Electroceutical Device Eases Chronic Back and Leg Pain 
|
By HospiMedica International staff writers Posted on 15 Dec 2014 |

Image: The Freedom Spinal Cord Stimulation (SCS) system (Photo courtesy of Stimwave Technologies).
A new injectable neuromodulation device powered by an externally worn transmitter aids in the relief of chronic back and leg pain.
The Stimwave Freedom Spinal Cord Stimulation (SCS) system is based on a tiny injectable microchip that delivers small pulses of energy to electrodes near surrounding nerves, triggering a reaction that enables the brain to remap specific pain signals, thus providing pain relief. The stimulator, which is so narrow (1.3 mm) that it can be implanted through a standard needle, eliminates the need for long electrical leads necessary in pacemaker-like devices. Containing 4 electrode arrays, the stimulator is fixed in place by a suture sleeve cap anchor, serving as a permanent, long-term device.
The SCS is powered by a wearable antenna assembly (WAA) that uses wireless radiofrequency (RF) signals; wireless Bluetooth technology is also used to program the WAA with specific stimulation parameters. For added safety, the neurostimulator only accepts communication from a specific WAA. The system also features the ability to allow the patient to have a whole body 3 T or 1.5 T magnetic resonance imaging (MRI) scan, without removing the implant. The Stimwave Freedom SCS system is a product of Stimwave Technologies (Miami Beach, FL, USA), and has been approved by the US Food and Drug Administration (FDA).
“This technology is no longer an academic-type science experiment, but a real, viable innovation that can immediately start being utilized by patients in pain,” said Laura Tyler Perryman, MSc, MBA, co-inventor of the SCS system and CEO and chairman of Stimwave Technologies. “Historically with SCS, dependency on pain medications can be drastically reduced or even eliminated.”
“I am extremely excited about this new technology developed by Stimwave and recent clearance by the FDA,” said David Kloth, MD, medical director of the Connecticut Pain Care Center and author of the book Pain Wise. “Now people in pain will have additional options including the ability to receive a permanent implant with a far less invasive and complicated surgery, while avoiding the cumbersome long-term issues with recharging, as is the case today with other systems.”
SCS, in the simplest form, consists of stimulating electrodes implanted in the epidural space. It has notable analgesic properties and, at the present, is used mostly in the treatment of failed back surgery syndrome, complex regional pain syndrome, and refractory pain due to ischemia.
Related Links:
Stimwave Technologies
The Stimwave Freedom Spinal Cord Stimulation (SCS) system is based on a tiny injectable microchip that delivers small pulses of energy to electrodes near surrounding nerves, triggering a reaction that enables the brain to remap specific pain signals, thus providing pain relief. The stimulator, which is so narrow (1.3 mm) that it can be implanted through a standard needle, eliminates the need for long electrical leads necessary in pacemaker-like devices. Containing 4 electrode arrays, the stimulator is fixed in place by a suture sleeve cap anchor, serving as a permanent, long-term device.
The SCS is powered by a wearable antenna assembly (WAA) that uses wireless radiofrequency (RF) signals; wireless Bluetooth technology is also used to program the WAA with specific stimulation parameters. For added safety, the neurostimulator only accepts communication from a specific WAA. The system also features the ability to allow the patient to have a whole body 3 T or 1.5 T magnetic resonance imaging (MRI) scan, without removing the implant. The Stimwave Freedom SCS system is a product of Stimwave Technologies (Miami Beach, FL, USA), and has been approved by the US Food and Drug Administration (FDA).
“This technology is no longer an academic-type science experiment, but a real, viable innovation that can immediately start being utilized by patients in pain,” said Laura Tyler Perryman, MSc, MBA, co-inventor of the SCS system and CEO and chairman of Stimwave Technologies. “Historically with SCS, dependency on pain medications can be drastically reduced or even eliminated.”
“I am extremely excited about this new technology developed by Stimwave and recent clearance by the FDA,” said David Kloth, MD, medical director of the Connecticut Pain Care Center and author of the book Pain Wise. “Now people in pain will have additional options including the ability to receive a permanent implant with a far less invasive and complicated surgery, while avoiding the cumbersome long-term issues with recharging, as is the case today with other systems.”
SCS, in the simplest form, consists of stimulating electrodes implanted in the epidural space. It has notable analgesic properties and, at the present, is used mostly in the treatment of failed back surgery syndrome, complex regional pain syndrome, and refractory pain due to ischemia.
Related Links:
Stimwave Technologies
Latest Critical Care News
- Angiography-Based FFR Approach Matches Gold Standard Results Without Wires
- Eye Imaging AI Identifies Elevated Cardiovascular Risk
- Noninvasive Monitoring Device Enables Earlier Intervention in Heart Failure
- Automated IV Labeling Solution Improves Infusion Safety and Efficiency
- First-Of-Its-Kind AI Tool Detects Pulmonary Hypertension from Standard ECGs
- 4D Digital Twin Heart Model Improves CRT Outcomes
- AI Turns Glucose Data Into Actionable Insights for Diabetes Care
- Microscale Wireless Implant Tracks Brain Activity Over Time
- Smart Mask Delivers Continuous, Battery-Free Breath Monitoring
- Routine Blood Pressure Readings May Identify Risk of Future Cognitive Decline
- CGM-Based Algorithm Enhances Insulin Dose Adjustment in Type 2 Diabetes
- Fish Scale–Based Implants Offer New Approach to Corneal Repair
- Dual-Function Wound Patch Combines Infection Sensing and Treatment
- Smartwatch Signals and Blood Tests Team Up for Early Warning on Insulin Resistance
- Smart Fabric Technology Aims to Prevent Pressure Injuries in Hospital Care
- Standardized Treatment Algorithm Improves Blood Pressure Control
Channels
Artificial Intelligence
view channelAI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
Accurately identifying long-term cardiovascular disease risk in asymptomatic adults remains challenging for clinicians. Missed or underestimated risk delays preventive therapy and increases the chance... Read more
Machine Learning Approach Enhances Liver Cancer Risk Stratification
Hepatocellular carcinoma, the most common form of primary liver cancer, is often detected late despite targeted surveillance programs. Current screening guidelines emphasize patients with known cirrhosis,... Read moreSurgical Techniques
view channel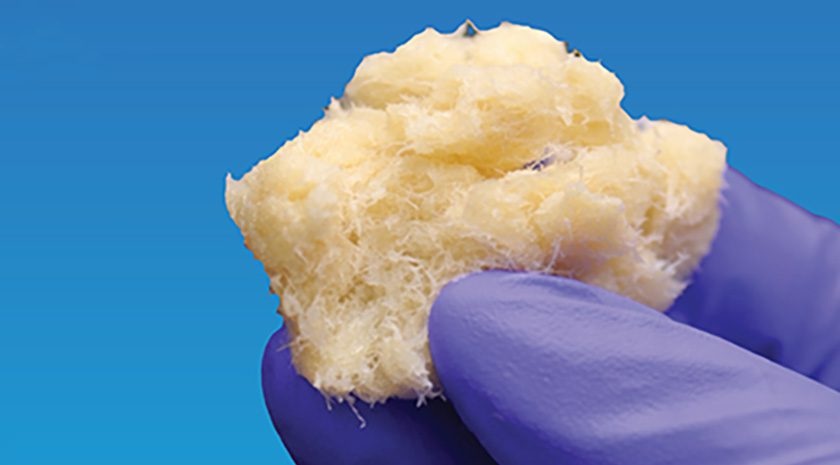
Fiber-Form Bone Graft Expands Intraoperative Options for Spinal Fusion
Spinal and orthopedic fusion procedures often require bone graft materials that handle predictably and support bone formation. Surgeons face added complexity in difficult anatomy and challenging fusion environments.... Read more
Ultrasound‑Aided Catheter Treatment Cuts Early Collapse in Pulmonary Embolism
Acute pulmonary embolism can cause rapid hemodynamic deterioration and early death in hospitalized and emergency patients. Systemic thrombolysis can dissolve clots but is limited by a high risk of major... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
Voice-Driven AI System Enables Structured GI Procedure Documentation
Documentation during gastrointestinal (GI) procedures often competes with real-time clinical decision-making and imposes a significant cognitive burden on physicians. Manual data entry and post-procedure... Read more
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read moreBusiness
view channel