Cervical Interbody Implants Treat Up to Four Levels
|
By HospiMedica International staff writers Posted on 11 Apr 2017 |

Image: The CoRoent Small Interbody System (Photo courtesy of NuVasive).
An innovative intervertebral body fusion system treats disc degeneration at up to four contiguous levels in the cervical spine.
The CoRoent Small Interbody System is an interbody cage manufactured from polyether ether ketone (PEEK) - Optima that provides stabilization of the spinal motion segment while fusion is taking place. A hollow core allows for packing of autogenous and/or allogeneic bone graft products, such as cancellous, cortical, and/or corticocancellous bone graft material in order to help promote a solid fusion. Rows of teeth on the surface of each end of the device serve to grip the adjacent vertebrae in order to resist migration and expulsion of the device.
Implants included in the system include the CoRoent Small (S), the CoRoent Small Lordotic (SL), the CoRoent Small Lordotic Plus (SLP), the CoRoent Small Hyperlordotic (SHL), and the CoRoent Small Contoured (SC) cages. The implants also include radiopaque marker pins composed of titanium alloy or tantalum. The CoRoent Small Interbody System is a product of NuVasive, and has been approved by the U.S. Food and Drug Administration (FDA).
“The ability to treat multi-level cervical disc degeneration in patients displaying cervical radiculopathy and myelopathy is a meaningful advancement for spine surgeons,” said Jason Hannon, president and CEO of NuVasive. “This first and only clearance is an example of NuVasive's commitment to defining the components necessary to properly address an unmet clinical need and expand our presence and competitive positioning in the cervical market.”
Interbody fusion devices are prostheses used in spinal fusion procedures to replace the intervertebral disc of the spine, enhancing stability in the region while the spine fuses by maintaining foraminal height and decompression. Once placed, the cages resist flexion and extension of the spine, as well as axial forces across the ventral and middle columns. Over time, the packed bone graft material is gradually replaced by natural bone, forming a solid section.
The CoRoent Small Interbody System is an interbody cage manufactured from polyether ether ketone (PEEK) - Optima that provides stabilization of the spinal motion segment while fusion is taking place. A hollow core allows for packing of autogenous and/or allogeneic bone graft products, such as cancellous, cortical, and/or corticocancellous bone graft material in order to help promote a solid fusion. Rows of teeth on the surface of each end of the device serve to grip the adjacent vertebrae in order to resist migration and expulsion of the device.
Implants included in the system include the CoRoent Small (S), the CoRoent Small Lordotic (SL), the CoRoent Small Lordotic Plus (SLP), the CoRoent Small Hyperlordotic (SHL), and the CoRoent Small Contoured (SC) cages. The implants also include radiopaque marker pins composed of titanium alloy or tantalum. The CoRoent Small Interbody System is a product of NuVasive, and has been approved by the U.S. Food and Drug Administration (FDA).
“The ability to treat multi-level cervical disc degeneration in patients displaying cervical radiculopathy and myelopathy is a meaningful advancement for spine surgeons,” said Jason Hannon, president and CEO of NuVasive. “This first and only clearance is an example of NuVasive's commitment to defining the components necessary to properly address an unmet clinical need and expand our presence and competitive positioning in the cervical market.”
Interbody fusion devices are prostheses used in spinal fusion procedures to replace the intervertebral disc of the spine, enhancing stability in the region while the spine fuses by maintaining foraminal height and decompression. Once placed, the cages resist flexion and extension of the spine, as well as axial forces across the ventral and middle columns. Over time, the packed bone graft material is gradually replaced by natural bone, forming a solid section.
Latest Surgical Techniques News
- Fiber-Form Bone Graft Expands Intraoperative Options for Spinal Fusion
- Ultrasound‑Aided Catheter Treatment Cuts Early Collapse in Pulmonary Embolism
- Ultrasound Technology Aims to Replace Invasive BPH Procedures
- Continuous Monitoring with Wearables Enhances Postoperative Patient Safety
- New Approach Enables Customized Muscle Tissue Without Biomaterial Scaffolds
- Robot-Assisted Brain Angiography Improves Procedural Outcomes
- Brain Mapping Technology Enhances Precision in Brain Tumor Resection
- Handheld Robotic System Expands Options for Total Knee Surgery
- VR Experience Reduces Patient Anxiety Before Kidney Stone Procedure
- Injectable Mini Livers Offer Hope for Patients Awaiting Transplant
- Pulsed Field Ablation Technology Cleared in Europe for Persistent AFib
- AI-Powered Imaging Brings Real-Time Margin Clarity to Breast Cancer Surgery
- Minimally Invasive Device Safely Treats Challenging Brain Aneurysms
- Surgical Robot Makes Complex Liver Tumor Surgery Safer and Less Invasive
- Neurostimulation Implant Reduces Seizure Burden in Drug-Resistant Epilepsy
- Minimally Invasive Procedure Effectively Treats Small Kidney Cancers
Channels
Artificial Intelligence
view channelAI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
Accurately identifying long-term cardiovascular disease risk in asymptomatic adults remains challenging for clinicians. Missed or underestimated risk delays preventive therapy and increases the chance... Read more
Machine Learning Approach Enhances Liver Cancer Risk Stratification
Hepatocellular carcinoma, the most common form of primary liver cancer, is often detected late despite targeted surveillance programs. Current screening guidelines emphasize patients with known cirrhosis,... Read moreCritical Care
view channel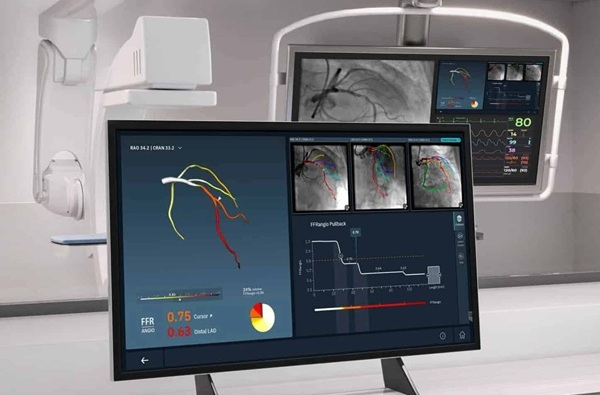
Angiography-Based FFR Approach Matches Gold Standard Results Without Wires
Accurately determining whether a coronary stenosis limits blood flow is essential to guide percutaneous coronary intervention, yet wire-based physiologic testing remains underused due to added procedural... Read more
Eye Imaging AI Identifies Elevated Cardiovascular Risk
Many adults at risk for atherosclerotic cardiovascular disease are not identified until they undergo formal primary care assessment. Delayed risk recognition can postpone initiation of statins and lifestyle... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel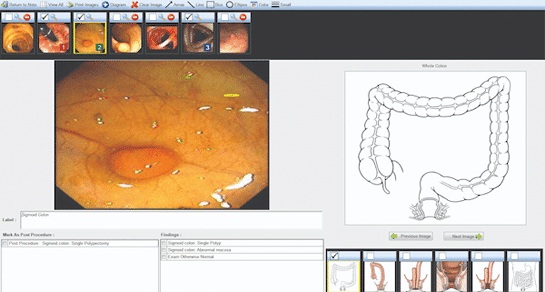
Voice-Driven AI System Enables Structured GI Procedure Documentation
Documentation during gastrointestinal (GI) procedures often competes with real-time clinical decision-making and imposes a significant cognitive burden on physicians. Manual data entry and post-procedure... Read more
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read moreBusiness
view channel