance Cardio-Oncology Care
- Medtronic and GE HealthCare Broaden Alliance Across Monitoring and Care Solutions
- Quantum Surgical Acquires NeuWave from Johnson & Johnson
- Medtronic to Acquire Coronary Artery Medtech Company CathWorks
- Medtronic and Mindray Expand Strategic Partnership to Ambulatory Surgery Centers in the U.S.
- FDA Clearance Expands Robotic Options for Minimally Invasive Heart Surgery
- WHX in Dubai (formerly Arab Health) to debut specialised Biotech & Life Sciences Zone as sector growth accelerates globally
Channels
Artificial Intelligence
view channel
FDA-Cleared AI System Detects Sepsis Earlier and Reduces Mortality
Sepsis remains one of the deadliest complications for hospitalized patients, in part because its early signs overlap with other conditions. Each hour of delayed recognition measurably decreases survival,... Read moreFacial Image Analysis Tracks Biological Aging, Predicts Cancer Outcomes
Biological aging is the progressive loss of physiological function that may diverge from chronological age. In cancer care, clinicians need simple tools that reflect dynamic changes in patient resilience... Read moreCritical Care
view channel
High-Frequency Ultrasound Disables Viruses While Sparing Human Cells
Respiratory viral infections such as influenza A and COVID-19 continue to challenge patient management. Antiviral drugs are generally difficult to develop, and safe, broadly applicable options remain limited.... Read more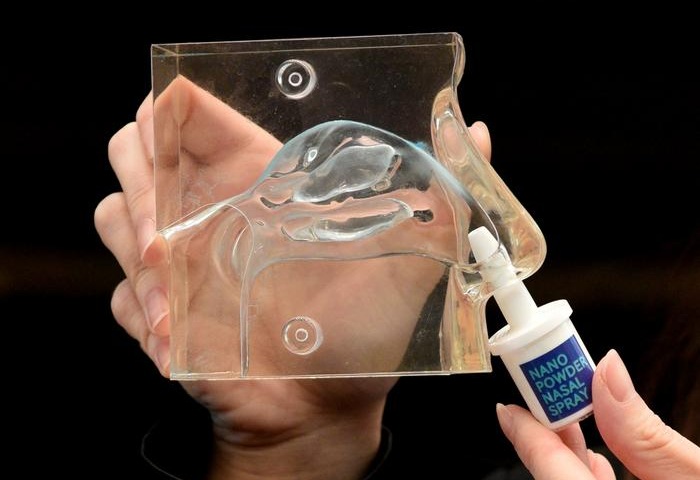
New Nasal Spray Enables Prehospital Neuroprotection in Ischemic Stroke
Ischemic stroke, caused by arterial occlusion in the brain, requires time‑critical intervention to limit neuronal loss. Many patients arrive outside the narrow window for thrombolysis or thrombectomy,... Read more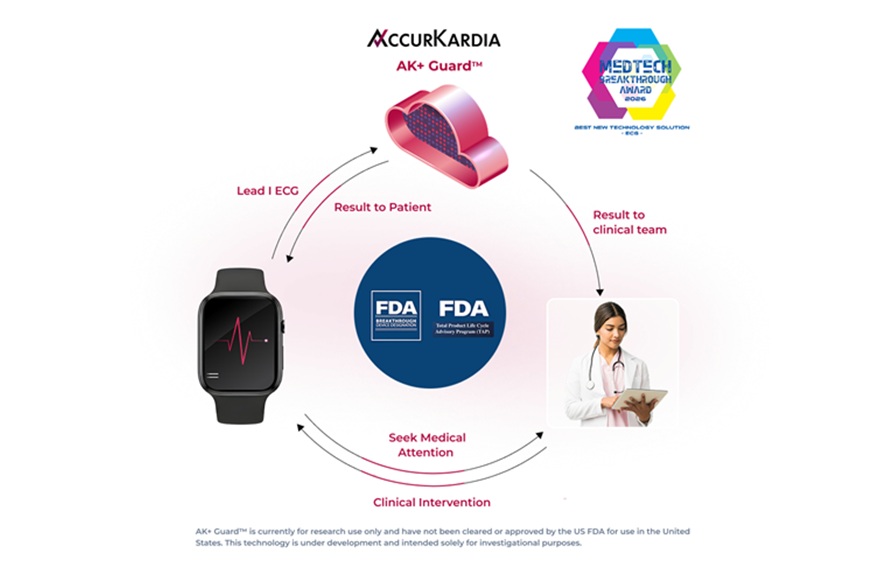
Single-Lead AI ECG Tool Detects Moderate-to-Severe Hyperkalemia Outside Clinic
Hyperkalemia can escalate quickly and is commonly monitored through laboratory blood draws, leaving risk between clinic visits. Clinicians need timely, accessible tools to identify moderate to severe elevations... Read moreSurgical Techniques
view channel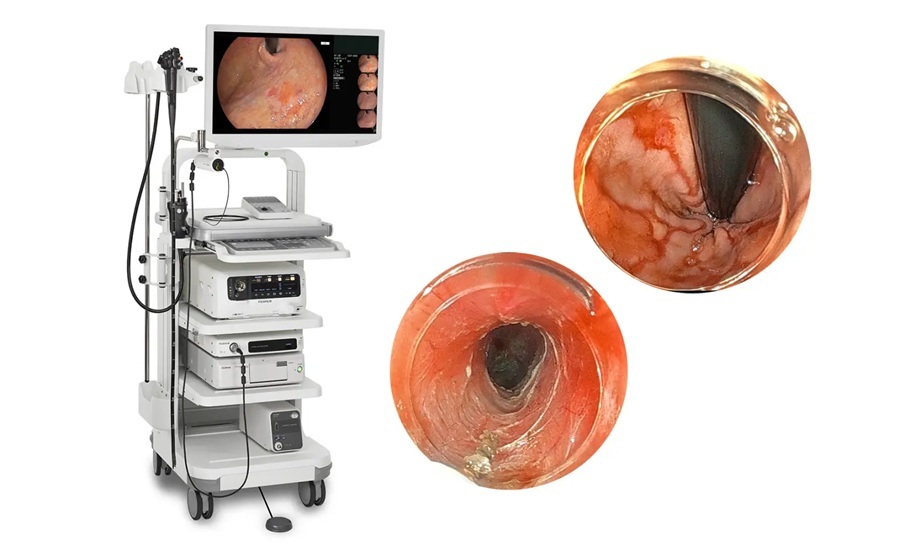
Advanced Endoscopy Platform Targets Challenging Upper GI Procedures
Fujifilm’s ELUXEO 8000 Endoscopic Imaging System, used with the EG-840TP ultra‑slim therapeutic gastroscope, was named “Best New Endoscopy Technology Solution” in the 10th annual MedTech Breakthrough Awards program.... Read more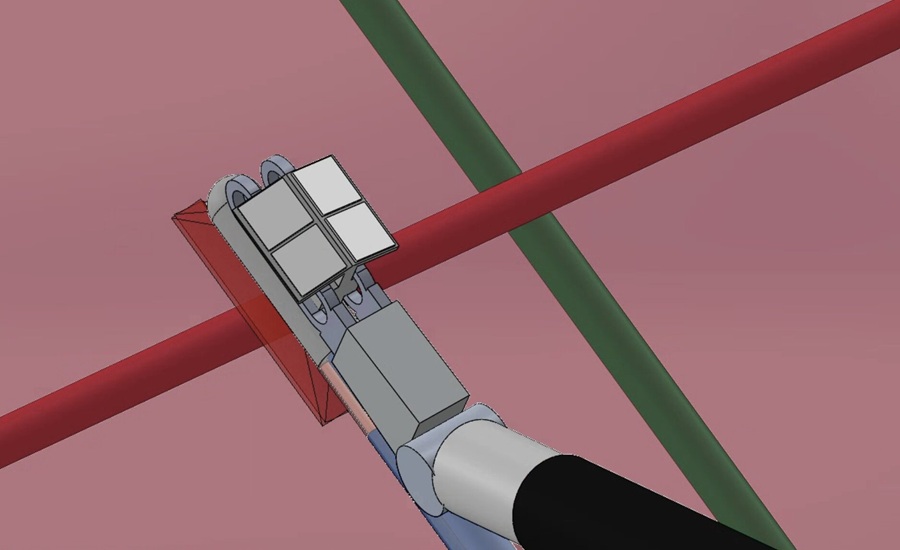
Photoacoustic Imaging System Maps Hidden Nerves and Vessels in Robotic Laparoscopy
Hidden neurovascular structures can be difficult to visualize during robot-assisted laparoscopy, increasing the risk of iatrogenic injury. Such injuries can cause hemorrhage, neurological deficits, and... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read more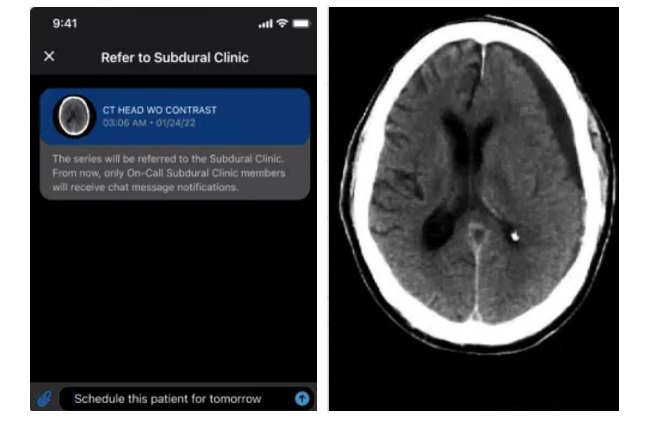
AI System Detects and Quantifies Chronic Subdural Hematoma
Viz.ai (San Francisco, CA, USA) announced a strategic commercialization collaboration with Johnson & Johnson (New Brunswick, NJ, USA) to expand access in the United States to the Viz Subdural solution... Read more
Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
Patients leaving skilled nursing facilities often lose continuous physiologic monitoring, increasing the risk of undetected infection and delayed intervention. Nursing home residents are seven times more... Read more
Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
Cardiogenic shock remains a difficult, time-sensitive emergency, with delayed identification driving poor outcomes and persistently high mortality. Many cases go undocumented even at advanced stages, hindering... Read morePoint of Care
view channel