Ablation Device Treats Heavy Menstrual Bleeding
|
By HospiMedica International staff writers Posted on 06 Sep 2015 |

Image: The Minerva Surgical Endometrial Ablation System (Photo courtesy of Minerva Surgical).
A new endometrial ablation system treats menorrhagia in premenopausal women due to benign causes, and for whom childbearing is complete.
The Minerva Surgical Endometrial Ablation System uses three separate ablation mechanisms to provide a safer and more effective means of treating excessive menstrual bleeding. The system is based on an impedance controlled treatment cycle specific to individual patient conditions. The first ablation step uses bipolar radiofrequency (RF) energy to ablate the endometrium. The second step uses the high voltage RF electrical current to ionize fully contained and circulated argon gas within a plasma formation array (PFA) sealed silicone membrane.
When the stretchable silicone membrane is deployed it conforms to the uterus; once energized, the argon gas is ionized, turning it in to a stream of plasma which heats the interior surface of the silicone membrane. The thermal energy is then conducted uniformly to the tissue in contact with the membrane. In the third step, released intra-cavitary fluids fills the gaps missed in the first two steps and further ablate remaining endometrial tissue. The system integration results in a faster procedure time and improved tissue contact.
Other features of the system include easy insertion, seating, and a non-stick atraumatic removal due to the lubricous nature of the silicone membrane array; a patented uterine integrity test (UIT) that automatically checks the integrity of the uterine cavity including the array membrane; and a cervical sealing balloon that positively seals the cervix canal near the internal os. The Endometrial Ablation System is a product of Minerva Surgical (Redwood City, CA, USA), and has been approved by the US Food and Drug Administration (FDA).
Menorrhagia, described as heavy and prolonged menstrual bleeding which disrupts a woman's normal activities, is one of the most common complaints in gynecology. Bleeding generally lasts for more than a week, requiring women to change their pad or tampon every two hours or less. Women with menorrhagia may also pass large blood clots and experience anemia due to the volume of blood loss. Constant lower abdominal and pelvic pain, tiredness, fatigue, and shortness of breath due to the heavy bleeding are also common.
Related Links:
Minerva Surgical
Minerva video
The Minerva Surgical Endometrial Ablation System uses three separate ablation mechanisms to provide a safer and more effective means of treating excessive menstrual bleeding. The system is based on an impedance controlled treatment cycle specific to individual patient conditions. The first ablation step uses bipolar radiofrequency (RF) energy to ablate the endometrium. The second step uses the high voltage RF electrical current to ionize fully contained and circulated argon gas within a plasma formation array (PFA) sealed silicone membrane.
When the stretchable silicone membrane is deployed it conforms to the uterus; once energized, the argon gas is ionized, turning it in to a stream of plasma which heats the interior surface of the silicone membrane. The thermal energy is then conducted uniformly to the tissue in contact with the membrane. In the third step, released intra-cavitary fluids fills the gaps missed in the first two steps and further ablate remaining endometrial tissue. The system integration results in a faster procedure time and improved tissue contact.
Other features of the system include easy insertion, seating, and a non-stick atraumatic removal due to the lubricous nature of the silicone membrane array; a patented uterine integrity test (UIT) that automatically checks the integrity of the uterine cavity including the array membrane; and a cervical sealing balloon that positively seals the cervix canal near the internal os. The Endometrial Ablation System is a product of Minerva Surgical (Redwood City, CA, USA), and has been approved by the US Food and Drug Administration (FDA).
Menorrhagia, described as heavy and prolonged menstrual bleeding which disrupts a woman's normal activities, is one of the most common complaints in gynecology. Bleeding generally lasts for more than a week, requiring women to change their pad or tampon every two hours or less. Women with menorrhagia may also pass large blood clots and experience anemia due to the volume of blood loss. Constant lower abdominal and pelvic pain, tiredness, fatigue, and shortness of breath due to the heavy bleeding are also common.
Related Links:
Minerva Surgical
Minerva video
Latest Surgical Techniques News
- Fracture Plating System Combines Anatomical Fit with Streamlined Instrumentation
- Surgical Robotic System Gains CE Mark for Minimally Invasive Procedures
- Pink Noise Stimulation Approach Could Support Safer Anesthesia
- BD Launches Elyra Laser Platform for Kidney Stone and Soft Tissue Procedures
- Anesthesia-Sparing System Targets Faster Ureteral Stone Treatment
- Stretchable Bioelectronic Implant Lowers Blood Pressure in Preclinical Study
- FDA-Cleared Nerve Stimulator Advances Intraoperative Peripheral Nerve Assessment
- Intravascular Lithotripsy Catheter Advances Treatment of Calcified Coronary Disease
- Handheld AI Endomicroscope Enables Real-Time Precancer Detection at Point of Care
- Photoacoustic Imaging System Maps Hidden Nerves and Vessels in Robotic Laparoscopy
- Smart Soft Sensors Provide Real-Time Force Feedback in Minimally Invasive Surgery
- Advanced Endoscopy Platform Targets Challenging Upper GI Procedures
- Ultrasonic Fine-Needle Biopsy Tool Improves Salivary Gland Tumor Diagnosis
- Robotic Microsurgery System Gains CE Mark for Clinical Use in Europe
- New Endoscopic Device Offers Minimally Invasive Approach to Obesity Management
- Handheld Probe Enables Real-Time Tumor Mapping in Breast-Conserving Surgery
Channels
Artificial Intelligence
view channel
AI Tool Predicts Unplanned Care and Symptom Burden in Cancer Survivors
Unplanned emergency visits and hospitalizations remain common in cancer survivorship, when routine clinical contact often tapers while new symptoms emerge. These events reflect unmet needs and disrupt... Read more
Automated Phone Speech Test Identifies Alzheimer’s Pathology for Prescreening
Alzheimer’s disease assessment and trial recruitment often rely on costly, invasive biomarker testing and clinic-based cognitive evaluations, limiting scalability as populations age. Providers and trial... Read moreCritical Care
view channel
Ring-Type Cuffless Monitor Becomes First Added to Official Hypertension Guidelines
Detecting nocturnal and morning hypertension often requires out-of-office assessment, but conventional cuff-type monitors can disrupt sleep. New national guidance in South Korea expands 24-hour monitoring... Read more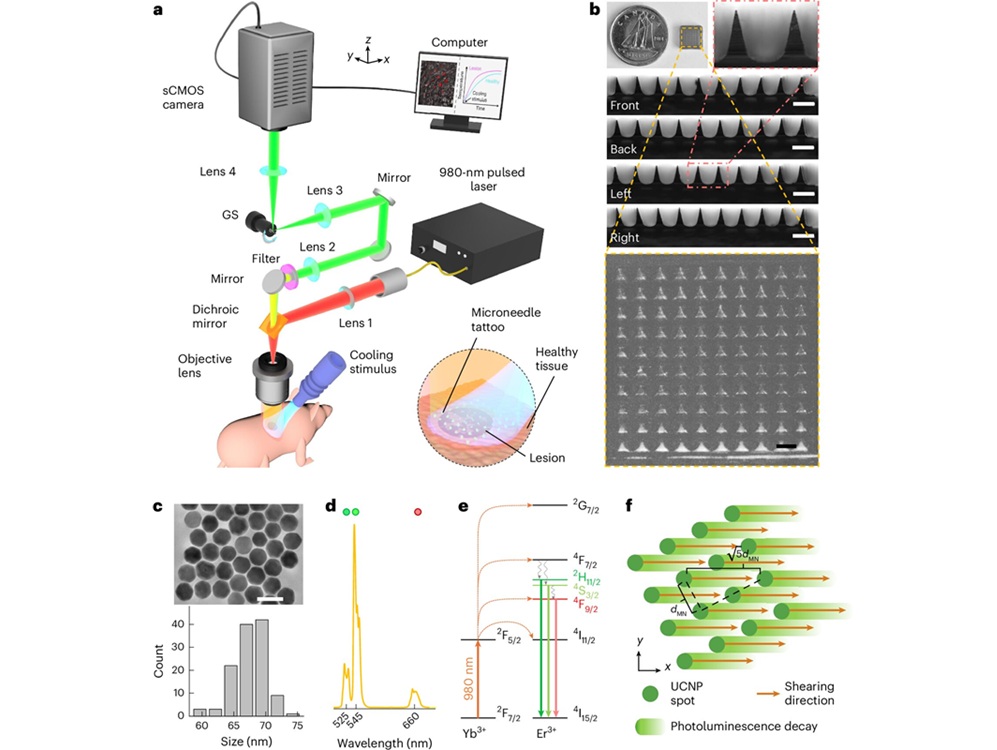
“Intelligent Tattoo” Method Detects Early Melanoma Signals
Melanoma is a lethal skin cancer in which earlier detection drives better outcomes. Current evaluation relies on visual inspection followed by biopsy, which can miss nascent lesions and lead to unnecessary... Read more
Reusable Intermittent Catheters Reduce Antibiotic Use Without Increasing Urinary Tract Infections
Intermittent self-catheterization, used to empty the bladder several times a day, can leave patients vulnerable to recurrent urinary tract infections and repeated antibiotic use. Reliance on single-use... Read morePatient Care
view channel
AI Avatar Doctor Improves Patient Understanding Before Radiotherapy
Radiation oncology consultations require patients to grasp complex concepts quickly, yet anxiety and information overload often undermine understanding and informed consent. Poor comprehension can also... Read more
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read moreHealth IT
view channel
AI-Native EHR Achieves EU Medical Device Certification
InterSystems (Boston, MA, USA) announced that its IntelliCare electronic health record (EHR) solutions have been certified as Class IIa medical devices under the European Union Medical Device Regulation... Read more
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read morePoint of Care
view channel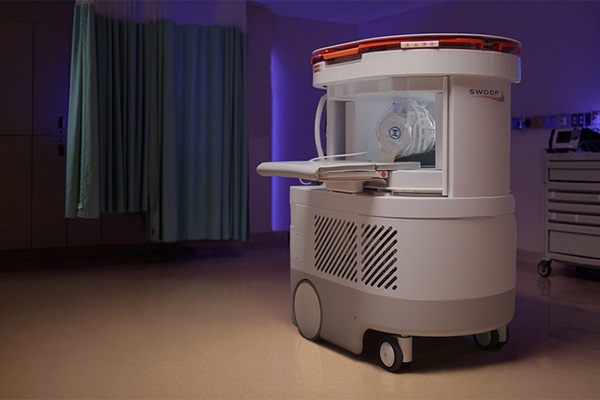
Portable MRI System Accelerates Emergency Brain Imaging and Triage
Emergency departments frequently face delays accessing conventional magnetic resonance imaging (MRI) for patients with suspected neurological emergencies. Such waits can slow triage, prolong boarding,... Read more