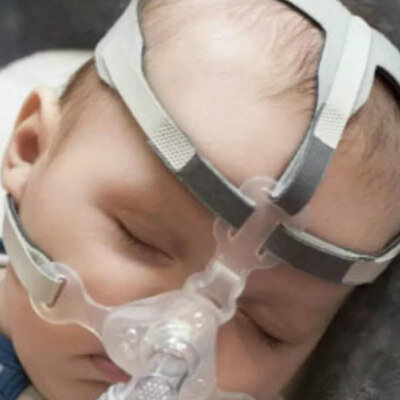
Disruptive Endovascular Solution Offers Minimally Invasive Alternative for Femoropopliteal Bypass Procedure
|
By HospiMedica International staff writers Posted on 10 May 2023 |

Currently, the "gold standard" treatment for patients with long blockages in the superficial femoral artery (SFA) is the invasive open femoropopliteal bypass. Endovascular techniques employed for these blockages often have high complication rates. Now, a new system offers patients an alternative treatment method, combining the advantages of both open and endovascular procedures.
The DETOUR System, developed by Endologix LLC (Irvine, CA, USA), enables Percutaneous Transmural Arterial Bypass (PTAB), a unique therapy that delivers a fully percutaneous femoropopliteal bypass through the femoral vein. Designed to treat patients with moderate to severe peripheral arterial disease and long SFA blockages, the DETOUR System employs the ENDOCROSS Device and TORUS Stent Graft.
The DETOUR2 Investigational Device Exemption (IDE) study showcased a 12-month primary patency rate of 72.1% in SFA lesions with an average length of 32.7cm. Notably, the trial achieved a 100% technical success rate in 202 patients, along with a 30-day Major Adverse Event Rate of 7%. This composite outcome includes death, CD-TLR, target limb amputation, symptomatic deep vein thrombosis (DVT), pulmonary embolism (PE), and the requirement for transfusion of packed red blood cells (PBRC) during the index procedure. The average hospital stay was just 1.1 days. The DETOUR System, currently an investigational device that has received FDA Breakthrough Device Designation, was recognized as the "Medical Device Engineering Breakthrough" winner at the 7th annual MedTech Breakthrough Awards.
“This award from MedTech Breakthrough is another strong validation of our strategy to expand our product portfolio into the large peripheral vascular market opportunity. Patients with long SFA blockages face tradeoffs with current treatment options. We know that open surgery of any kind is associated with complications that can negatively affect patient quality-of-life,” said Matt Thompson, MD, President and CEO of Endologix. “What makes PTAB with the DETOUR System truly unique is that, as demonstrated in our IDE study's 12-month results, it offers a minimally invasive therapy with comparable patency to open surgery while avoiding many of the complications associated with more invasive procedures. Once approved, the DETOUR System will offer a fully percutaneous femoropopliteal bypass and will provide a disruptive, innovative therapy for the treatment of long-segment SFA disease, thereby expanding the treatment options available for these patients.”
“While open surgery is still technically the gold standard for long-term durability, the reality is that not every patient is a candidate for that procedure and it can carry more risk for the patient,” said James Johnson, managing director, MedTech Breakthrough. “The DETOUR System has the potential to introduce a minimally invasive alternative for patients in need of a femoropopliteal bypass. The breakthrough solution may offer the best of both worlds: a durable, minimally invasive endovascular solution with the patency of open femoropopliteal bypass. The low MAE rate, coupled with good primary patency, provides supportive evidence for the feasibility of this new technology in a challenging patient population. Congratulations to the Endologix team on being for our 2023 ‘Medical Device Engineering Breakthrough’ award.”
Related Links:
Endologix LLC
Latest Critical Care News
- Heat-Activated Skin Patch Targets Melanoma Lesions
- Automated Dispensing System Enhances Medication Access and Efficiency
- Angiography-Based FFR Approach Matches Gold Standard Results Without Wires
- Eye Imaging AI Identifies Elevated Cardiovascular Risk
- Noninvasive Monitoring Device Enables Earlier Intervention in Heart Failure
- Automated IV Labeling Solution Improves Infusion Safety and Efficiency
- First-Of-Its-Kind AI Tool Detects Pulmonary Hypertension from Standard ECGs
- 4D Digital Twin Heart Model Improves CRT Outcomes
- AI Turns Glucose Data Into Actionable Insights for Diabetes Care
- Microscale Wireless Implant Tracks Brain Activity Over Time
- Smart Mask Delivers Continuous, Battery-Free Breath Monitoring
- Routine Blood Pressure Readings May Identify Risk of Future Cognitive Decline
- CGM-Based Algorithm Enhances Insulin Dose Adjustment in Type 2 Diabetes
- Fish Scale–Based Implants Offer New Approach to Corneal Repair
- Dual-Function Wound Patch Combines Infection Sensing and Treatment
- Smartwatch Signals and Blood Tests Team Up for Early Warning on Insulin Resistance
Channels
Artificial Intelligence
view channelAI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
Accurately identifying long-term cardiovascular disease risk in asymptomatic adults remains challenging for clinicians. Missed or underestimated risk delays preventive therapy and increases the chance... Read more
Machine Learning Approach Enhances Liver Cancer Risk Stratification
Hepatocellular carcinoma, the most common form of primary liver cancer, is often detected late despite targeted surveillance programs. Current screening guidelines emphasize patients with known cirrhosis,... Read moreSurgical Techniques
view channel
Endoscope Enables Fallopian Tube Imaging and Cell Collection for Ovarian Cancer Surveillance
Early detection of ovarian cancer remains challenging because symptoms are nonspecific and available screening tests often fail to identify disease at a curable stage. Many high‑grade serous carcinomas... Read more
New Modular Stem System Improves Fixation and Workflow in Total Ankle Replacement
Total ankle replacement demands stable fixation, preservation of bone stock, and consistent intraoperative efficiency to support durable outcomes. Surgeons often weigh exposure and soft-tissue disruption... Read more
Implantable Brain-Computer Interface Supports Stroke Recovery and Assistive Function
Stroke leaves many survivors with chronic motor deficits that limit independence, and cognitive decline is a growing concern in aging populations. Stroke is a leading cause of long-term disability in the... Read more
Patient-Specific Cardiac Digital Twin Guides Ventricular Tachycardia Ablation
Catheter ablation for ventricular tachycardia after myocardial infarction is lengthy, technically demanding, and prone to recurrence. Repeat procedures add scar burden and keep many patients on antiarrhythmic drugs.... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
Voice-Driven AI System Enables Structured GI Procedure Documentation
Documentation during gastrointestinal (GI) procedures often competes with real-time clinical decision-making and imposes a significant cognitive burden on physicians. Manual data entry and post-procedure... Read more
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read moreBusiness
view channel
New Partnership Expands Access to Predictive Tool for Patient Monitoring
Spacelabs Healthcare has signed an agreement with DEPTH Health, Inc. to make the Rothman Index available to hospitals and health systems through DEPTH’s Real-Time Advisor for Clinical Expert Routing (RACER)... Read more