Wearable Defibrillator Protects Children at Risk
|
By HospiMedica International staff writers Posted on 28 Dec 2015 |
A novel wearable defibrillator now offers an advanced protection and monitoring option for children at risk of sudden cardiac arrest.
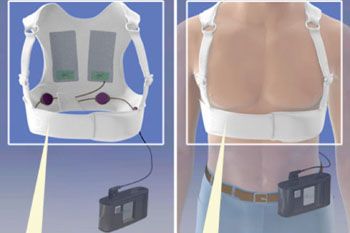
The LifeVest is a wearable defibrillator, which unlike an implantable cardioverter defibrillator (ICD) is worn on the body (under clothing), rather than implanted into it. Weighing less than one kilogram, the device consists of two main components: an electrode belt and vest-like garment that surround the patient’s chest, and a monitor that the patient wears around the waist. The pediatric device is intended only for children weighing at least 18 kg and with a chest size of 66 cm or more, which is about the size of an average eight year old.
The device continuously monitors the patient's heart using dry, non-adhesive sensing electrodes that detect abnormal heart rhythms. If such a life-threatening cardiac rhythm is detected, the device alerts the patient prior to delivering a shock, thus allowing a conscious patient to postpone the treatment shock. If, on the other hand, the patient is unconscious, the device releases a special gel over the therapy electrodes and delivers an electrical shock to restore normal rhythm.
The LifeVest Wearable Cardioverter Defibrillator is a product of Zoll Medical Corporation (Zoll; Chelmsford, MA, USA), and has been approved by the US Food and Drug Administration (FDA) for children who are at risk for sudden cardiac arrest, but are not candidates for an ICD due to certain medical conditions or lack of parental consent. The device was previously approved in 2001 for patients 18 years of age and older.
“The pediatric medical community is often forced to use adult devices off-label without appropriate labeling or instructions for use in pediatric patients,” said Vasum Peiris, MD, MPH, chief medical officer of pediatrics and special populations in the FDA Center for Devices and Radiological Health (CDRH). “Doctors now have important information that may help them safely prescribe this life-saving device to young patients who may benefit from the device.”
Ventricular fibrillation (VF) and ventricular tachycardia (VT) are life-threatening abnormal heart rhythms that are the most common cause of sudden cardiac arrest (SCA), according to the US National Heart, Lung and Blood Institute (Bethesda, MD, USA). Certain diseases and conditions that can lead to SCA include heart disease, inherited disorders, and structural changes in the heart due to infection or congenital heart disease. Most people who have SCA die from it, often within minutes. Rapid treatment with a defibrillator can save lives.
Related Links:
Zoll Medical Corporation
US National Heart, Lung and Blood Institute
The LifeVest is a wearable defibrillator, which unlike an implantable cardioverter defibrillator (ICD) is worn on the body (under clothing), rather than implanted into it. Weighing less than one kilogram, the device consists of two main components: an electrode belt and vest-like garment that surround the patient’s chest, and a monitor that the patient wears around the waist. The pediatric device is intended only for children weighing at least 18 kg and with a chest size of 66 cm or more, which is about the size of an average eight year old.
The device continuously monitors the patient's heart using dry, non-adhesive sensing electrodes that detect abnormal heart rhythms. If such a life-threatening cardiac rhythm is detected, the device alerts the patient prior to delivering a shock, thus allowing a conscious patient to postpone the treatment shock. If, on the other hand, the patient is unconscious, the device releases a special gel over the therapy electrodes and delivers an electrical shock to restore normal rhythm.
The LifeVest Wearable Cardioverter Defibrillator is a product of Zoll Medical Corporation (Zoll; Chelmsford, MA, USA), and has been approved by the US Food and Drug Administration (FDA) for children who are at risk for sudden cardiac arrest, but are not candidates for an ICD due to certain medical conditions or lack of parental consent. The device was previously approved in 2001 for patients 18 years of age and older.
“The pediatric medical community is often forced to use adult devices off-label without appropriate labeling or instructions for use in pediatric patients,” said Vasum Peiris, MD, MPH, chief medical officer of pediatrics and special populations in the FDA Center for Devices and Radiological Health (CDRH). “Doctors now have important information that may help them safely prescribe this life-saving device to young patients who may benefit from the device.”
Ventricular fibrillation (VF) and ventricular tachycardia (VT) are life-threatening abnormal heart rhythms that are the most common cause of sudden cardiac arrest (SCA), according to the US National Heart, Lung and Blood Institute (Bethesda, MD, USA). Certain diseases and conditions that can lead to SCA include heart disease, inherited disorders, and structural changes in the heart due to infection or congenital heart disease. Most people who have SCA die from it, often within minutes. Rapid treatment with a defibrillator can save lives.
Related Links:
Zoll Medical Corporation
US National Heart, Lung and Blood Institute
Read the full article by registering today, it's FREE! 

Register now for FREE to HospiMedica.com and get access to news and events that shape the world of Hospital Medicine. 
- Free digital version edition of HospiMedica International sent by email on regular basis
- Free print version of HospiMedica International magazine (available only outside USA and Canada).
- Free and unlimited access to back issues of HospiMedica International in digital format
- Free HospiMedica International Newsletter sent every week containing the latest news
- Free breaking news sent via email
- Free access to Events Calendar
- Free access to LinkXpress new product services
- REGISTRATION IS FREE AND EASY!
Sign in: Registered website members
Sign in: Registered magazine subscribers
Latest Critical Care News
- AI Tool Predicts In-Hospital Cardiac Arrest Minutes in Advance
- Magnetic Control System Enables Precise Navigation of Miniature Medical Devices
- Noninvasive AI Test Aims to Enable Earlier Pulmonary Hypertension Detection
- Blood-Filtering Device May Prolong Pregnancy in Severe Early Preeclampsia
- Noninvasive Skull Sensor Detects Early Deterioration in Acute Brain Injury
- Automated Robotic Imaging System Enhances Full-Body Skin Cancer Screening
- Bacteria-Resistant Urinary Catheter Coating Reduces Infections and Antibiotic Use
- New Brain-Computer Interface Targets Treatment-Resistant Depression
- Sensor-Integrated Airway Stent Enables Continuous Remote Monitoring
- Graphene-Based Material Selectively Eliminates Bacteria While Sparing Human Cells
- Flexible Plastic Film Uses Nanostructures to Destroy Viruses
- Finger Cuff Algorithm Enables Noninvasive Screening for Aortic Stenosis
- Vibration-Based Tools Aim to Improve Newborn Screening for Heart Defects
- Dried Platelet Biologic Reduces Hemorrhage After Traumatic Brain Injury
- Minimally Invasive Probe Measures Key Metabolic Markers Simultaneously
- AI Expands Across Heart Failure Care Continuum to Improve Management
Channels
Artificial Intelligence
view channel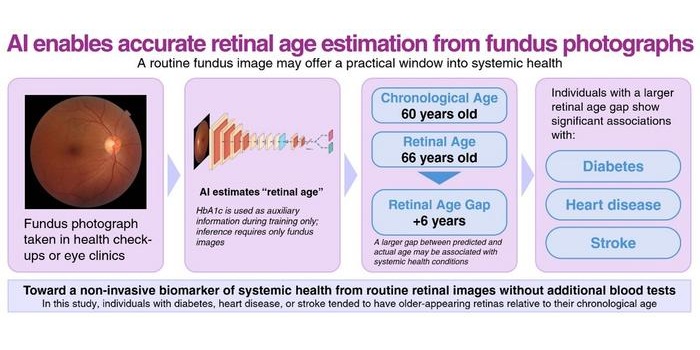
AI Model Uses Eye Imaging to Identify Risk of Major Systemic Diseases
Early detection of systemic disease risk remains a persistent challenge in population health screening. Cardiometabolic conditions such as diabetes, heart disease, and stroke often progress without symptoms... Read more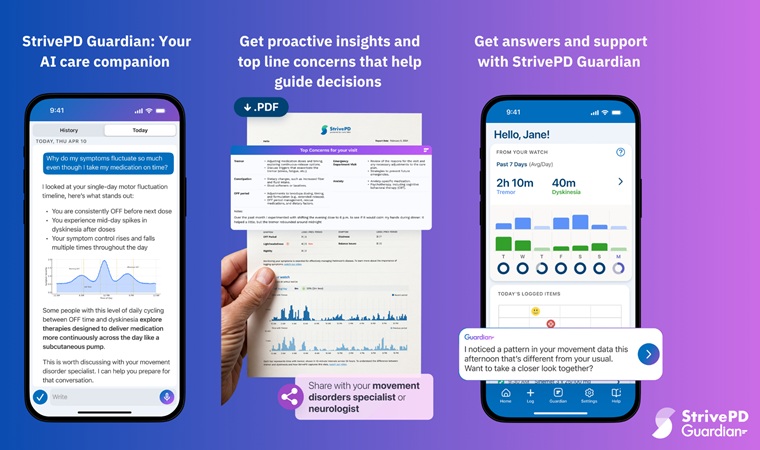
AI Platform Interprets Real-Time Wearable Data for Parkinson’s Management
Parkinson’s disease presents fluctuating motor and non-motor symptoms that complicate day-to-day self-management and clinical decision-making. Care teams require timely, longitudinal insight into medication... Read moreSurgical Techniques
view channel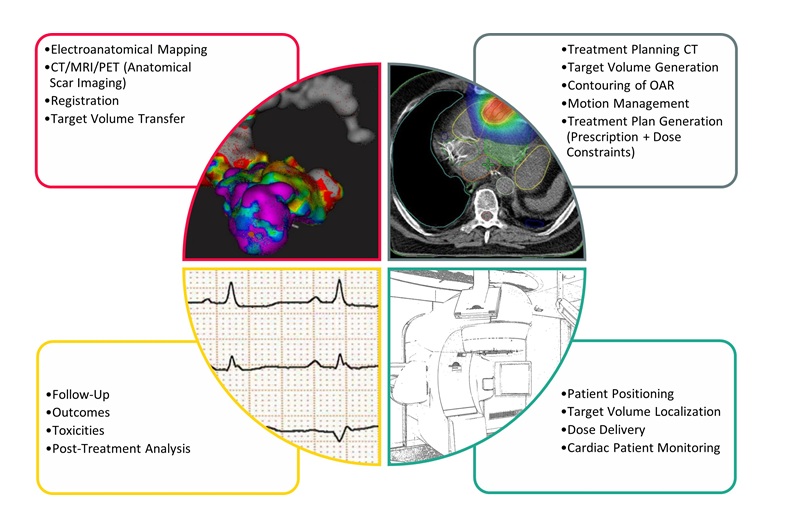
Noninvasive Cardiac Radiotherapy Reduces Ventricular Tachycardia Events
Life-threatening ventricular tachycardia is a rapid heart rhythm originating in the ventricles that can trigger sudden cardiac death. Patients who continue to have ventricular tachycardia despite guideline-based... Read more
Augmented Reality System for Knee Replacement Receives FDA Clearance
Total knee arthroplasty requires precise implant alignment while maintaining efficient, reproducible workflows. Many operating rooms, particularly ambulatory surgical centers, need guidance technologies... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel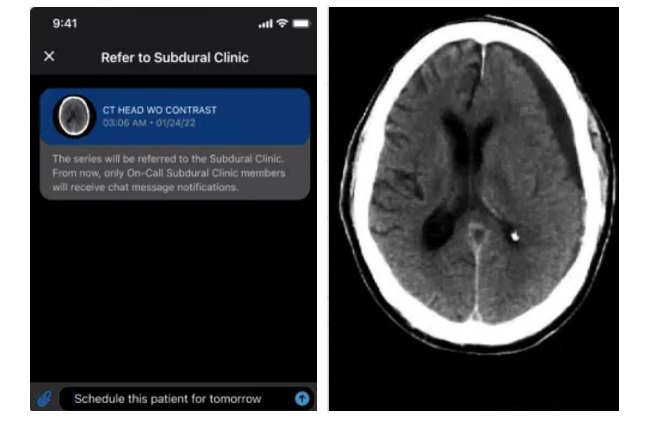
AI System Detects and Quantifies Chronic Subdural Hematoma
Viz.ai (San Francisco, CA, USA) announced a strategic commercialization collaboration with Johnson & Johnson (New Brunswick, NJ, USA) to expand access in the United States to the Viz Subdural solution... Read more
Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
Patients leaving skilled nursing facilities often lose continuous physiologic monitoring, increasing the risk of undetected infection and delayed intervention. Nursing home residents are seven times more... Read more
Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
Cardiogenic shock remains a difficult, time-sensitive emergency, with delayed identification driving poor outcomes and persistently high mortality. Many cases go undocumented even at advanced stages, hindering... Read morePoint of Care
view channelBusiness
view channel
Johnson & Johnson Launches AI-Driven Cardiac Mapping System
Johnson & Johnson has introduced the CARTOSOUND SONATA Module for the CARTO System at the Heart Rhythm Society (HRS) 2026 meeting in Chicago. The module uses artificial intelligence with the CARTO... Read more