Monitoring Technology Keeps COVID-19 Vaccines Safe
|
By HospiMedica International staff writers Posted on 01 Dec 2020 |

Image: The OneVue Sense monitors vaccine storage temperatures (Photo courtesy of Primex)
An automated environmental monitoring platform can help administrators prepare for the pending global rollout of COVID-19 vaccines.
The Primex (Lake Geneva, WI, USA) OneVue Sense sensor incorporates a cryogenic probe that can monitor storage units for temperatures as low as minus 200 degrees Celsius. Once paired with the OneVue Monitor automated monitoring software solution, the integrated system provides real-time alerts if vaccine storage temperatures fluctuate outside of the specified temperature range, helping maintain vaccine safety and effectiveness. The system also archives all recorded temperatures to ensure compliance.
In addition to environmental temperature monitoring, the OneVue monitoring software also provides indoor air quality data, differential pressure reading, and contact closure monitoring solutions, which can be used to monitor hospital isolation rooms and to ensure that freezer storage doors remain closed. The cloud-based software powering the OneVue system is fully customizable, enabling users to schedule reports, alerts, and messaging to suit specific needs, and to synchronize many different types of devices across a facility.
“For nearly 50 years, healthcare providers have trusted Primex to deliver proven and reliable healthcare solutions. We are proud to be part of this monumental public health effort to ensure safe, reliable, and effective COVID-19 vaccines reach the public,” said Paul Shekoski, CEO of Primex. “Any breach in temperature control can degrade a vaccine, making it lose its full potency. With the Primex OneVue platform, customers have peace of mind knowing that the vaccine will always remain at the proper temperature.”
The current leading COVID-19 vaccine candidates are expected to require extremely cold storage temperatures, with some, such as the Pfizer (New York, NY, USA) candidate, requiring storage and triage at temperatures as low as minus 70 degrees Celsius.
Related Links:
Primex
Pfizer
The Primex (Lake Geneva, WI, USA) OneVue Sense sensor incorporates a cryogenic probe that can monitor storage units for temperatures as low as minus 200 degrees Celsius. Once paired with the OneVue Monitor automated monitoring software solution, the integrated system provides real-time alerts if vaccine storage temperatures fluctuate outside of the specified temperature range, helping maintain vaccine safety and effectiveness. The system also archives all recorded temperatures to ensure compliance.
In addition to environmental temperature monitoring, the OneVue monitoring software also provides indoor air quality data, differential pressure reading, and contact closure monitoring solutions, which can be used to monitor hospital isolation rooms and to ensure that freezer storage doors remain closed. The cloud-based software powering the OneVue system is fully customizable, enabling users to schedule reports, alerts, and messaging to suit specific needs, and to synchronize many different types of devices across a facility.
“For nearly 50 years, healthcare providers have trusted Primex to deliver proven and reliable healthcare solutions. We are proud to be part of this monumental public health effort to ensure safe, reliable, and effective COVID-19 vaccines reach the public,” said Paul Shekoski, CEO of Primex. “Any breach in temperature control can degrade a vaccine, making it lose its full potency. With the Primex OneVue platform, customers have peace of mind knowing that the vaccine will always remain at the proper temperature.”
The current leading COVID-19 vaccine candidates are expected to require extremely cold storage temperatures, with some, such as the Pfizer (New York, NY, USA) candidate, requiring storage and triage at temperatures as low as minus 70 degrees Celsius.
Related Links:
Primex
Pfizer
Latest Critical Care News
- Automated IV Labeling Solution Improves Infusion Safety and Efficiency
- First-Of-Its-Kind AI Tool Detects Pulmonary Hypertension from Standard ECGs
- 4D Digital Twin Heart Model Improves CRT Outcomes
- AI Turns Glucose Data Into Actionable Insights for Diabetes Care
- Microscale Wireless Implant Tracks Brain Activity Over Time
- Smart Mask Delivers Continuous, Battery-Free Breath Monitoring
- Routine Blood Pressure Readings May Identify Risk of Future Cognitive Decline
- CGM-Based Algorithm Enhances Insulin Dose Adjustment in Type 2 Diabetes
- Fish Scale–Based Implants Offer New Approach to Corneal Repair
- Dual-Function Wound Patch Combines Infection Sensing and Treatment
- Smartwatch Signals and Blood Tests Team Up for Early Warning on Insulin Resistance
- Smart Fabric Technology Aims to Prevent Pressure Injuries in Hospital Care
- Standardized Treatment Algorithm Improves Blood Pressure Control
- Combined Infection Control Strategy Limits Drug-Resistant Outbreak in NICU
- AI Helps Predict Which Heart-Failure Patients Will Worsen Within a Year
- Algorithm Allows Paramedics to Predict Brain Damage Risk After Cardiac Arrest
Channels
Artificial Intelligence
view channel
Machine Learning Approach Enhances Liver Cancer Risk Stratification
Hepatocellular carcinoma, the most common form of primary liver cancer, is often detected late despite targeted surveillance programs. Current screening guidelines emphasize patients with known cirrhosis,... Read more
New AI Approach Monitors Brain Health Using Passive Wearable Data
Brain health spans cognitive and emotional functions and can fluctuate even in adults without diagnosed disease. Detecting early changes remains difficult in routine care and burdens specialty services... Read moreSurgical Techniques
view channel
Continuous Monitoring with Wearables Enhances Postoperative Patient Safety
Postoperative hypoxemia on general surgical wards is common and often missed by intermittent vital sign checks. Undetected low oxygen levels can delay recovery and raise the risk of complications that... Read more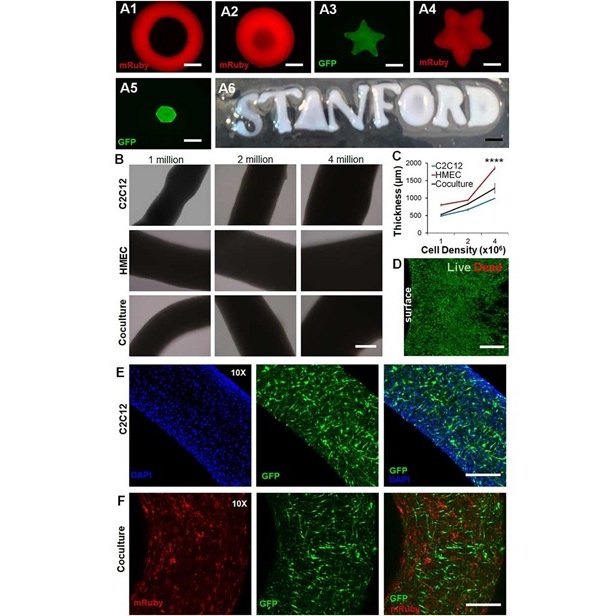
New Approach Enables Customized Muscle Tissue Without Biomaterial Scaffolds
Volumetric muscle loss is a traumatic loss of skeletal muscle that often leads to permanent functional impairment and limited reconstructive options. Current experimental strategies struggle to deliver... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read moreBusiness
view channel