Cryotherapy System Reduces Chronic Rhinitis Symptoms 
|
By HospiMedica International staff writers Posted on 06 Sep 2021 |
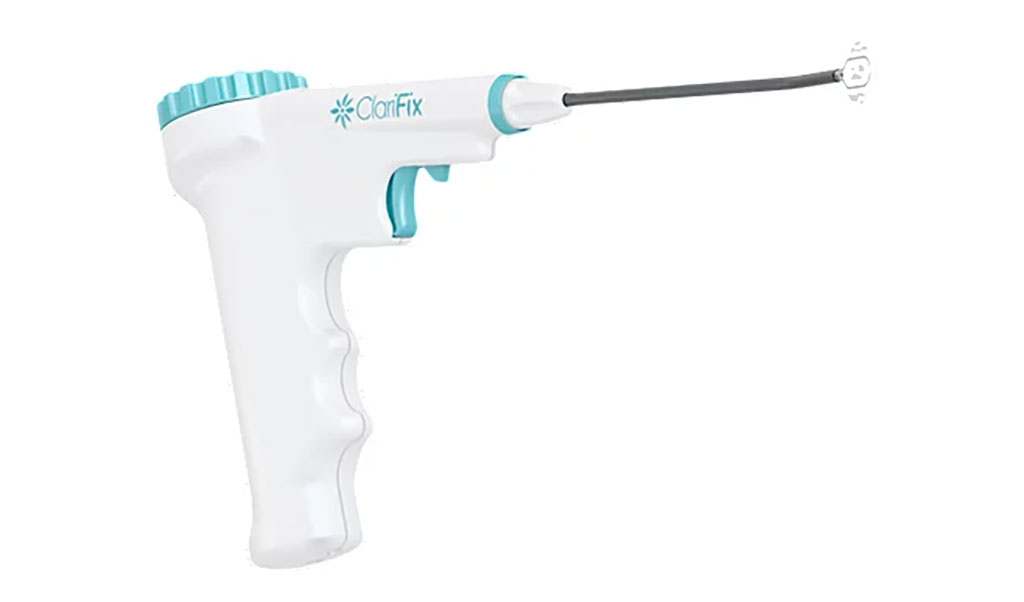
Image: The ClariFix cryotherapy device (Photo courtesy of Stryker)
A novel cryotherapy device freezes the posterior nasal nerve, thereby disrupting parasympathetic innervation of the nasal mucosa and reducing the symptoms of chronic rhinitis.
The Stryker Corporation (Kalamazoo, MI, USA) ClariFix cryotherapy device is a single patient-use, disposable cryosurgical device that consists of a handle attached to a cannula with a cryoprobe at the distal end. The handheld device is provided sterile, with two optional nitrous oxide (N2O) cryogenic canister sizes available. To perform cryosurgery, the desired cryogen canister is inserted into the ClariFix handle. A canister cap is then tightened onto the handle, piercing the canister. Once a mechanical valve is opened, cryogen flows down the cannula into the Cryoprobe.
The Cryoprobe is placed into contact with the targeted posterior nasal nerve via direct visualization, and can be rotated to ensure proper positioning. Once in the desired position, cryogen is released to flow into the Cryoprobe, partially evaporating to cool the Cryoprobe to -80°C. A freeze zone forms in the adjacent tissue, damaging the nerve. Once the area has been treated, cryosurgery is stopped by closing the valve. As soon as the Cryoprobe has thawed, it can be safely removed. Further cycles can be initiated per physician discretion.
Chronic rhinitis is a frequent inflammation of the nasal cavity with symptoms such as a runny nose, nasal congestion, and post-nasal drip. Symptoms of rhinitis include rhinorrhea (runny nose), nasal itching, nasal congestion, sneezing, and post-nasal drip, an accumulation of mucus in the back of the nose and throat leading to (or giving the sensation of) mucus dripping downward from the back of the nose.
Related Links:
Stryker Corporation
The Stryker Corporation (Kalamazoo, MI, USA) ClariFix cryotherapy device is a single patient-use, disposable cryosurgical device that consists of a handle attached to a cannula with a cryoprobe at the distal end. The handheld device is provided sterile, with two optional nitrous oxide (N2O) cryogenic canister sizes available. To perform cryosurgery, the desired cryogen canister is inserted into the ClariFix handle. A canister cap is then tightened onto the handle, piercing the canister. Once a mechanical valve is opened, cryogen flows down the cannula into the Cryoprobe.
The Cryoprobe is placed into contact with the targeted posterior nasal nerve via direct visualization, and can be rotated to ensure proper positioning. Once in the desired position, cryogen is released to flow into the Cryoprobe, partially evaporating to cool the Cryoprobe to -80°C. A freeze zone forms in the adjacent tissue, damaging the nerve. Once the area has been treated, cryosurgery is stopped by closing the valve. As soon as the Cryoprobe has thawed, it can be safely removed. Further cycles can be initiated per physician discretion.
Chronic rhinitis is a frequent inflammation of the nasal cavity with symptoms such as a runny nose, nasal congestion, and post-nasal drip. Symptoms of rhinitis include rhinorrhea (runny nose), nasal itching, nasal congestion, sneezing, and post-nasal drip, an accumulation of mucus in the back of the nose and throat leading to (or giving the sensation of) mucus dripping downward from the back of the nose.
Related Links:
Stryker Corporation
Latest Critical Care News
- New Nasal Spray Enables Prehospital Neuroprotection in Ischemic Stroke
- AI-Enhanced ECG Screens for Heart Failure Risk in Resource-Limited Settings
- Single-Lead AI ECG Tool Detects Moderate-to-Severe Hyperkalemia Outside Clinic
- Reduced-Intensity Transplant Regimen Expands Donor Access in Sickle Cell Disease
- Battery-Free ECG Patch Enables Continuous Arrhythmia Monitoring
- Spinal Cord Interface Restores Bladder Control in Preclinical Study
- Rapid Clotting Gel Improves Emergency Bleeding Control
- AI Tool Predicts In-Hospital Cardiac Arrest Minutes in Advance
- Magnetic Control System Enables Precise Navigation of Miniature Medical Devices
- Shelf-Stable Synthetic Platelets Could Enable Prehospital Hemorrhage Control
- Noninvasive AI Test Aims to Enable Earlier Pulmonary Hypertension Detection
- Blood-Filtering Device May Prolong Pregnancy in Severe Early Preeclampsia
- Noninvasive Skull Sensor Detects Early Deterioration in Acute Brain Injury
- Automated Robotic Imaging System Enhances Full-Body Skin Cancer Screening
- Bacteria-Resistant Urinary Catheter Coating Reduces Infections and Antibiotic Use
- New Brain-Computer Interface Targets Treatment-Resistant Depression
Channels
Artificial Intelligence
view channel
FDA-Cleared AI System Detects Sepsis Earlier and Reduces Mortality
Sepsis remains one of the deadliest complications for hospitalized patients, in part because its early signs overlap with other conditions. Each hour of delayed recognition measurably decreases survival,... Read moreFacial Image Analysis Tracks Biological Aging, Predicts Cancer Outcomes
Biological aging is the progressive loss of physiological function that may diverge from chronological age. In cancer care, clinicians need simple tools that reflect dynamic changes in patient resilience... Read moreSurgical Techniques
view channel
New Endoscopic Device Offers Minimally Invasive Approach to Obesity Management
Obesity is increasing worldwide, and many patients face a gap between lifestyle interventions, pharmacotherapy, and bariatric surgery. Endoscopic approaches are emerging as additional treatment options... Read more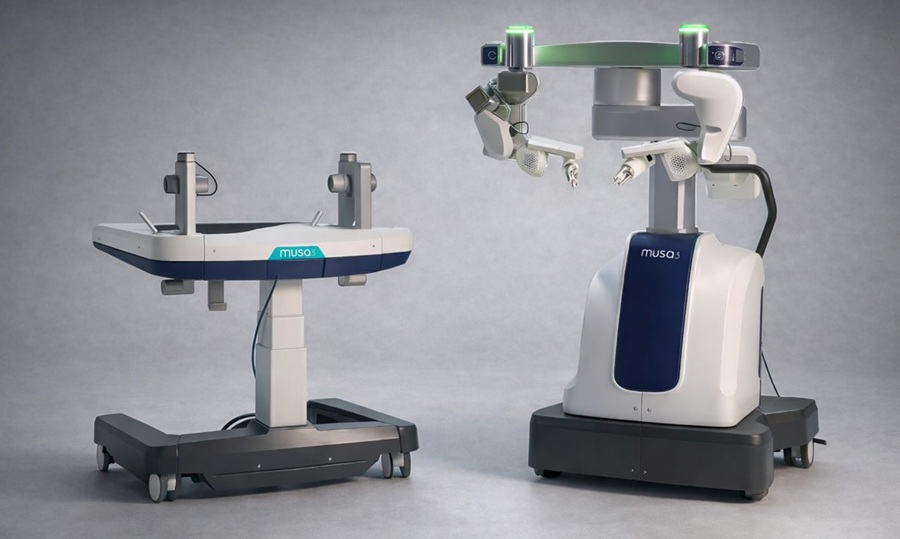
Robotic Microsurgery System Gains CE Mark for Clinical Use in Europe
Microsure (Eindhoven, The Netherlands) announced on May 7, 2026 that its MUSA-3 system received CE mark approval, enabling clinical use in Europe. The milestone marks the company’s transition from product... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read more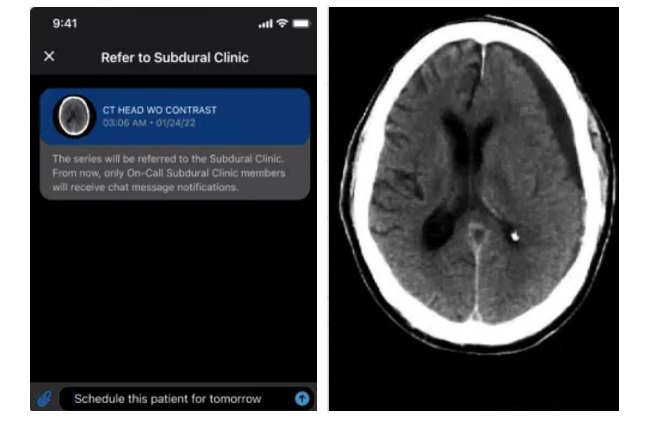
AI System Detects and Quantifies Chronic Subdural Hematoma
Viz.ai (San Francisco, CA, USA) announced a strategic commercialization collaboration with Johnson & Johnson (New Brunswick, NJ, USA) to expand access in the United States to the Viz Subdural solution... Read more
Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
Patients leaving skilled nursing facilities often lose continuous physiologic monitoring, increasing the risk of undetected infection and delayed intervention. Nursing home residents are seven times more... Read more
Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
Cardiogenic shock remains a difficult, time-sensitive emergency, with delayed identification driving poor outcomes and persistently high mortality. Many cases go undocumented even at advanced stages, hindering... Read morePoint of Care
view channel
Point-of-Care Viscoelastic Testing System Supports Obstetric Bleeding Management
HemoSonics (Durham, NC, USA) announced on May 5, 2026 that the company's Quantra Hemostasis System for Obstetric Procedures won Silver in the 2026 Edison Awards in the Women’s Health and Reproductive Innovations... Read moreBusiness
view channel
Olympus Partnership Aims to Expand Access to Robot-Assisted Endoscopic Therapy
Olympus has signed an exclusive global distribution agreement with EndoRobotics Co., Ltd., under which robot-assisted technologies developed by EndoRobotics will be distributed worldwide as part of the... Read more