Zoll Medical Acquires CSA Systems Manufacturer Respicardia
|
By HospiMedica International staff writers Posted on 14 Apr 2021 |
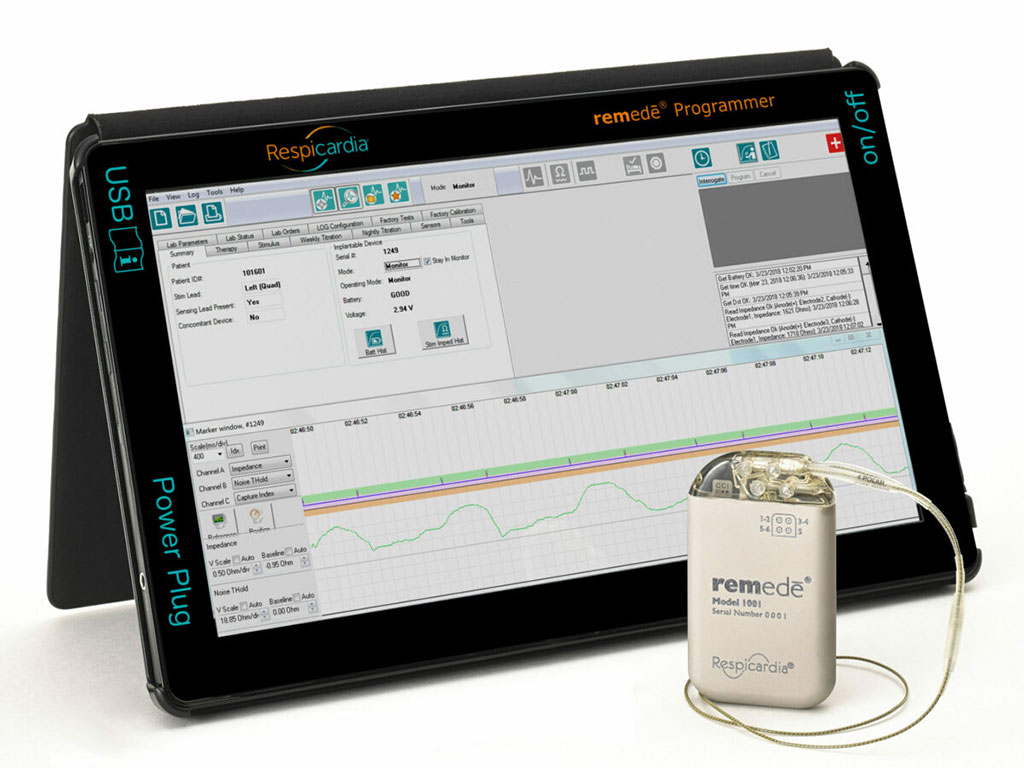
Image: Respicardia’s remedē System (Photo courtesy of Respicardia, Inc.)
ZOLL Medical Corporation (Chelmsford, MA, USA), an Asahi Kasei company, has acquired Respicardia, Inc. (Minnetonka, MN, USA), a provider of novel implantable neurostimulators for the treatment of moderate to severe Central Sleep Apnea (CSA).
CSA occurs when the brain fails to send appropriate signals to the breathing muscles that stimulate a regular breathing pattern. Respicardia’s remedē System is a revolutionary treatment for CSA, and is the only implantable device approved by the US Food and Drug Administration (FDA) to treat moderate to severe CSA in adult patients with reduced cardiac function. Respicardia’s remedē System is implanted by a cardiac electrophysiologist during a minimally invasive outpatient procedure. The remedē System delivers electrical pulses to one of the phrenic nerves, which send signals to the diaphragm, restoring a more normal breathing pattern during sleep.
ZOLL, which develops and markets medical devices and software solutions that help advance emergency care and save lives, has been a significant investor in Respicardia for more than three years. With products for defibrillation and cardiac monitoring, circulation enhancement and CPR feedback, supersaturated oxygen therapy, data management, ventilation, and therapeutic temperature management, ZOLL provides a comprehensive set of technologies that help clinicians, EMS and fire professionals, as well as lay rescuers, improve patient outcomes in critical cardiopulmonary conditions.
“ZOLL and Respicardia both develop innovative therapies for conditions with large unmet clinical needs, and we share a deep commitment to improving patient outcomes,” said Jon Rennert, CEO of ZOLL Medical. “With this acquisition, ZOLL will combine its expertise in cardiac and respiratory care with Respicardia’s novel remedē System to make a meaningful difference in the health and quality of life for many patients.”
“Respicardia is excited to be joining the ZOLL family,” said Peter Sommerness, CEO of Respicardia. “As an innovator of safe and effective therapies that address the unmet needs of respiratory and cardiovascular patients, we see tremendous potential ahead with the support of ZOLL’s strong brand and global reach.”
Related Links:
ZOLL Medical Corporation
Respicardia, Inc.
CSA occurs when the brain fails to send appropriate signals to the breathing muscles that stimulate a regular breathing pattern. Respicardia’s remedē System is a revolutionary treatment for CSA, and is the only implantable device approved by the US Food and Drug Administration (FDA) to treat moderate to severe CSA in adult patients with reduced cardiac function. Respicardia’s remedē System is implanted by a cardiac electrophysiologist during a minimally invasive outpatient procedure. The remedē System delivers electrical pulses to one of the phrenic nerves, which send signals to the diaphragm, restoring a more normal breathing pattern during sleep.
ZOLL, which develops and markets medical devices and software solutions that help advance emergency care and save lives, has been a significant investor in Respicardia for more than three years. With products for defibrillation and cardiac monitoring, circulation enhancement and CPR feedback, supersaturated oxygen therapy, data management, ventilation, and therapeutic temperature management, ZOLL provides a comprehensive set of technologies that help clinicians, EMS and fire professionals, as well as lay rescuers, improve patient outcomes in critical cardiopulmonary conditions.
“ZOLL and Respicardia both develop innovative therapies for conditions with large unmet clinical needs, and we share a deep commitment to improving patient outcomes,” said Jon Rennert, CEO of ZOLL Medical. “With this acquisition, ZOLL will combine its expertise in cardiac and respiratory care with Respicardia’s novel remedē System to make a meaningful difference in the health and quality of life for many patients.”
“Respicardia is excited to be joining the ZOLL family,” said Peter Sommerness, CEO of Respicardia. “As an innovator of safe and effective therapies that address the unmet needs of respiratory and cardiovascular patients, we see tremendous potential ahead with the support of ZOLL’s strong brand and global reach.”
Related Links:
ZOLL Medical Corporation
Respicardia, Inc.
Latest Business News
- GE HealthCare Leads Major European Initiative to Advance Cardio-Oncology Care
- Medtronic and GE HealthCare Broaden Alliance Across Monitoring and Care Solutions
- Quantum Surgical Acquires NeuWave from Johnson & Johnson
- Medtronic to Acquire Coronary Artery Medtech Company CathWorks
- Medtronic and Mindray Expand Strategic Partnership to Ambulatory Surgery Centers in the U.S.
- FDA Clearance Expands Robotic Options for Minimally Invasive Heart Surgery
- WHX in Dubai (formerly Arab Health) to debut specialised Biotech & Life Sciences Zone as sector growth accelerates globally
- WHX in Dubai (formerly Arab Health) to bring together key UAE government entities during the groundbreaking 2026 edition
- Interoperability Push Fuels Surge in Healthcare IT Market
- Philips and Masimo Partner to Advance Patient Monitoring Measurement Technologies
- B. Braun Acquires Digital Microsurgery Company True Digital Surgery
- CMEF 2025 to Promote Holistic and High-Quality Development of Medical and Health Industry
- Bayer and Broad Institute Extend Research Collaboration to Develop New Cardiovascular Therapies
- Medtronic Partners with Corsano to Expand Acute Care & Monitoring Portfolio in Europe
- Expanded Collaboration to Transform OR Technology Through AI and Automation
- Becton Dickinson to Spin Out Biosciences and Diagnostic Solutions Business
Channels
Artificial Intelligence
view channel
Machine Learning Approach Enhances Liver Cancer Risk Stratification
Hepatocellular carcinoma, the most common form of primary liver cancer, is often detected late despite targeted surveillance programs. Current screening guidelines emphasize patients with known cirrhosis,... Read more
New AI Approach Monitors Brain Health Using Passive Wearable Data
Brain health spans cognitive and emotional functions and can fluctuate even in adults without diagnosed disease. Detecting early changes remains difficult in routine care and burdens specialty services... Read moreCritical Care
view channel
Automated IV Labeling Solution Improves Infusion Safety and Efficiency
Medication administration in high-acuity settings is often complicated by multiple concurrent infusions, making accurate line identification essential. In a 10-hospital intensive care unit study, 60% of... Read more
First-Of-Its-Kind AI Tool Detects Pulmonary Hypertension from Standard ECGs
Pulmonary hypertension is a progressive, life‑threatening disease that is frequently missed early because symptoms such as dyspnea are nonspecific and diagnostic delays can exceed two years.... Read moreSurgical Techniques
view channel
Continuous Monitoring with Wearables Enhances Postoperative Patient Safety
Postoperative hypoxemia on general surgical wards is common and often missed by intermittent vital sign checks. Undetected low oxygen levels can delay recovery and raise the risk of complications that... Read more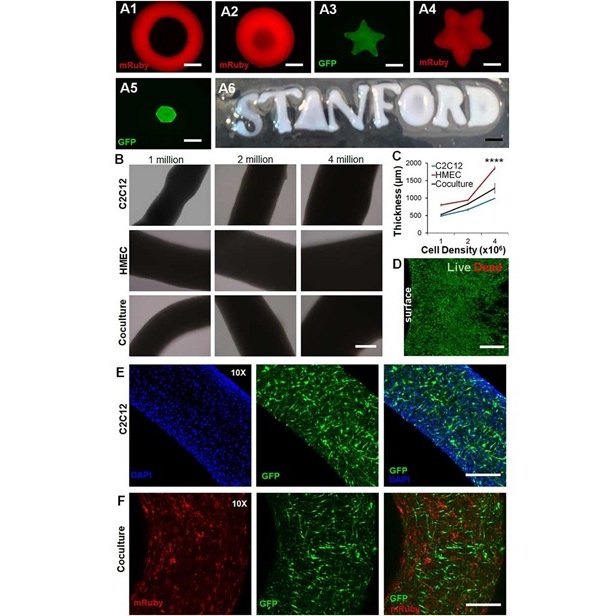
New Approach Enables Customized Muscle Tissue Without Biomaterial Scaffolds
Volumetric muscle loss is a traumatic loss of skeletal muscle that often leads to permanent functional impairment and limited reconstructive options. Current experimental strategies struggle to deliver... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more