Groundbreaking Technology Radically Improves Minimally Invasive Endoscopic Surgery
|
By HospiMedica International staff writers Posted on 11 Jan 2023 |
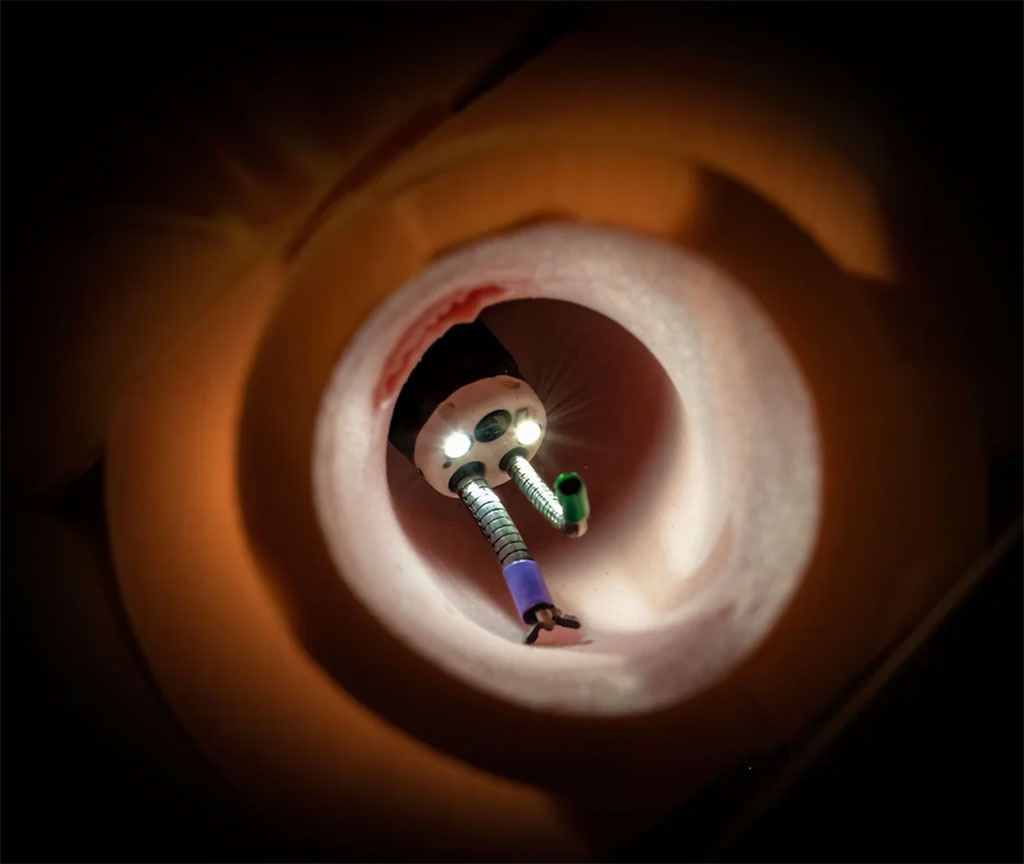
A next-generation endoscope technology aims to radically improve minimally invasive flexible endoscopic surgery by harnessing the power of intelligently micro-machined smart materials to create highly dexterous manipulators at millimeter and sub-millimeter scales, thereby enhancing therapeutic outcomes.
The disruptive platform technology from EndoTheia Inc. (Nashville, TN, USA), based on extremely thin-walled, laser-machined metallic tubes, enables the creation of highly flexible, steerable devices that can pass through standard endoscopes, while also carrying within themselves interventional tools. The steerability that these devices provide to endoscope-delivered tools opens the door for new diagnostic and therapeutic applications of flexible endoscopy. The technology can be used across a vast array of clinical specialties, including urology, gastroenterology, neurology, and otology.
EndoTheia’s proprietary technology exploits pre-programmed mechanical properties in concentric super-elastic tubes to enable tool steerability at the tips of flexible endoscopes without any modification to the endoscope itself. This eliminates parasitic bending along the instrument length like with tendon-actuated systems. By doing away with pullwires and discrete linkages, EndoTheia can create steerable tools with thinner walls and smaller diameters than ever before. It is this capability that allows the company to create dexterous instruments all the way down to 2F in diameter, with an open lumen allowing for the passage of adjunct tools, fluid flow, or suction/irrigation, adding value to the most demanding endoscopic applications. Coupling EndoTheia’s steerable tips with proprietary insertion shaft technology enables distal dexterity at the most demanding of curvatures and deflection angles, with high torsional trackability, axial stiffness, and bidirectional steerability.
EndoTheia has gained priority status from the FDA by receiving a Breakthrough Device designation from the agency for its next-generation endoscope technology. EndoTheia has demonstrated feasibility in animal, cadaver, and tissue model studies in surgeries for biliary disease, ureteroscopy kidney stone removal, endoscopic submucosal dissection (ESD), endoscopic neurosurgery, sinus surgery, and middle ear surgery. EndoTheia’s technology can provide dexterity in any procedures where tools are delivered through flexible endoscopes, including urology, neurology, interventional pulmonology, gastroenterology, colorectal, orthopedics, otolaryngology (ENT), and other sub-specialties.
“EndoTheia’s technology is currently the only viable option to add dexterity to flexible endoscopy, without re-engineering the endoscope itself,” said Robert J. Webster, III, PhD, Co-Founder and President of EndoTheia. “This empowers surgeons to provide much more accurate and precise therapeutic interventions in a wide range of clinical specialties.”
“We are excited that the FDA has recognized the huge potential of EndoTheia’s technology through Breakthrough Device designation,” added S. Duke Herrell, III, MD, FACS, Co-Founder and Chief Medical Officer of EndoTheia. “The FDA has affirmed our view that there is nothing else out there capable of doing what we do for flexible endoscopy, and that our technology represents a breakthrough treatment option for patients who currently face irreversibly debilitating disease.”
Related Links:
EndoTheia Inc.
Latest Surgical Techniques News
- Robot-Assisted Brain Angiography Improves Procedural Outcomes
- Brain Mapping Technology Enhances Precision in Brain Tumor Resection
- VR Experience Reduces Patient Anxiety Before Kidney Stone Procedure
- Injectable Mini Livers Offer Hope for Patients Awaiting Transplant
- Pulsed Field Ablation Technology Cleared in Europe for Persistent AFib
- AI-Powered Imaging Brings Real-Time Margin Clarity to Breast Cancer Surgery
- Minimally Invasive Device Safely Treats Challenging Brain Aneurysms
- Surgical Robot Makes Complex Liver Tumor Surgery Safer and Less Invasive
- Neurostimulation Implant Reduces Seizure Burden in Drug-Resistant Epilepsy
- Minimally Invasive Procedure Effectively Treats Small Kidney Cancers
- Fluorescence Probe Paired with Engineered Enzymes Lights Up Tumors for Easier Surgical Removal
- Novel Hydrogel Could Become Bone Implant of the Future
- Skull Implant Design Could Shape Surgical Outcomes
- Redesigned Surgical Laser Cuts Bone Deeper and Faster Than Before
- New Method Offers Less Invasive Detection of Susceptibility to Rare Anesthesia Reaction
- Laser Ablation Plus Immunotherapy Improves Survival in Recurrent Glioblastoma
Channels
Artificial Intelligence
view channel
AI Model Identifies Rare Endocrine Disorder from Hand Images
Acromegaly is a rare, intractable disease that typically begins in middle age and causes enlargement of the hands and feet, changes in facial appearance, and abnormal bone and organ growth.... Read moreCritical Care
view channel
Algorithm Allows Paramedics to Predict Brain Damage Risk After Cardiac Arrest
Out-of-hospital cardiac arrest carries a high risk of death, with survival rates below 10%. Even when the heart is restarted, clinicians often face uncertainty about the extent of brain injury.... Read more
Combined Infection Control Strategy Limits Drug-Resistant Outbreak in NICU
Neonatal sepsis remains a leading cause of death in low- and middle-income countries, where overcrowded facilities and limited resources heighten transmission risks from multidrug-resistant pathogens.... Read morePatient Care
view channel
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read more
VR Training Tool Combats Contamination of Portable Medical Equipment
Healthcare-associated infections (HAIs) impact one in every 31 patients, cause nearly 100,000 deaths each year, and cost USD 28.4 billion in direct medical expenses. Notably, up to 75% of these infections... Read more
Portable Biosensor Platform to Reduce Hospital-Acquired Infections
Approximately 4 million patients in the European Union acquire healthcare-associated infections (HAIs) or nosocomial infections each year, with around 37,000 deaths directly resulting from these infections,... Read moreFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read moreHealth IT
view channel
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read moreBusiness
view channelMedtronic and GE HealthCare Broaden Alliance Across Monitoring and Care Solutions
Medtronic announced a multiyear renewal and significant expansion of its global strategic alliance with GE HealthCare across Patient Care Solutions. Building on more than three decades of collaboration,... Read more
Quantum Surgical Acquires NeuWave from Johnson & Johnson
Quantum Surgical announced that it has acquired NeuWave Medical from Johnson & Johnson. NeuWave’s microwave ablation system is used in percutaneous tumor ablation procedures, and the acquisition supports... Read more
Medtronic to Acquire Coronary Artery Medtech Company CathWorks
Medtronic plc (Galway, Ireland) has announced that it will exercise its option to acquire CathWorks (Kfar Saba, Israel), a privately held medical device company, which aims to transform how coronary artery... Read more