Innovative ICDs Automatically Adapt to MRI Settings
|
By HospiMedica International staff writers Posted on 17 Feb 2016 |
A new line of implantable cardioverter defibrillators (ICDs) and cardiac resynchronization devices (CRT-Ds) can automatically detect MRI environments.
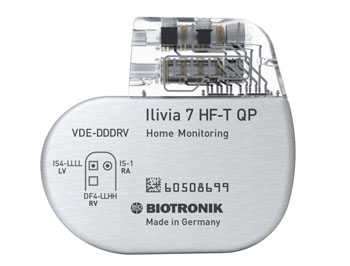
The Biotronik Ilivia ICDs and CRT-Ds feature both ProMRI, a technology that allows patients with a cardiac system to safely access MRI scanning, and MRI AutoDetect, which allows the cardiologist to activate a time window lasting up to 14 days, during which all device functionality is maintained until the patient undergoes the actual scan; at that point the device automatically switches into MRI mode. Once the scan is completed, all device functionality is once again restored automatically, all without the need for the cardiologist’s intervention.
Soon after the scan, the cardiologist receives a full report via Biotronik Home Monitoring, a system designed to replace device interrogation with customizable fully programmable alerts, with options to specify alert thresholds and ranges, and call-back notifications that direct the patient to contact the clinic. Other features include MultiPole Pacing (MPP) to improve therapy for CRT patients, a closed loop stimulation (CLS) algorithm for more adaptive pacing, and DX technology for ICDs and CRT-Ds. Biotronik Ilivia ICDs and CRT-Ds are products of Biotronik (Berlin, Germany).
“MRI AutoDetect will not only reduce the amount of time the patient’s device stays in MRI mode, but I expect it will also greatly improve the workflow between the cardiologist and radiologist, which will ultimately benefit our patients,” said Manuel Ortega, senior vice president of Biotronik. “MRI AutoDetect is the next step in ensuring that patients get the full benefit of both their implanted devices and diagnostic imaging.”
“Patients aren’t fully protected if they have an event while their devices are programmed in MRI mode, a period that currently can last a day or more depending on hospital workflows,” said Richard Kobza, MD, head of cardiology at Luzerner Kantonsspital (Lucerne, Switzerland). “Reducing the amount of time a device is in MRI mode is particularly crucial for ICD and CRT-D patients, and with MRI AutoDetect the only time these patients won’t be able to benefit from full device therapy is the short 30-minute window they spend in the MRI machine itself.”
At present, when a cardiac device patient needs to undergo a MRI scan, a cardiologist must program his or her device into a special mode that temporarily reduces the device’s functionality until the MRI scan is completed. Following the scan, the cardiologist must switch off the device’s MRI mode to allow the patient to once again have the full therapeutic benefits of their implanted device.
Related Links:
Biotronik
The Biotronik Ilivia ICDs and CRT-Ds feature both ProMRI, a technology that allows patients with a cardiac system to safely access MRI scanning, and MRI AutoDetect, which allows the cardiologist to activate a time window lasting up to 14 days, during which all device functionality is maintained until the patient undergoes the actual scan; at that point the device automatically switches into MRI mode. Once the scan is completed, all device functionality is once again restored automatically, all without the need for the cardiologist’s intervention.
Soon after the scan, the cardiologist receives a full report via Biotronik Home Monitoring, a system designed to replace device interrogation with customizable fully programmable alerts, with options to specify alert thresholds and ranges, and call-back notifications that direct the patient to contact the clinic. Other features include MultiPole Pacing (MPP) to improve therapy for CRT patients, a closed loop stimulation (CLS) algorithm for more adaptive pacing, and DX technology for ICDs and CRT-Ds. Biotronik Ilivia ICDs and CRT-Ds are products of Biotronik (Berlin, Germany).
“MRI AutoDetect will not only reduce the amount of time the patient’s device stays in MRI mode, but I expect it will also greatly improve the workflow between the cardiologist and radiologist, which will ultimately benefit our patients,” said Manuel Ortega, senior vice president of Biotronik. “MRI AutoDetect is the next step in ensuring that patients get the full benefit of both their implanted devices and diagnostic imaging.”
“Patients aren’t fully protected if they have an event while their devices are programmed in MRI mode, a period that currently can last a day or more depending on hospital workflows,” said Richard Kobza, MD, head of cardiology at Luzerner Kantonsspital (Lucerne, Switzerland). “Reducing the amount of time a device is in MRI mode is particularly crucial for ICD and CRT-D patients, and with MRI AutoDetect the only time these patients won’t be able to benefit from full device therapy is the short 30-minute window they spend in the MRI machine itself.”
At present, when a cardiac device patient needs to undergo a MRI scan, a cardiologist must program his or her device into a special mode that temporarily reduces the device’s functionality until the MRI scan is completed. Following the scan, the cardiologist must switch off the device’s MRI mode to allow the patient to once again have the full therapeutic benefits of their implanted device.
Related Links:
Biotronik
Read the full article by registering today, it's FREE! 

Register now for FREE to HospiMedica.com and get access to news and events that shape the world of Hospital Medicine. 
- Free digital version edition of HospiMedica International sent by email on regular basis
- Free print version of HospiMedica International magazine (available only outside USA and Canada).
- Free and unlimited access to back issues of HospiMedica International in digital format
- Free HospiMedica International Newsletter sent every week containing the latest news
- Free breaking news sent via email
- Free access to Events Calendar
- Free access to LinkXpress new product services
- REGISTRATION IS FREE AND EASY!
Sign in: Registered website members
Sign in: Registered magazine subscribers
Latest Critical Care News
- Bedside CSF Monitor Detects Early Infection in Fluid Drains
- Wearable Ultrasound Patch Noninvasively Paces Heart to Stabilize Arrhythmias
- AI ECG Tool Detects Cardiac Amyloidosis for Early Screening
- Cuffless Wearable Enables Continuous Blood Pressure Monitoring for Hypertension Care
- AI-Guided System Supports Cardiac Ultrasound Training on Cart-Based Systems
- AI ECG Index Tracks Pubertal Maturation in Children and Adolescents
- Noninvasive AI Tool Enables Pressure-Guided Heart Failure Management
- Regenerative Therapies Aim to Support Recovery After Traumatic Brain Injury
- Ring-Type Cuffless Monitor Becomes First Added to Official Hypertension Guidelines
- “Intelligent Tattoo” Method Detects Early Melanoma Signals
- Implantable Wireless Light Device Advances Bladder Cancer Treatment
- Reusable Intermittent Catheters Reduce Antibiotic Use Without Increasing Urinary Tract Infections
- Smart Wristband Technology Detects Cardiac Arrest and Alerts Responders
- FDA-Cleared Home Sleep Test Enables Multi-Night Diagnosis of Sleep Apnea
- AI-Enabled Wearable Patches Reveal Undetected Hormone Disruption in Infertility
- AI Method Turns Toe Scan into Rapid PAD Screening Tool
Channels
Artificial Intelligence
view channel
AI Platform Supports Noninvasive Remote Hemodynamic Monitoring in Heart Failure
Heart failure remains a leading cause of hospitalization in adults over 65, affecting more than 6.7 million people in the U.S. Clinicians often lose visibility into hemodynamic deterioration once patients... Read more
AI Tool Predicts Unplanned Care and Symptom Burden in Cancer Survivors
Unplanned emergency visits and hospitalizations remain common in cancer survivorship, when routine clinical contact often tapers while new symptoms emerge. These events reflect unmet needs and disrupt... Read moreSurgical Techniques
view channel
3D Map of Heart Electrical Wiring Aims to Guide Congenital Heart Repair
Tetralogy of Fallot is one of the most common congenital heart problems and often requires surgery in infancy. Many survivors later develop conduction abnormalities because the cardiac electrical system... Read more
Small Cryoprobe Outperforms Forceps in Lung Biopsy Trial
Accurate diagnosis of pulmonary nodules, post-transplant allograft dysfunction, and diffuse parenchymal lung disease depends on high-quality tissue obtained during bronchoscopy. Conventional forceps biopsy... Read morePatient Care
view channel
AI Avatar Doctor Improves Patient Understanding Before Radiotherapy
Radiation oncology consultations require patients to grasp complex concepts quickly, yet anxiety and information overload often undermine understanding and informed consent. Poor comprehension can also... Read more
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read moreHealth IT
view channel
AI-Native EHR Achieves EU Medical Device Certification
InterSystems (Boston, MA, USA) announced that its IntelliCare electronic health record (EHR) solutions have been certified as Class IIa medical devices under the European Union Medical Device Regulation... Read more
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read morePoint of Care
view channel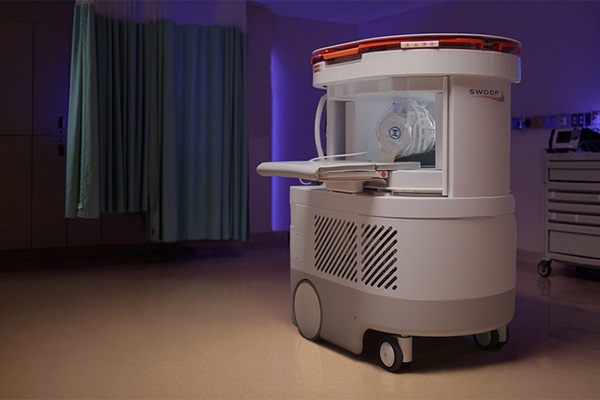
Portable MRI System Accelerates Emergency Brain Imaging and Triage
Emergency departments frequently face delays accessing conventional magnetic resonance imaging (MRI) for patients with suspected neurological emergencies. Such waits can slow triage, prolong boarding,... Read more