Breakthrough Biodegradable Capsule Aids Hemophilia Treatment
|
By Daniel Beris Posted on 13 Dec 2016 |
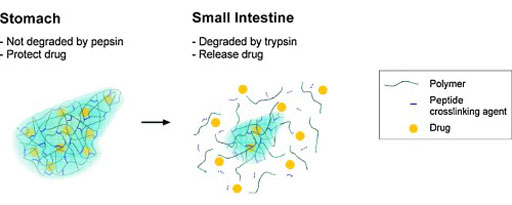
Image: A hemophilia capsule only degrades in the small intestine (Photo courtesy of UT Austin).
A new study describes how an innovative biodegradable capsule that uses hydrophilic carriers for oral delivery of hematological factor IX (hFIX) can treat hemophilia B.
Developed by researchers at the University of Texas (UT; Austin, USA), the oral delivery system is based on a pH-responsive polymer hydrogel microcarrier system that carries the prophylactic hFIX particles. The poly (methacrylic acid)-grafted-poly(ethylene glycol) [P(MAA-g-EG)] polymer, synthesized using ultraviolet (UV) polymerization, is subsequently joined with an enzymatically degradable peptide crosslinking agent that allows for site-specific degradation (by trypsin) in the small intestine.
As it moves through the body, the particle-containing capsule resists the major gastric enzyme in order to remain intact while in the stomach, protecting the encapsulated drug. When in the small intestine, the capsule begins to swell with the increase in pH, and is subsequently degraded by the minor intestinal enzyme, slowly releasing the drug over time. In study models, the system successfully transported hFIX, and was able to deliver adequate levels of the drug to the target site in the small intestine. The study was published on November 15, 2016, in the International Journal of Pharmaceutics.
“While an oral delivery platform will be beneficial to all hemophilia B patients, patients in developing countries will benefit the most,” said lead author Sarena Horava, PhD, of the UT Cockrell School of Engineering. “In many developing countries, the median life expectancy for hemophilia patients is 11 years due to the lack of access to treatment, but our new oral delivery of factor IX can now overcome these issues and improve the worldwide use of this therapy.”
Hemophilia B is a blood clotting disorder caused by a mutation of the factor IX gene, leading to a deficiency of factor IX. It is the second-most common form of hemophilia, and is sometimes called Christmas disease, after Stephen Christmas, the first patient described with the affliction. Presentation of hemophilia B is consistent with easy bruising, urinary tract bleed, and nosebleeds. Current protein replacement therapies rely on intravenous (IV) injections and infusions.
Related Links:
University of Texas
Developed by researchers at the University of Texas (UT; Austin, USA), the oral delivery system is based on a pH-responsive polymer hydrogel microcarrier system that carries the prophylactic hFIX particles. The poly (methacrylic acid)-grafted-poly(ethylene glycol) [P(MAA-g-EG)] polymer, synthesized using ultraviolet (UV) polymerization, is subsequently joined with an enzymatically degradable peptide crosslinking agent that allows for site-specific degradation (by trypsin) in the small intestine.
As it moves through the body, the particle-containing capsule resists the major gastric enzyme in order to remain intact while in the stomach, protecting the encapsulated drug. When in the small intestine, the capsule begins to swell with the increase in pH, and is subsequently degraded by the minor intestinal enzyme, slowly releasing the drug over time. In study models, the system successfully transported hFIX, and was able to deliver adequate levels of the drug to the target site in the small intestine. The study was published on November 15, 2016, in the International Journal of Pharmaceutics.
“While an oral delivery platform will be beneficial to all hemophilia B patients, patients in developing countries will benefit the most,” said lead author Sarena Horava, PhD, of the UT Cockrell School of Engineering. “In many developing countries, the median life expectancy for hemophilia patients is 11 years due to the lack of access to treatment, but our new oral delivery of factor IX can now overcome these issues and improve the worldwide use of this therapy.”
Hemophilia B is a blood clotting disorder caused by a mutation of the factor IX gene, leading to a deficiency of factor IX. It is the second-most common form of hemophilia, and is sometimes called Christmas disease, after Stephen Christmas, the first patient described with the affliction. Presentation of hemophilia B is consistent with easy bruising, urinary tract bleed, and nosebleeds. Current protein replacement therapies rely on intravenous (IV) injections and infusions.
Related Links:
University of Texas
Latest Critical Care News
- Low-Frequency Wireless Sensor Monitors Arterial Stiffening and Blood Pressure
- FDA-Cleared Transseptal Access Device Enables Site-Specific Left Atrial Puncture
- AI Tool Estimates CPAP Effect on Cardiovascular Risk in Sleep Apnea
- Wearable AI Tool Predicts Hospitalization Risk in Heart Failure
- Real-Time Imaging Guides CPR to Improve Perfusion
- AI Tool Predicts Post-Therapy Barrett’s Esophagus Recurrence
- New Technology Turns Earbuds into Sensors for Cardiac Function Tracking

- Wearable AI Tool Estimates Vascular Age for Cardiovascular Risk
- New Brain Stimulation Approach Targets Deep Brain Areas Without Surgery
- Injectable Microgel Reduces Blood Loss in Infant Surgery
- Standardized FMT Protocol May Improve Survival in Severe C. difficile Infection
- Heat-Activated Skin Patch Targets Melanoma Lesions
- Automated Dispensing System Enhances Medication Access and Efficiency
- Angiography-Based FFR Approach Matches Gold Standard Results Without Wires
- Eye Imaging AI Identifies Elevated Cardiovascular Risk
- Noninvasive Monitoring Device Enables Earlier Intervention in Heart Failure
Channels
Artificial Intelligence
view channel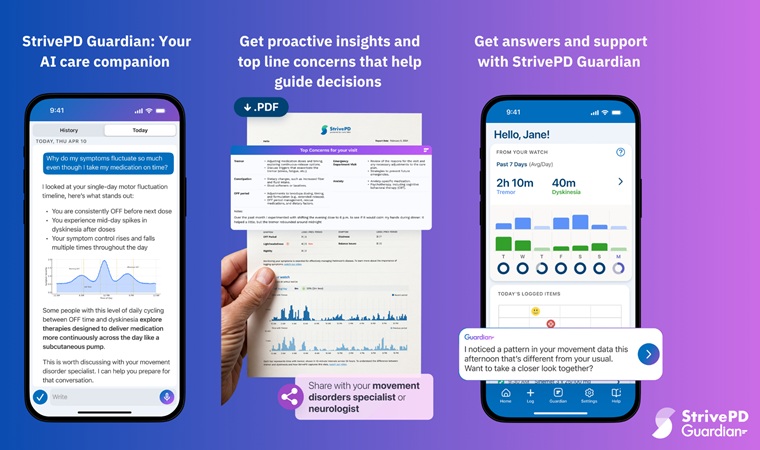
AI Platform Interprets Real-Time Wearable Data for Parkinson’s Management
Parkinson’s disease presents fluctuating motor and non-motor symptoms that complicate day-to-day self-management and clinical decision-making. Care teams require timely, longitudinal insight into medication... Read more
Algorithm Identifies Cardiac Arrest Hotspots to Guide AED Placement
Out-of-hospital sudden cardiac arrest is common and usually fatal, and survival depends on rapid defibrillation. Many communities deploy automated external defibrillators without precise guidance, which... Read moreSurgical Techniques
view channel
Spine Surgical Table and Smart Stretcher Streamline Positioning and Transfers
Spine procedures often require manual patient rotation and complex positioning that add time and physical strain for operating room teams. Emergency and procedural areas also face frequent transfers and... Read more
MRI-Guided Ultrasound Ablation Shows Faster Recovery Than Prostatectomy
Localized, intermediate-risk prostate cancer is commonly managed with robotic prostatectomy, yet surgery can carry short-term recovery burdens for patients. Faster convalescence and preservation of function... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
Cardiogenic shock remains a difficult, time-sensitive emergency, with delayed identification driving poor outcomes and persistently high mortality. Many cases go undocumented even at advanced stages, hindering... Read more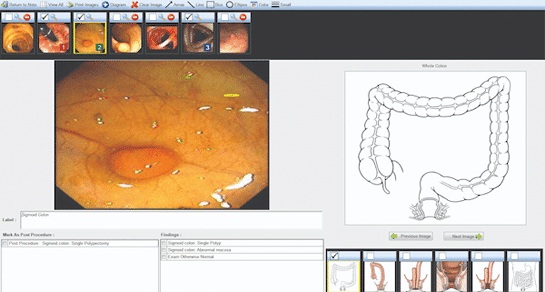
Voice-Driven AI System Enables Structured GI Procedure Documentation
Documentation during gastrointestinal (GI) procedures often competes with real-time clinical decision-making and imposes a significant cognitive burden on physicians. Manual data entry and post-procedure... Read more
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read morePoint of Care
view channelBusiness
view channel
New Partnership Advances Physical AI into Perioperative Workflows
Perioperative operations often span sterile processing, operating rooms, logistics, and equipment coordination across disconnected systems, teams, and data layers, creating coordination challenges.... Read more