Placenta Stem Cells Could Help Repair the Mothers' Heart
|
By HospiMedica International staff writers Posted on 23 Nov 2011 |
Stem cells from the placenta can travel to injured tissues in the mother's heart during pregnancy and create new cardiac cells, according to a new study.
Researchers at Mount Sinai Medical Center (MSMC; New York, NY, USA) induced heart attacks in pregnant mice and then checked to see if the fetal cells had transferred to the mother. To make the fetal cells easy to spot, they were genetically tagged with green fluorescent protein (GFP). The researchers found that the fetal cells selectively and specifically migrated to the injured zone of the maternal hearts, and not to noninjured organs within the same mouse. Two weeks after the animal's induced heart attack, fetal cells comprised 2% of the maternal heart; furthermore, the fetus-derived cells differentiated into heart muscle cells and cells that form blood vessels or the endothelial lining of blood vessels and other organs.
The cells could similarly differentiate when isolated from the mother's heart and grown in Petri dishes, even forming beating cardiac cells in culture. About 40% of the fetal cells that incorporated into the mothers' hearts expressed Cdx2, a marker of trophoblast stem cells, which form when embryonic stem cells develop; though embryonic stem cells lead to all the cell types in fetus, trophoblast stem cells give rise to the placenta.
The researchers are now conducting lineage-tracing experiments to confirm the precise origin of the cells. The nonexpressing cells (60%) could still be from the placenta, and may have switched off Cdx2 or other early progenitor markers and differentiated into cardiac cell types. The study was presented at the American Heart Association (AHA) Scientific Sessions 2011, held during November 2011 in Orlando (FL, USA).
“If we can confirm that trophoblast stem cells are directly involved in mediating cardiac repair in the injury state, then we may just have found that elusive cell type, other than the embryonic stem cell, that can truly become a beating heart muscle cell,” said senior author and study presenter cardiologist Hina Chaudhry, MD. “You could bank placentas and isolate stem cells and potentially use them in other individuals and not just for peripartum cardiomyopathy, but for a broad spectrum of heart diseases.”
The placenta is an organ that connects the developing fetus to the uterine wall to allow nutrient uptake, waste elimination, and gas exchange via the mother's blood supply. The placenta functions as a fetomaternal organ with two components: the fetal placenta (Chorion frondosum), which develops from the same sperm and egg cells that form the fetus, and the maternal placenta (Decidua basalis), which develops from the maternal uterine tissue.
Related Links:
Mount Sinai Medical Center
Researchers at Mount Sinai Medical Center (MSMC; New York, NY, USA) induced heart attacks in pregnant mice and then checked to see if the fetal cells had transferred to the mother. To make the fetal cells easy to spot, they were genetically tagged with green fluorescent protein (GFP). The researchers found that the fetal cells selectively and specifically migrated to the injured zone of the maternal hearts, and not to noninjured organs within the same mouse. Two weeks after the animal's induced heart attack, fetal cells comprised 2% of the maternal heart; furthermore, the fetus-derived cells differentiated into heart muscle cells and cells that form blood vessels or the endothelial lining of blood vessels and other organs.
The cells could similarly differentiate when isolated from the mother's heart and grown in Petri dishes, even forming beating cardiac cells in culture. About 40% of the fetal cells that incorporated into the mothers' hearts expressed Cdx2, a marker of trophoblast stem cells, which form when embryonic stem cells develop; though embryonic stem cells lead to all the cell types in fetus, trophoblast stem cells give rise to the placenta.
The researchers are now conducting lineage-tracing experiments to confirm the precise origin of the cells. The nonexpressing cells (60%) could still be from the placenta, and may have switched off Cdx2 or other early progenitor markers and differentiated into cardiac cell types. The study was presented at the American Heart Association (AHA) Scientific Sessions 2011, held during November 2011 in Orlando (FL, USA).
“If we can confirm that trophoblast stem cells are directly involved in mediating cardiac repair in the injury state, then we may just have found that elusive cell type, other than the embryonic stem cell, that can truly become a beating heart muscle cell,” said senior author and study presenter cardiologist Hina Chaudhry, MD. “You could bank placentas and isolate stem cells and potentially use them in other individuals and not just for peripartum cardiomyopathy, but for a broad spectrum of heart diseases.”
The placenta is an organ that connects the developing fetus to the uterine wall to allow nutrient uptake, waste elimination, and gas exchange via the mother's blood supply. The placenta functions as a fetomaternal organ with two components: the fetal placenta (Chorion frondosum), which develops from the same sperm and egg cells that form the fetus, and the maternal placenta (Decidua basalis), which develops from the maternal uterine tissue.
Related Links:
Mount Sinai Medical Center
Latest Critical Care News
- Automated IV Labeling Solution Improves Infusion Safety and Efficiency
- First-Of-Its-Kind AI Tool Detects Pulmonary Hypertension from Standard ECGs
- 4D Digital Twin Heart Model Improves CRT Outcomes
- AI Turns Glucose Data Into Actionable Insights for Diabetes Care
- Microscale Wireless Implant Tracks Brain Activity Over Time
- Smart Mask Delivers Continuous, Battery-Free Breath Monitoring
- Routine Blood Pressure Readings May Identify Risk of Future Cognitive Decline
- CGM-Based Algorithm Enhances Insulin Dose Adjustment in Type 2 Diabetes
- Fish Scale–Based Implants Offer New Approach to Corneal Repair
- Dual-Function Wound Patch Combines Infection Sensing and Treatment
- Smartwatch Signals and Blood Tests Team Up for Early Warning on Insulin Resistance
- Smart Fabric Technology Aims to Prevent Pressure Injuries in Hospital Care
- Standardized Treatment Algorithm Improves Blood Pressure Control
- Combined Infection Control Strategy Limits Drug-Resistant Outbreak in NICU
- AI Helps Predict Which Heart-Failure Patients Will Worsen Within a Year
- Algorithm Allows Paramedics to Predict Brain Damage Risk After Cardiac Arrest
Channels
Artificial Intelligence
view channel
Machine Learning Approach Enhances Liver Cancer Risk Stratification
Hepatocellular carcinoma, the most common form of primary liver cancer, is often detected late despite targeted surveillance programs. Current screening guidelines emphasize patients with known cirrhosis,... Read more
New AI Approach Monitors Brain Health Using Passive Wearable Data
Brain health spans cognitive and emotional functions and can fluctuate even in adults without diagnosed disease. Detecting early changes remains difficult in routine care and burdens specialty services... Read moreSurgical Techniques
view channel
Continuous Monitoring with Wearables Enhances Postoperative Patient Safety
Postoperative hypoxemia on general surgical wards is common and often missed by intermittent vital sign checks. Undetected low oxygen levels can delay recovery and raise the risk of complications that... Read more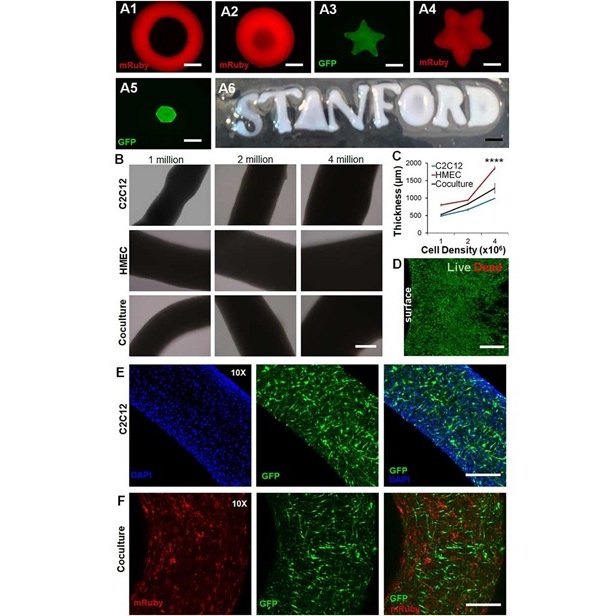
New Approach Enables Customized Muscle Tissue Without Biomaterial Scaffolds
Volumetric muscle loss is a traumatic loss of skeletal muscle that often leads to permanent functional impairment and limited reconstructive options. Current experimental strategies struggle to deliver... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read moreBusiness
view channel