Wearable AI Device Monitors Key Biometrics of Volunteers in World's First Phase 3 Trials Of Inactivated COVID-19 Vaccine
|
By HospiMedica International staff writers Posted on 19 Aug 2020 |

Illustration
A fitness tracker has become the first wearable device to be used in the world's first Phase 3 trials of an inactivated COVID-19 vaccine and will be used to measure key biometrics of the volunteers.
This is the first trial of its kind to feature wearable technology and will include thousands of volunteers who are participating in the trials in Abu Dhabi, UAE. The inactivated COVID-19 vaccine has been developed by the Wuhan Institute of Biological Products under the China National Biotec Group (CNBG) affiliated to the China National Pharmaceutical Group Co., Ltd. (Sinopharm Beijing, China). The trials are as a result of a cooperation partnership between G42 Healthcare (Abu Dhabi, UAE) and Sinopharm CNBG, the world’s sixth largest vaccine manufacturer. The Phase 3 trials are open to individual volunteers aged between 18 and 60 living in the UAE and Bahrain and will last for six to 12 months, with the volunteers required to be available for follow ups during this time.
G42 Healthcare has now joined hands with wearable technology firm WHOOP (Boston, MA, USA) to launch the Whoop4Humanity initiative to ensure that volunteers who now join the clinical trials, including those coming in for the second dose, can further enhance their participation in the trails by using the new WHOOP Strap 3.0 wearable device which will enable them to check their daily heart rate, respiratory rate, heart rate variability, sleep performance, and more.
The WHOOP Strap 3.0 wearable device can be a powerful tool to boost the comprehensive volunteer healthcare management program by helping thousands of vaccine volunteers to self-monitor their well-being while measuring their fitness, sleep, and recovery levels during the trials. It provides valuable insights and actionable feedback around specific markers, thus encouraging better decision-making for volunteers' health and contributing to the trials' overall success.
“As a digital business, this new partnership is a logical extension of the highly-advanced AI and super computer solutions we are already using in the trials process. New volunteers will be invited to engage with the technology developed by WHOOP to enhance their individual volunteer experience and contribute to the advanced research involved in these sophisticated trials,” said Ashish Koshy, CEO of G42 Healthcare. “We believe that giving volunteers an opportunity to use the WHOOP Strap 3.0 wearable device will reinforce their confidence in the trial by giving them the ability to check on several daily health data points. This will complement the world-class clinical expertise that the healthcare professionals in the UAE are providing to the volunteers and ensure their safety and well-being throughout the trials process.”
“WHOOP is proud to partner with G42 Healthcare and participate in their innovative Phase III COVID-19 vaccine trial,” said Will Ahmed, WHOOP Founder & CEO. “The continuous biometric monitoring offered by WHOOP will help volunteers evaluate any changes to their health and help researchers vastly increase their understanding of the vaccine's impact. This partnership represents the future of healthcare: using continuous monitoring and artificial intelligence to better understand the effect of drugs, vaccines, and other treatments.”
Related Links:
WHOOP
G42 Healthcare
China National Pharmaceutical Group Co., Ltd.
This is the first trial of its kind to feature wearable technology and will include thousands of volunteers who are participating in the trials in Abu Dhabi, UAE. The inactivated COVID-19 vaccine has been developed by the Wuhan Institute of Biological Products under the China National Biotec Group (CNBG) affiliated to the China National Pharmaceutical Group Co., Ltd. (Sinopharm Beijing, China). The trials are as a result of a cooperation partnership between G42 Healthcare (Abu Dhabi, UAE) and Sinopharm CNBG, the world’s sixth largest vaccine manufacturer. The Phase 3 trials are open to individual volunteers aged between 18 and 60 living in the UAE and Bahrain and will last for six to 12 months, with the volunteers required to be available for follow ups during this time.
G42 Healthcare has now joined hands with wearable technology firm WHOOP (Boston, MA, USA) to launch the Whoop4Humanity initiative to ensure that volunteers who now join the clinical trials, including those coming in for the second dose, can further enhance their participation in the trails by using the new WHOOP Strap 3.0 wearable device which will enable them to check their daily heart rate, respiratory rate, heart rate variability, sleep performance, and more.
The WHOOP Strap 3.0 wearable device can be a powerful tool to boost the comprehensive volunteer healthcare management program by helping thousands of vaccine volunteers to self-monitor their well-being while measuring their fitness, sleep, and recovery levels during the trials. It provides valuable insights and actionable feedback around specific markers, thus encouraging better decision-making for volunteers' health and contributing to the trials' overall success.
“As a digital business, this new partnership is a logical extension of the highly-advanced AI and super computer solutions we are already using in the trials process. New volunteers will be invited to engage with the technology developed by WHOOP to enhance their individual volunteer experience and contribute to the advanced research involved in these sophisticated trials,” said Ashish Koshy, CEO of G42 Healthcare. “We believe that giving volunteers an opportunity to use the WHOOP Strap 3.0 wearable device will reinforce their confidence in the trial by giving them the ability to check on several daily health data points. This will complement the world-class clinical expertise that the healthcare professionals in the UAE are providing to the volunteers and ensure their safety and well-being throughout the trials process.”
“WHOOP is proud to partner with G42 Healthcare and participate in their innovative Phase III COVID-19 vaccine trial,” said Will Ahmed, WHOOP Founder & CEO. “The continuous biometric monitoring offered by WHOOP will help volunteers evaluate any changes to their health and help researchers vastly increase their understanding of the vaccine's impact. This partnership represents the future of healthcare: using continuous monitoring and artificial intelligence to better understand the effect of drugs, vaccines, and other treatments.”
Related Links:
WHOOP
G42 Healthcare
China National Pharmaceutical Group Co., Ltd.
Latest AI News
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data
- AI Tool Maps Early Risk Patterns in Bloodstream Infections
- AI Model Identifies Rare Endocrine Disorder from Hand Images
- AI Tool Promises to Reduce Length of Hospital Stays and Free Up Beds
- Machine Learning Model Cuts Canceled Liver Transplants By 60%
Channels
Artificial Intelligence
view channel
Machine Learning Approach Enhances Liver Cancer Risk Stratification
Hepatocellular carcinoma, the most common form of primary liver cancer, is often detected late despite targeted surveillance programs. Current screening guidelines emphasize patients with known cirrhosis,... Read more
New AI Approach Monitors Brain Health Using Passive Wearable Data
Brain health spans cognitive and emotional functions and can fluctuate even in adults without diagnosed disease. Detecting early changes remains difficult in routine care and burdens specialty services... Read moreCritical Care
view channel
Automated IV Labeling Solution Improves Infusion Safety and Efficiency
Medication administration in high-acuity settings is often complicated by multiple concurrent infusions, making accurate line identification essential. In a 10-hospital intensive care unit study, 60% of... Read more
First-Of-Its-Kind AI Tool Detects Pulmonary Hypertension from Standard ECGs
Pulmonary hypertension is a progressive, life‑threatening disease that is frequently missed early because symptoms such as dyspnea are nonspecific and diagnostic delays can exceed two years.... Read moreSurgical Techniques
view channel
Continuous Monitoring with Wearables Enhances Postoperative Patient Safety
Postoperative hypoxemia on general surgical wards is common and often missed by intermittent vital sign checks. Undetected low oxygen levels can delay recovery and raise the risk of complications that... Read more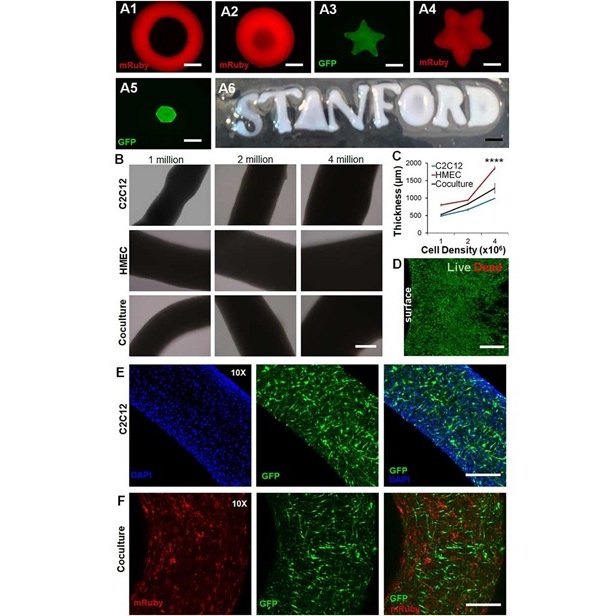
New Approach Enables Customized Muscle Tissue Without Biomaterial Scaffolds
Volumetric muscle loss is a traumatic loss of skeletal muscle that often leads to permanent functional impairment and limited reconstructive options. Current experimental strategies struggle to deliver... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read moreBusiness
view channel