Closure System Permanently Seals Varicose Veins 
|
By HospiMedica International staff writers Posted on 01 Mar 2015 |
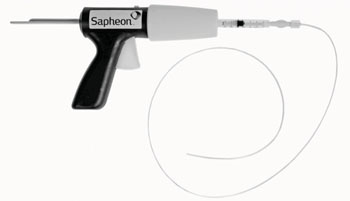
Image: The VenaSeal Closure System (Photo courtesy of Sapheon).
A novel embolic system uses an adhesive agent that polymerizes inside superficial varicose veins of the legs, sealing them shut.
The VenaSeal Closure System is composed of a specially formulated adhesive (n-butyl-2-cyanoacrylate) and a delivery system that includes a catheter, guidewire, dispenser gun, dispenser tips, and syringes. The VenaSeal Closure System does not require tumescent anesthesia to be injected peripherally into the leg, and because there are no pre-procedures drugs involved, patients can usually return to their normal activities right after the treatment, with just a Band-Aid in place.
For example, to seal the great saphenous vein (GSV), the catheter is first guided via ultrasound (US) towards the saphenofemoral junction (SFJ); once properly placed near the SFJ, the adhesive is injected into the lumen, sealing the vein. The procedure is then repeated every three cm along the GSV. Once closed, blood is immediately rerouted through other healthy veins in the leg. The VenaSeal Closure system is a product of Sapheon (Morrisville, NC, USA), a part of Covidien (Dublin, Ireland), and has been approved by the US Food and Drug Administration (FDA).
“Cyanoacrylate embolization is an effective and durable solution to the problem of incompetent great saphenous veins; long-term effectiveness is high,” said Prof. Thomas Proebstle, MD, of Mainz University (Germany), who led the European multicenter clinical study of the system. “The elimination of the need for perivenous tumescent anesthesia and post-interventional compression stockings, along with the associated side effects, results in significantly improved treatment.”
“This new system is the first to permanently treat varicose veins by sealing them with an adhesive, thereby giving patients another treatment option for this common condition,” said William Maisel, MD, MPH, acting director of the office of device evaluation in the FDA’s Center for Devices and Radiological Health (CDRH). “Because the VenaSeal system does not incorporate heat application or cutting, the in-office procedure can allow patients to quickly return to their normal activities, with less bruising.”
Varicose veins (also known as venous reflux disease) occur when valves inside the veins break and blood is unable to circulate properly. This is a common condition that affects both men (25%) and women (40%). Varicose veins can occur anywhere from the groin to the ankle.
Related Links:
Sapheon
Covidien
The VenaSeal Closure System is composed of a specially formulated adhesive (n-butyl-2-cyanoacrylate) and a delivery system that includes a catheter, guidewire, dispenser gun, dispenser tips, and syringes. The VenaSeal Closure System does not require tumescent anesthesia to be injected peripherally into the leg, and because there are no pre-procedures drugs involved, patients can usually return to their normal activities right after the treatment, with just a Band-Aid in place.
For example, to seal the great saphenous vein (GSV), the catheter is first guided via ultrasound (US) towards the saphenofemoral junction (SFJ); once properly placed near the SFJ, the adhesive is injected into the lumen, sealing the vein. The procedure is then repeated every three cm along the GSV. Once closed, blood is immediately rerouted through other healthy veins in the leg. The VenaSeal Closure system is a product of Sapheon (Morrisville, NC, USA), a part of Covidien (Dublin, Ireland), and has been approved by the US Food and Drug Administration (FDA).
“Cyanoacrylate embolization is an effective and durable solution to the problem of incompetent great saphenous veins; long-term effectiveness is high,” said Prof. Thomas Proebstle, MD, of Mainz University (Germany), who led the European multicenter clinical study of the system. “The elimination of the need for perivenous tumescent anesthesia and post-interventional compression stockings, along with the associated side effects, results in significantly improved treatment.”
“This new system is the first to permanently treat varicose veins by sealing them with an adhesive, thereby giving patients another treatment option for this common condition,” said William Maisel, MD, MPH, acting director of the office of device evaluation in the FDA’s Center for Devices and Radiological Health (CDRH). “Because the VenaSeal system does not incorporate heat application or cutting, the in-office procedure can allow patients to quickly return to their normal activities, with less bruising.”
Varicose veins (also known as venous reflux disease) occur when valves inside the veins break and blood is unable to circulate properly. This is a common condition that affects both men (25%) and women (40%). Varicose veins can occur anywhere from the groin to the ankle.
Related Links:
Sapheon
Covidien
Latest Surgical Techniques News
- Intravascular Lithotripsy Catheter Advances Treatment of Calcified Coronary Disease
- Handheld AI Endomicroscope Enables Real-Time Precancer Detection at Point of Care
- Photoacoustic Imaging System Maps Hidden Nerves and Vessels in Robotic Laparoscopy
- Smart Soft Sensors Provide Real-Time Force Feedback in Minimally Invasive Surgery
- Advanced Endoscopy Platform Targets Challenging Upper GI Procedures
- Ultrasonic Fine-Needle Biopsy Tool Improves Salivary Gland Tumor Diagnosis
- Robotic Microsurgery System Gains CE Mark for Clinical Use in Europe
- New Endoscopic Device Offers Minimally Invasive Approach to Obesity Management
- Handheld Probe Enables Real-Time Tumor Mapping in Breast-Conserving Surgery
- Robotic Surgical System Enables Gastric Bypass in Space-Constrained Operating Rooms
- Optical Wireless Link Enables Cable-Free 4K Endoscopic Imaging
- Patient-Specific 3D-Printed Guide Improves Precision in Breast-Conserving Surgery
- Glasses-Free 3D Imaging Software Receives FDA Clearance for Surgical Planning
- Noninvasive Cardiac Radiotherapy Reduces Ventricular Tachycardia Events
- Augmented Reality System for Knee Replacement Receives FDA Clearance
- New AI Tool Predicts Complications Before Lung Cancer Surgery
Channels
Artificial Intelligence
view channel
FDA-Cleared AI System Detects Sepsis Earlier and Reduces Mortality
Sepsis remains one of the deadliest complications for hospitalized patients, in part because its early signs overlap with other conditions. Each hour of delayed recognition measurably decreases survival,... Read moreFacial Image Analysis Tracks Biological Aging, Predicts Cancer Outcomes
Biological aging is the progressive loss of physiological function that may diverge from chronological age. In cancer care, clinicians need simple tools that reflect dynamic changes in patient resilience... Read moreCritical Care
view channel
AI Tool Predicts Risk of Out-of-Hospital Cardiac Arrest
Sudden cardiac arrest is a lethal event that often occurs without warning, causing more than 400,000 deaths in the U.S. each year and a survival rate of about 10%. Clinicians struggle to identify who in... Read more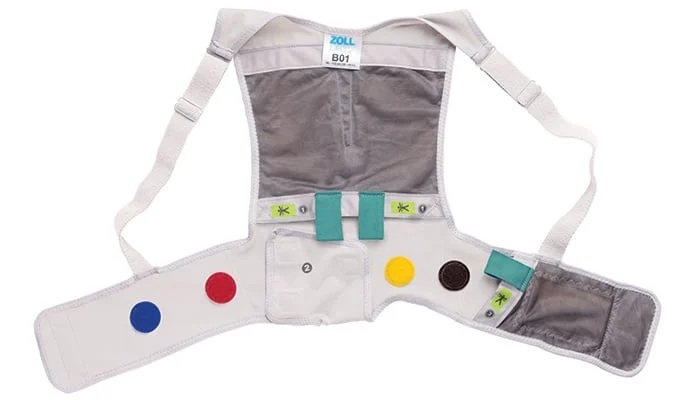
Wearable Defibrillator Supports Quicker Beta-Blocker Optimization in Women
Women with newly diagnosed heart failure with reduced ejection fraction (HFrEF) often do not reach optimal beta-blocker dosing during early treatment. Guideline-directed titration depends on achieving... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read more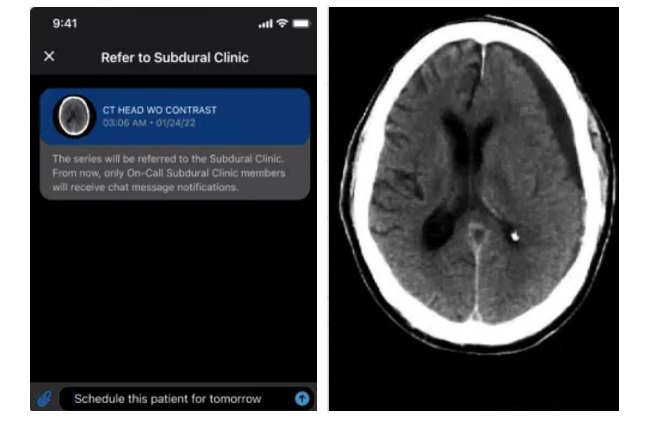
AI System Detects and Quantifies Chronic Subdural Hematoma
Viz.ai (San Francisco, CA, USA) announced a strategic commercialization collaboration with Johnson & Johnson (New Brunswick, NJ, USA) to expand access in the United States to the Viz Subdural solution... Read more
Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
Patients leaving skilled nursing facilities often lose continuous physiologic monitoring, increasing the risk of undetected infection and delayed intervention. Nursing home residents are seven times more... Read more
Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
Cardiogenic shock remains a difficult, time-sensitive emergency, with delayed identification driving poor outcomes and persistently high mortality. Many cases go undocumented even at advanced stages, hindering... Read morePoint of Care
view channel
Point-of-Care Viscoelastic Testing System Supports Obstetric Bleeding Management
HemoSonics (Durham, NC, USA) announced on May 5, 2026 that the company's Quantra Hemostasis System for Obstetric Procedures won Silver in the 2026 Edison Awards in the Women’s Health and Reproductive Innovations... Read moreBusiness
view channel
Olympus Partnership Aims to Expand Access to Robot-Assisted Endoscopic Therapy
Olympus has signed an exclusive global distribution agreement with EndoRobotics Co., Ltd., under which robot-assisted technologies developed by EndoRobotics will be distributed worldwide as part of the... Read more