FDA Offers Vote of Confidence for AI in Healthcare
|
By HospiMedica International staff writers Posted on 17 May 2018 |
FDA Commissioner Scott Gottlieb, MD, believes that artificial intelligence holds enormous promise for the future of medicine, and is actively developing a new regulatory framework to promote innovation and support the use of AI-based technologies in healthcare.
Speaking at the AcademyHealth (Washington DC, USA) annual Health Datapalooza, held during April 2018 in Washington (DC, USA), Commissioner Gottlieb said the FDA is fully expecting to see an ever-increasing number of AI-powered healthcare tools submitted for approval in the years to come, from imaging devices to technology derived from other industries such as finance that are already widely using AI platforms for fraud detection. The FDA, in turn, is developing improved strategies for regulation and launching new projects at the FDA's internal data science incubator.
The Information Exchange and Data Transformation (INFORMED) incubator will explore opportunities for machine learning and AI to improve existing clinical practices, such as open-access platforms and shared, continually reconciled database exchange technologies such as blockchain, which can enable the widespread and secure exchange of health data. Among the ongoing projects is collaboration with Project Data Sphere, a nonprofit open-access cancer data repository, aimed at developing algorithms for classification of tumor dynamics using medical imaging data.
“We’re implementing a new approach to the review of artificial intelligence. FDA's regulatory approach will focus on the ways in which real-world data flows. This includes structured and unstructured data from pathology slides, electronic medical records, wearable devices, and insurance claims data,” said Dr. Gottlieb. “We want to better understand, and unlock ways, that this data can be used to inform development and validation of AI devices.”
“FDA also plans to ensure its regulatory framework and software validation tools are sufficiently flexible to keep pace with the unique attributes of this rapidly advancing field. That said, the agency still needs to establish appropriate guardrails for patients,” concluded Dr. Gottlieb. “It’ll look at developing new clinical endpoints and signal detection methods for evaluation of the safety and effectiveness of therapies. These efforts also will help us develop new approaches for understanding variations in individual patient experience using diverse data sets from clinical trials, EHRs, and biometric monitoring devices.”
The FDA has also recently launched a fellowship program with Harvard University (Boston, MA, USA) on AI and machine learning, which is focused on designing, developing, and implementing algorithms for regulatory science applications. One such example is innovative clinical decision support software that uses AI algorithms to help alert neurovascular specialists of brain deterioration.
Related Links:
AcademyHealth
Speaking at the AcademyHealth (Washington DC, USA) annual Health Datapalooza, held during April 2018 in Washington (DC, USA), Commissioner Gottlieb said the FDA is fully expecting to see an ever-increasing number of AI-powered healthcare tools submitted for approval in the years to come, from imaging devices to technology derived from other industries such as finance that are already widely using AI platforms for fraud detection. The FDA, in turn, is developing improved strategies for regulation and launching new projects at the FDA's internal data science incubator.
The Information Exchange and Data Transformation (INFORMED) incubator will explore opportunities for machine learning and AI to improve existing clinical practices, such as open-access platforms and shared, continually reconciled database exchange technologies such as blockchain, which can enable the widespread and secure exchange of health data. Among the ongoing projects is collaboration with Project Data Sphere, a nonprofit open-access cancer data repository, aimed at developing algorithms for classification of tumor dynamics using medical imaging data.
“We’re implementing a new approach to the review of artificial intelligence. FDA's regulatory approach will focus on the ways in which real-world data flows. This includes structured and unstructured data from pathology slides, electronic medical records, wearable devices, and insurance claims data,” said Dr. Gottlieb. “We want to better understand, and unlock ways, that this data can be used to inform development and validation of AI devices.”
“FDA also plans to ensure its regulatory framework and software validation tools are sufficiently flexible to keep pace with the unique attributes of this rapidly advancing field. That said, the agency still needs to establish appropriate guardrails for patients,” concluded Dr. Gottlieb. “It’ll look at developing new clinical endpoints and signal detection methods for evaluation of the safety and effectiveness of therapies. These efforts also will help us develop new approaches for understanding variations in individual patient experience using diverse data sets from clinical trials, EHRs, and biometric monitoring devices.”
The FDA has also recently launched a fellowship program with Harvard University (Boston, MA, USA) on AI and machine learning, which is focused on designing, developing, and implementing algorithms for regulatory science applications. One such example is innovative clinical decision support software that uses AI algorithms to help alert neurovascular specialists of brain deterioration.
Related Links:
AcademyHealth
Latest AI News
- Algorithm Identifies Cardiac Arrest Hotspots to Guide AED Placement
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data
- AI Tool Maps Early Risk Patterns in Bloodstream Infections
- AI Model Identifies Rare Endocrine Disorder from Hand Images
- AI Tool Promises to Reduce Length of Hospital Stays and Free Up Beds
Channels
Critical Care
view channel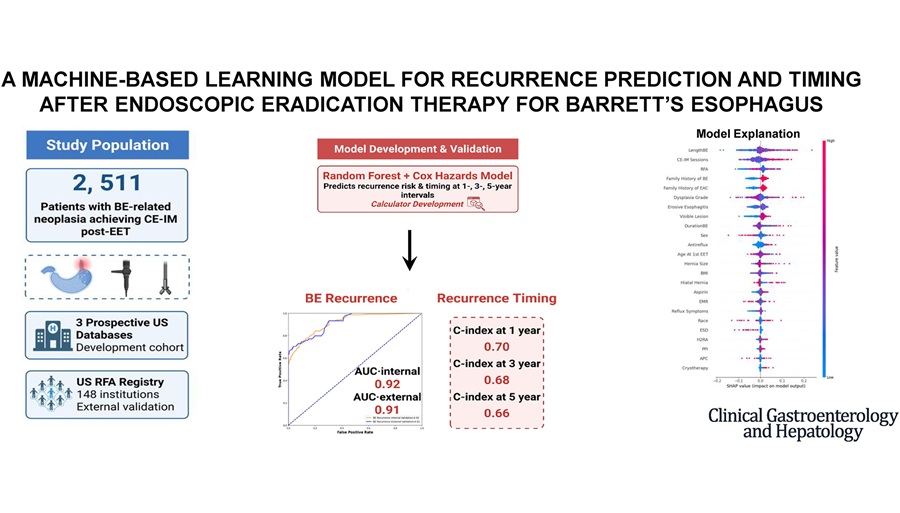
AI Tool Predicts Post-Therapy Barrett’s Esophagus Recurrence
Barrett’s esophagus (BE) is the only known precursor to esophageal adenocarcinoma, an aggressive cancer with high mortality. After endoscopic eradication therapy, disease can recur, making long-term surveillance... Read more
New Technology Turns Earbuds into Sensors for Cardiac Function Tracking
Continuous assessment of cardiac mechanics outside the clinic remains difficult because existing tools often require chest-mounted sensors and brief, supervised recordings. Missed early changes in valve... Read moreSurgical Techniques
view channelEndovascular Stent Graft Enables Minimally Invasive Aortic Arch Repair
Open repair of aortic arch disease is complex and carries substantial risk for patients with significant comorbidities. Many individuals are not candidates for surgery, and endovascular options for this... Read more
Dynamic Pressure Overlay Reduces Perioperative Pressure Injuries
Perioperative pressure injuries are a persistent risk during long procedures, particularly when repositioning is limited. Time in the operating room beyond two hours is cited as an intrinsic risk factor,... Read more
Pulsed Field Ablation System Streamlines Atrial Fibrillation Procedures
Atrial fibrillation is the most common cardiac arrhythmia, affecting more than 50 million people worldwide and straining capacity for rhythm-control procedures. Electrophysiology teams continue to seek... Read more
Single-Use System Enables Minimally Invasive Decompression for Lumbar Spinal Stenosis
Lumbar spinal stenosis is frequently driven by hypertrophic bone that narrows the canal and produces pain. Conventional decompression often relies on larger incisions and bulky retractors, adding time,... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
Cardiogenic shock remains a difficult, time-sensitive emergency, with delayed identification driving poor outcomes and persistently high mortality. Many cases go undocumented even at advanced stages, hindering... Read more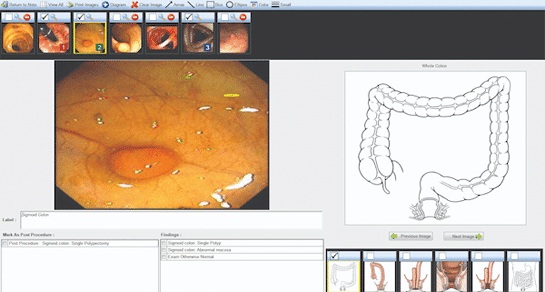
Voice-Driven AI System Enables Structured GI Procedure Documentation
Documentation during gastrointestinal (GI) procedures often competes with real-time clinical decision-making and imposes a significant cognitive burden on physicians. Manual data entry and post-procedure... Read more
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read moreBusiness
view channel
External Liver Assist System Receives FDA RMAT Designation
Acute liver failure can develop over days to weeks and is often fatal when transplantation is not possible or unavailable. Although some patients recover spontaneously, many deteriorate rapidly amid a... Read more