AI Tool Accurately Predicts Cancer Three Years Prior to Diagnosis
|
By HospiMedica International staff writers Posted on 24 Aug 2023 |

Over the past five decades, there has been a surge in cases of a specific type of esophageal and stomach cancer - esophageal adenocarcinoma (EAC) and gastric cardia adenocarcinoma (GCA). Both cancers have high fatality rates, although preventive measures can make a difference. Screenings can detect pre-cancerous changes, such as Barrett’s esophagus, often identified in individuals with long-standing gastroesophageal reflux disease (GERD). Although guidelines suggest screening for high-risk patients, many healthcare providers remain unfamiliar with this recommendation. Now, a new artificial intelligence (AI) tool offers accurate predictions for these forms of esophageal and stomach cancer at least three years in advance of a diagnosis.
Researchers at Michigan Medicine (Ann Arbor, MI, USA) have developed an automated tool integrated into the electronic health record (EHR). This tool has the potential to bridge the awareness gap between healthcare providers and patients with an elevated risk of developing EAC and GCA. The researchers employed a specific type of AI for analyzing data related to EAC and GCA rates across more than 10 million U.S. veterans in order to develop and validate the Kettles Esophageal and Cardia Adenocarcinoma prediction tool, or K-ECAN for short.
K-ECAN leverages readily accessible data from the EHR, including patient demographics, weight, prior diagnoses, and routine lab results, to gauge an individual's risk of developing EAC and GCA. Outperforming published guidelines and previously validated prediction tools, K-ECAN accurately predicts cancer at least three years ahead of a diagnosis. Integrating this AI tool into the EHR could automatically notify healthcare providers about patients at a higher risk of developing EAC and GCA.
“Symptoms of GERD, like heartburn, are an important risk factor for esophageal adenocarcinoma,” said Joel Rubenstein, M.D., M.S., professor of internal medicine at Michigan Medicine. “But most people with GERD symptoms will never develop esophageal adenocarcinoma and gastric cardia adenocarcinoma. In addition, roughly half of the patients with this form of cancer never experienced prior GERD symptoms at all. This makes K-ECAN particularly useful because it can identify people who are at elevated risk, regardless of whether they have GERD symptoms or not.”
Related Links:
Michigan Medicine
Latest AI News
- Automated Phone Speech Test Identifies Alzheimer’s Pathology for Prescreening
- FDA-Cleared AI System Detects Sepsis Earlier and Reduces Mortality
- Facial Image Analysis Tracks Biological Aging, Predicts Cancer Outcomes
- AI Model Uses Eye Imaging to Identify Risk of Major Systemic Diseases
- AI Platform Interprets Real-Time Wearable Data for Parkinson’s Management
- Algorithm Identifies Cardiac Arrest Hotspots to Guide AED Placement
- AI Analysis of Pericardial Fat Refines Long-Term Heart Disease Risk
- Machine Learning Approach Enhances Liver Cancer Risk Stratification
- New AI Approach Monitors Brain Health Using Passive Wearable Data
- AI Tool Maps Early Risk Patterns in Bloodstream Infections
- AI Model Identifies Rare Endocrine Disorder from Hand Images
Channels
Critical Care
view channel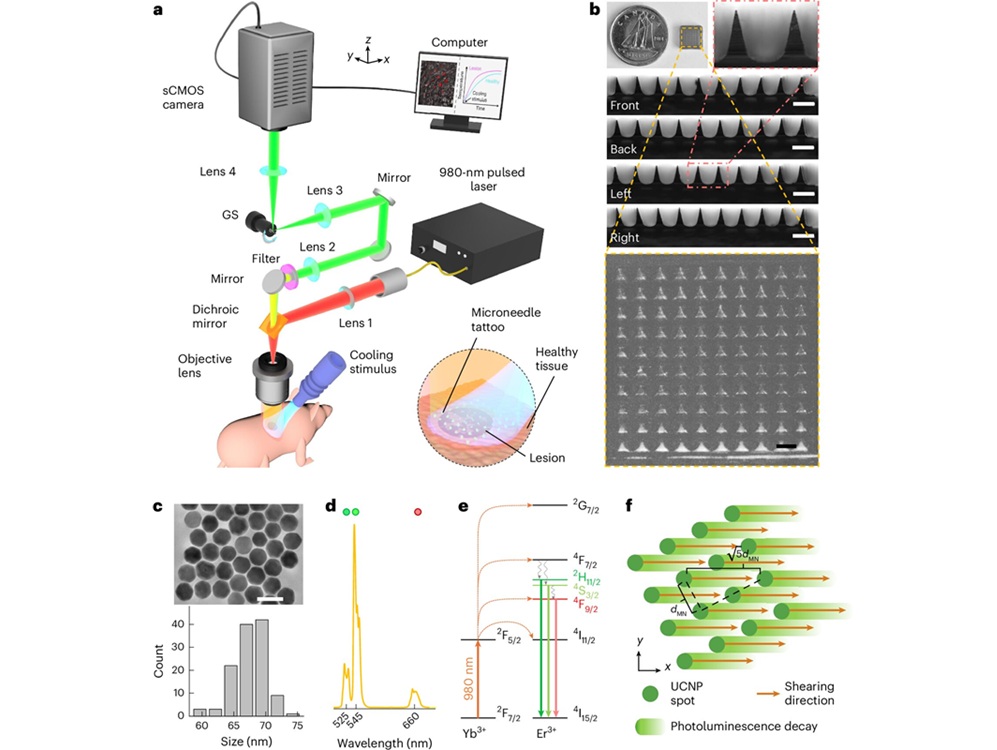
“Intelligent Tattoo” Method Detects Early Melanoma Signals
Melanoma is a lethal skin cancer in which earlier detection drives better outcomes. Current evaluation relies on visual inspection followed by biopsy, which can miss nascent lesions and lead to unnecessary... Read more
Reusable Intermittent Catheters Reduce Antibiotic Use Without Increasing Urinary Tract Infections
Intermittent self-catheterization, used to empty the bladder several times a day, can leave patients vulnerable to recurrent urinary tract infections and repeated antibiotic use. Reliance on single-use... Read moreSurgical Techniques
view channel
Pink Noise Stimulation Approach Could Support Safer Anesthesia
Maintaining stable unconsciousness during general anesthesia while minimizing drug exposure remains a core perioperative challenge. Sudden nociceptive surges can disrupt anesthetic depth, increasing neurological... Read more
Surgical Robotic System Gains CE Mark for Minimally Invasive Procedures
Cornerstone Robotics’ (CSR; Hong Kong, China) Sentire Endoscopic Surgical System (Sentire Surgical System) has received European Union CE mark certification under the Medical Device Regulation (MDR) for... Read morePatient Care
view channel
AI Avatar Doctor Improves Patient Understanding Before Radiotherapy
Radiation oncology consultations require patients to grasp complex concepts quickly, yet anxiety and information overload often undermine understanding and informed consent. Poor comprehension can also... Read more
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read moreHealth IT
view channel
AI-Native EHR Achieves EU Medical Device Certification
InterSystems (Boston, MA, USA) announced that its IntelliCare electronic health record (EHR) solutions have been certified as Class IIa medical devices under the European Union Medical Device Regulation... Read more
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read morePoint of Care
view channel