Ingestible Balloon Helps Patients Lose Weight
|
By HospiMedica International staff writers Posted on 04 Oct 2016 |

Image: An ingestible intragastric balloon system helps reduce weight (Photo courtesy of Obalon).
An ingestible intragastric balloon system facilitates weight loss in obese adults who failed to lose weight through diet and exercise.
The Obalon system consists of a balloon folded inside a capsule that is swallowed by the patient, with no sedation or anesthesia required. Once the balloon reaches the stomach, it is remotely inflated via an attached micro-catheter that is subsequently removed, leaving behind a buoyant balloon with a volume of 250cc (about the size of a small orange) that weighs less than six grams. Over the following three months of treatment, two additional balloons are swallowed and inflated, bringing the total maximum displaced volume to750cc.
The nonsurgical weight loss system is fully reversible throughout the course, and at the end of the six-month treatment period, all three balloons are mandatorily removed via endoscopy under conscious sedation. The system is intended for use as an adjunct to a moderate intensity diet and to a behavior modification program. The Obalon balloon system is a product of Obalon (Carlsbad, CA, USA), and has been approved by the U.S. Food and Drug Administration (FDA).
“We’re excited to bring this novel technology to the approximately 70 million adults in the United States who are struggling with obesity. For physicians, we believe that the Obalon technology represents an important new option for both their practices and their patients who are obese and looking to lose weight,” said Andy Rasdal, CEO of Obalon. “I’d also like to recognize the FDA for their efficient and interactive review of our product, which represents another option for physicians and patients in the battle against the obesity epidemic.”
“The clinical trial design for the Obalon balloon system was highly rigorous and the system demonstrated a favorable safety profile,” said Shelby Sullivan, MD, director of the gastroenterology bariatric and metabolic program at the University of Colorado School of Medicine (Aurora, USA). “We were also pleased to see that patients tolerated the administrations of the Obalon balloon remarkably well, with no recovery time needed. This is in contrast to other currently approved intragastric balloon devices, where most patients cannot immediately return to normal activities.”
Related Links:
Obalon
The Obalon system consists of a balloon folded inside a capsule that is swallowed by the patient, with no sedation or anesthesia required. Once the balloon reaches the stomach, it is remotely inflated via an attached micro-catheter that is subsequently removed, leaving behind a buoyant balloon with a volume of 250cc (about the size of a small orange) that weighs less than six grams. Over the following three months of treatment, two additional balloons are swallowed and inflated, bringing the total maximum displaced volume to750cc.
The nonsurgical weight loss system is fully reversible throughout the course, and at the end of the six-month treatment period, all three balloons are mandatorily removed via endoscopy under conscious sedation. The system is intended for use as an adjunct to a moderate intensity diet and to a behavior modification program. The Obalon balloon system is a product of Obalon (Carlsbad, CA, USA), and has been approved by the U.S. Food and Drug Administration (FDA).
“We’re excited to bring this novel technology to the approximately 70 million adults in the United States who are struggling with obesity. For physicians, we believe that the Obalon technology represents an important new option for both their practices and their patients who are obese and looking to lose weight,” said Andy Rasdal, CEO of Obalon. “I’d also like to recognize the FDA for their efficient and interactive review of our product, which represents another option for physicians and patients in the battle against the obesity epidemic.”
“The clinical trial design for the Obalon balloon system was highly rigorous and the system demonstrated a favorable safety profile,” said Shelby Sullivan, MD, director of the gastroenterology bariatric and metabolic program at the University of Colorado School of Medicine (Aurora, USA). “We were also pleased to see that patients tolerated the administrations of the Obalon balloon remarkably well, with no recovery time needed. This is in contrast to other currently approved intragastric balloon devices, where most patients cannot immediately return to normal activities.”
Related Links:
Obalon
Latest Critical Care News
- Magnetic Control System Enables Precise Navigation of Miniature Medical Devices
- Noninvasive Skull Sensor Detects Early Deterioration in Acute Brain Injury
- Bacteria-Resistant Urinary Catheter Coating Reduces Infections and Antibiotic Use
- Sensor-Integrated Airway Stent Enables Continuous Remote Monitoring
- Graphene-Based Material Selectively Eliminates Bacteria While Sparing Human Cells
- Flexible Plastic Film Uses Nanostructures to Destroy Viruses
- Finger Cuff Algorithm Enables Noninvasive Screening for Aortic Stenosis
- Vibration-Based Tools Aim to Improve Newborn Screening for Heart Defects
- Dried Platelet Biologic Reduces Hemorrhage After Traumatic Brain Injury
- Minimally Invasive Probe Measures Key Metabolic Markers Simultaneously
- AI Expands Across Heart Failure Care Continuum to Improve Management
- Wearable Cardiac Monitor with Cellular Transmission Enhances Remote Arrhythmia Detection
- Microneedle Sensor Enables Continuous Monitoring of Drug Clearance
- 3D Brain Imaging Marker Predicts Surgical Outcomes in Dementia-Causing Hydrocephalus
- Printed Artificial Neurons Stimulate Natural Brain Circuits
- Review Advances Precision Care Pathway for Meningioma Management
Channels
Artificial Intelligence
view channel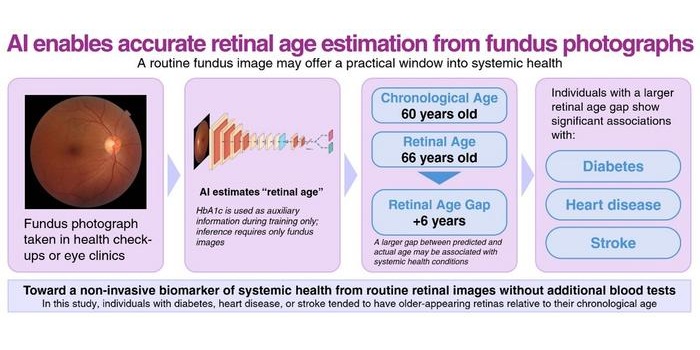
AI Model Uses Eye Imaging to Identify Risk of Major Systemic Diseases
Early detection of systemic disease risk remains a persistent challenge in population health screening. Cardiometabolic conditions such as diabetes, heart disease, and stroke often progress without symptoms... Read more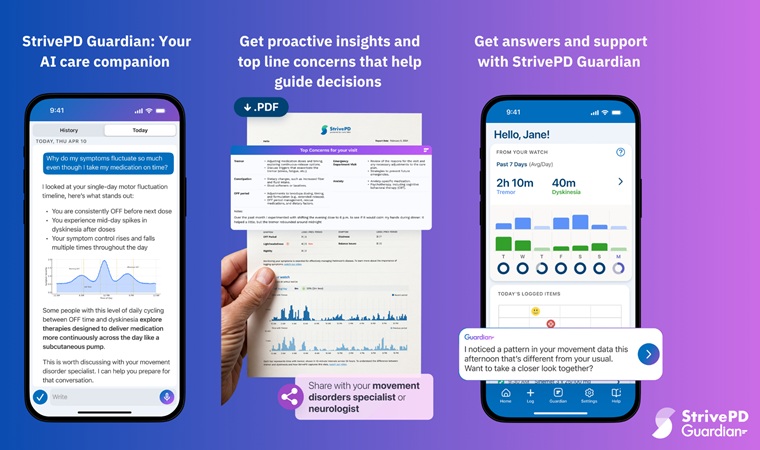
AI Platform Interprets Real-Time Wearable Data for Parkinson’s Management
Parkinson’s disease presents fluctuating motor and non-motor symptoms that complicate day-to-day self-management and clinical decision-making. Care teams require timely, longitudinal insight into medication... Read moreSurgical Techniques
view channel
New AI Tool Predicts Complications Before Lung Cancer Surgery
Lung cancer remains a leading cause of cancer mortality, and many surgical candidates present with complex comorbidities. Postoperative complications are common, making accurate and individualized risk... Read more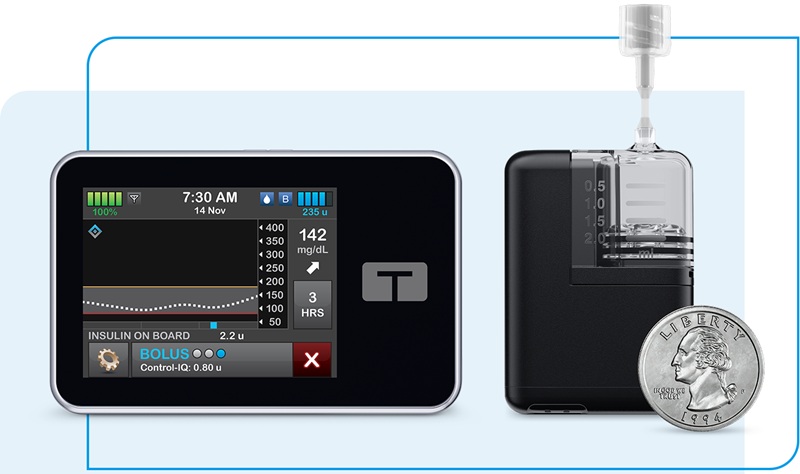
First Automated Insulin Delivery System Cleared for Type 1 Diabetes in Pregnancy
Managing glycemia during pregnancy complicated by type 1 diabetes requires tight targets and sustained time in a pregnancy-specific glucose range. Clinicians and patients often rely on automated insulin... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel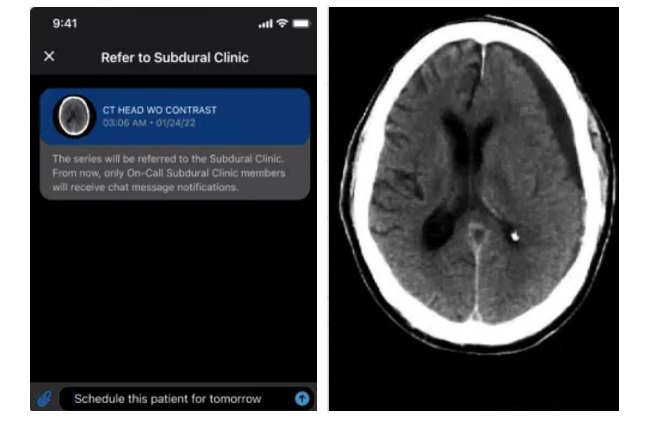
AI System Detects and Quantifies Chronic Subdural Hematoma
Viz.ai (San Francisco, CA, USA) announced a strategic commercialization collaboration with Johnson & Johnson (New Brunswick, NJ, USA) to expand access in the United States to the Viz Subdural solution... Read more
Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
Patients leaving skilled nursing facilities often lose continuous physiologic monitoring, increasing the risk of undetected infection and delayed intervention. Nursing home residents are seven times more... Read more
Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
Cardiogenic shock remains a difficult, time-sensitive emergency, with delayed identification driving poor outcomes and persistently high mortality. Many cases go undocumented even at advanced stages, hindering... Read morePoint of Care
view channelBusiness
view channel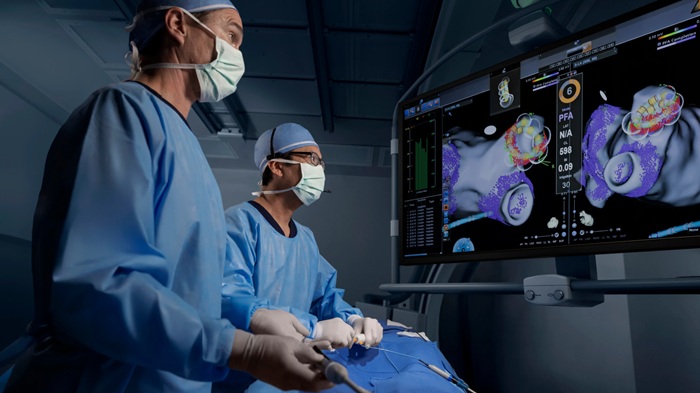
Johnson & Johnson Launches AI-Driven Cardiac Mapping System
Johnson & Johnson has introduced the CARTOSOUND SONATA Module for the CARTO System at the Heart Rhythm Society (HRS) 2026 meeting in Chicago. The module uses artificial intelligence with the CARTO... Read more