Pediatric Breathalyzer Identifies Disease by Analyzing Exhalations
|
By HospiMedica International staff writers Posted on 19 Oct 2016 |
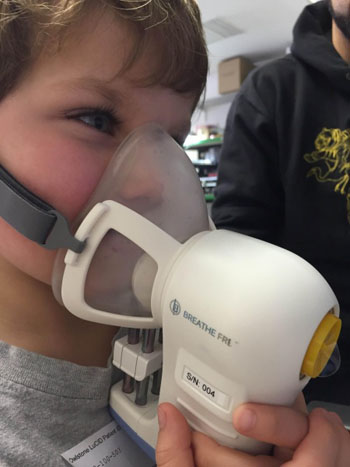
Image: The ReCIVA breathalyzer (Photo courtesy of Owlstone Medical).
A novel breath-testing device aids early stage diagnostics and therapy response in both adults and child asthma patients.
The Respiration Collector for In Vitro Analysis (ReCIVA) device is designed to accurately and selectively detect volatile organic compounds (VOCs) in breath that have been shown to correlate to inflammatory subtype in asthma, and can thus help guide better treatment decisions. ReCIVA is used in combination with the proprietary field asymmetric ion mobility spectrometer (FAIMS) sensor platform, a technology that can be programmed in software to detect targeted biomarkers of disease not only in breath, but in bodily fluids as well.
FAIMS can be also used in clinical diagnostics and precision medicine, with applications both in cancer and in a wide range of other medical conditions. The highly sensitive and selective tests allow for early diagnosis when treatment is more effective, resulting in more lives saved. The ReCIVA breathalyzer platform was adapted to a pediatric version intended for children five years of ages and up, offering a completely non-invasive way to test children’s breath.
Both the adult and pediatric versions of the breathalyzer have been implemented in the East Midlands Breathomics Pathology Node (EMBER), a £2.5 million project with the primary aims being development of breath-based systems for molecular pathology of disease, and clinically validating breathomics as a viable diagnostic modality. The ReCIVA breathalyzer is a product of Owlstone Medical (Cambridge, United Kingdom), and has received the European Community CE mark of approval.
“Breath analysis presents a significant opportunity to better predict how a child will respond to certain treatments, including steroids and expensive biologics,” said Billy Boyle, co-founder and CEO of Owlstone Medical. “In developing a pediatric breath sampler we are expanding the scope of projects such as EMBER to include children as an important group in the study of asthma, and more generally extending other biomarker and discovery studies using breath.”
“Choosing the right treatment for the right patient is especially challenging in children, as obtaining samples such as blood to measure disease activity can be difficult,” said professor of respiratory medicine Chris Brightling, PhD, of the University of Leicester (United Kingdom) and EMBER. “Breath analysis offers an excellent opportunity to sample the airway by simply breathing into a mask. This presents a new approach to understand disease and make better treatment decisions.”
Related Links:
Owlstone Medical
The Respiration Collector for In Vitro Analysis (ReCIVA) device is designed to accurately and selectively detect volatile organic compounds (VOCs) in breath that have been shown to correlate to inflammatory subtype in asthma, and can thus help guide better treatment decisions. ReCIVA is used in combination with the proprietary field asymmetric ion mobility spectrometer (FAIMS) sensor platform, a technology that can be programmed in software to detect targeted biomarkers of disease not only in breath, but in bodily fluids as well.
FAIMS can be also used in clinical diagnostics and precision medicine, with applications both in cancer and in a wide range of other medical conditions. The highly sensitive and selective tests allow for early diagnosis when treatment is more effective, resulting in more lives saved. The ReCIVA breathalyzer platform was adapted to a pediatric version intended for children five years of ages and up, offering a completely non-invasive way to test children’s breath.
Both the adult and pediatric versions of the breathalyzer have been implemented in the East Midlands Breathomics Pathology Node (EMBER), a £2.5 million project with the primary aims being development of breath-based systems for molecular pathology of disease, and clinically validating breathomics as a viable diagnostic modality. The ReCIVA breathalyzer is a product of Owlstone Medical (Cambridge, United Kingdom), and has received the European Community CE mark of approval.
“Breath analysis presents a significant opportunity to better predict how a child will respond to certain treatments, including steroids and expensive biologics,” said Billy Boyle, co-founder and CEO of Owlstone Medical. “In developing a pediatric breath sampler we are expanding the scope of projects such as EMBER to include children as an important group in the study of asthma, and more generally extending other biomarker and discovery studies using breath.”
“Choosing the right treatment for the right patient is especially challenging in children, as obtaining samples such as blood to measure disease activity can be difficult,” said professor of respiratory medicine Chris Brightling, PhD, of the University of Leicester (United Kingdom) and EMBER. “Breath analysis offers an excellent opportunity to sample the airway by simply breathing into a mask. This presents a new approach to understand disease and make better treatment decisions.”
Related Links:
Owlstone Medical
Latest Critical Care News
- Magnetic Control System Enables Precise Navigation of Miniature Medical Devices
- Noninvasive Skull Sensor Detects Early Deterioration in Acute Brain Injury
- Bacteria-Resistant Urinary Catheter Coating Reduces Infections and Antibiotic Use
- Sensor-Integrated Airway Stent Enables Continuous Remote Monitoring
- Graphene-Based Material Selectively Eliminates Bacteria While Sparing Human Cells
- Flexible Plastic Film Uses Nanostructures to Destroy Viruses
- Finger Cuff Algorithm Enables Noninvasive Screening for Aortic Stenosis
- Vibration-Based Tools Aim to Improve Newborn Screening for Heart Defects
- Dried Platelet Biologic Reduces Hemorrhage After Traumatic Brain Injury
- Minimally Invasive Probe Measures Key Metabolic Markers Simultaneously
- AI Expands Across Heart Failure Care Continuum to Improve Management
- Wearable Cardiac Monitor with Cellular Transmission Enhances Remote Arrhythmia Detection
- Microneedle Sensor Enables Continuous Monitoring of Drug Clearance
- 3D Brain Imaging Marker Predicts Surgical Outcomes in Dementia-Causing Hydrocephalus
- Printed Artificial Neurons Stimulate Natural Brain Circuits
- Review Advances Precision Care Pathway for Meningioma Management
Channels
Artificial Intelligence
view channel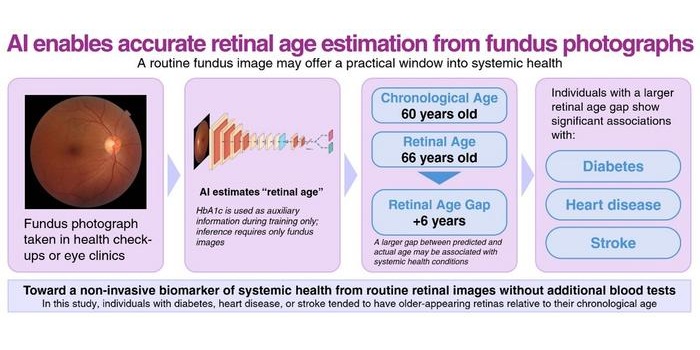
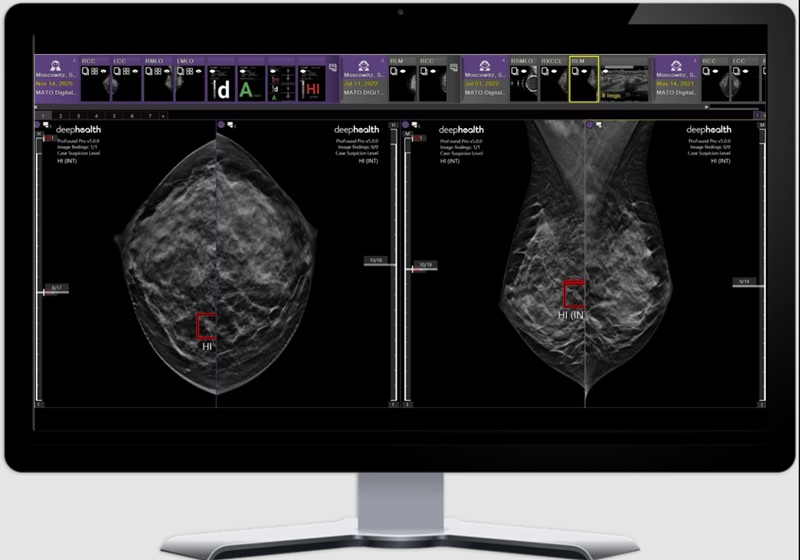
AI Model Uses Eye Imaging to Identify Risk of Major Systemic Diseases
Early detection of systemic disease risk remains a persistent challenge in population health screening. Cardiometabolic conditions such as diabetes, heart disease, and stroke often progress without symptoms... Read more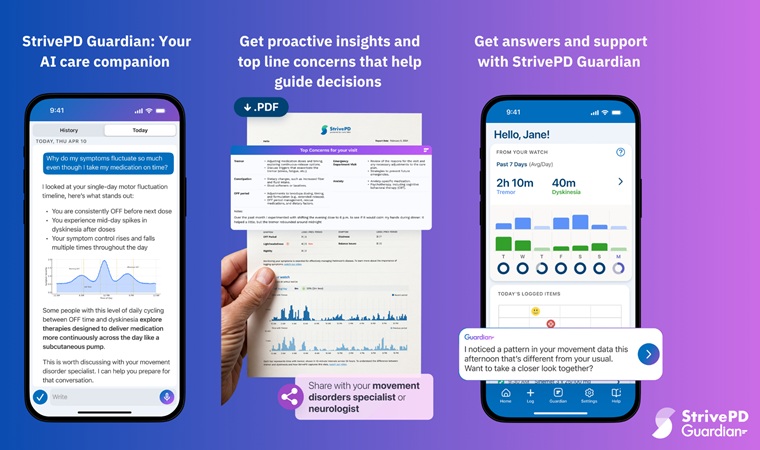
AI Platform Interprets Real-Time Wearable Data for Parkinson’s Management
Parkinson’s disease presents fluctuating motor and non-motor symptoms that complicate day-to-day self-management and clinical decision-making. Care teams require timely, longitudinal insight into medication... Read moreSurgical Techniques
view channel
New AI Tool Predicts Complications Before Lung Cancer Surgery
Lung cancer remains a leading cause of cancer mortality, and many surgical candidates present with complex comorbidities. Postoperative complications are common, making accurate and individualized risk... Read more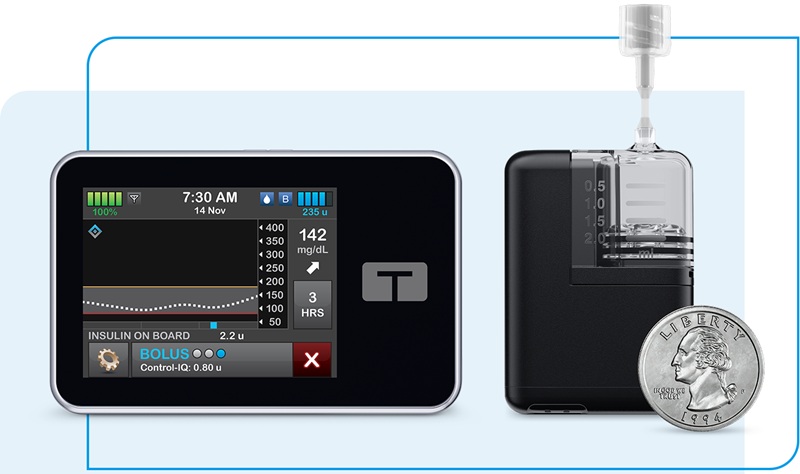
First Automated Insulin Delivery System Cleared for Type 1 Diabetes in Pregnancy
Managing glycemia during pregnancy complicated by type 1 diabetes requires tight targets and sustained time in a pregnancy-specific glucose range. Clinicians and patients often rely on automated insulin... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel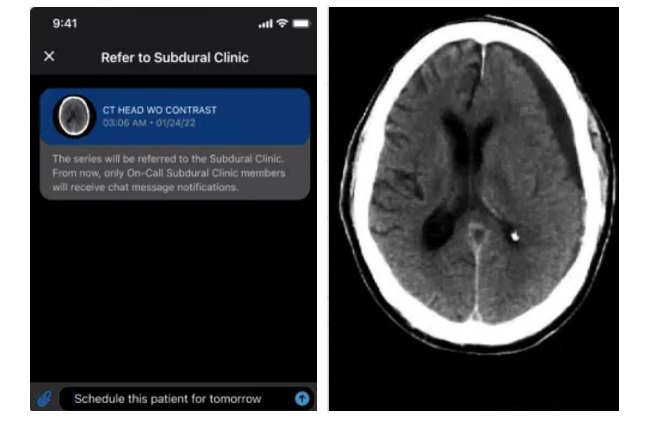
AI System Detects and Quantifies Chronic Subdural Hematoma
Viz.ai (San Francisco, CA, USA) announced a strategic commercialization collaboration with Johnson & Johnson (New Brunswick, NJ, USA) to expand access in the United States to the Viz Subdural solution... Read more
Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
Patients leaving skilled nursing facilities often lose continuous physiologic monitoring, increasing the risk of undetected infection and delayed intervention. Nursing home residents are seven times more... Read more
Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
Cardiogenic shock remains a difficult, time-sensitive emergency, with delayed identification driving poor outcomes and persistently high mortality. Many cases go undocumented even at advanced stages, hindering... Read morePoint of Care
view channelBusiness
view channel
Johnson & Johnson Launches AI-Driven Cardiac Mapping System
Johnson & Johnson has introduced the CARTOSOUND SONATA Module for the CARTO System at the Heart Rhythm Society (HRS) 2026 meeting in Chicago. The module uses artificial intelligence with the CARTO... Read more