US FDA Demands Proof of Antibacterial Soap Claims
|
By HospiMedica International staff writers Posted on 02 Jan 2014 |
Manufacturers of nonprescription antibacterial hand soaps and body washes will soon be required to show their products are more effective than plain soap in stopping the spread of infections.
The demand made by the US Food and Drug Administration (FDA; Silver Spring, MD, USA) does not apply to hand sanitizers and wipes, which are alcohol-based and are not used with water, or to antibacterial products used in the healthcare setting. Failing to meet these standards would mean that the antiseptic ingredients used in the products would have to be removed from over-the-counter (OTC) products, or that antibacterial claims are removed from product labeling. The FDA has been reviewing antibacterial active ingredients for the past several years, and hopes to finalize the rule by late 2016.
Over 2,200 antibacterial hand soaps and body washes are currently available for consumers in the United States alone, but there is no scientific evidence showing these products are any more effective at preventing illness than washing hands with plain soap and water. In fact, some data suggest that long-term exposure to certain active ingredients used in antibacterial products, such as triclosan and triclocarban, engender health risks such as bacterial resistance or hormonal imbalances (for example with estrogen, testosterone, and thyroid hormones).
“In order for antibacterial soaps and body washes to be considered generally recognized as effective, manufacturers would be required to conduct clinical trials demonstrating that their products are more effective than plain soap and water in preventing illness and the spread of certain infections when they're used by consumers,” said Sandra Kweder, MD, deputy director of the Office of New Drugs at the FDA's Center for Drug Evaluation and Research (CDER).
“Antibacterial soaps and body washes are used widely and frequently by consumers in everyday home, work, school, and public settings, where the risk of infection is relatively low,” said Janet Woodcock, MD, director of the CDER. “Due to consumers’ extensive exposure to the ingredients in antibacterial soaps, we believe there should be a clearly demonstrated benefit from using antibacterial soap to balance any potential risk.”
The proposed rule does not require the antibacterial soap products to be removed from the market at this time. When the proposed rule is finalized, either companies will have provided data to support an antibacterial claim, or if not, they will have to reformulate or re-label these products in order to continue marketing.
Related Links:
US Food and Drug Administration
The demand made by the US Food and Drug Administration (FDA; Silver Spring, MD, USA) does not apply to hand sanitizers and wipes, which are alcohol-based and are not used with water, or to antibacterial products used in the healthcare setting. Failing to meet these standards would mean that the antiseptic ingredients used in the products would have to be removed from over-the-counter (OTC) products, or that antibacterial claims are removed from product labeling. The FDA has been reviewing antibacterial active ingredients for the past several years, and hopes to finalize the rule by late 2016.
Over 2,200 antibacterial hand soaps and body washes are currently available for consumers in the United States alone, but there is no scientific evidence showing these products are any more effective at preventing illness than washing hands with plain soap and water. In fact, some data suggest that long-term exposure to certain active ingredients used in antibacterial products, such as triclosan and triclocarban, engender health risks such as bacterial resistance or hormonal imbalances (for example with estrogen, testosterone, and thyroid hormones).
“In order for antibacterial soaps and body washes to be considered generally recognized as effective, manufacturers would be required to conduct clinical trials demonstrating that their products are more effective than plain soap and water in preventing illness and the spread of certain infections when they're used by consumers,” said Sandra Kweder, MD, deputy director of the Office of New Drugs at the FDA's Center for Drug Evaluation and Research (CDER).
“Antibacterial soaps and body washes are used widely and frequently by consumers in everyday home, work, school, and public settings, where the risk of infection is relatively low,” said Janet Woodcock, MD, director of the CDER. “Due to consumers’ extensive exposure to the ingredients in antibacterial soaps, we believe there should be a clearly demonstrated benefit from using antibacterial soap to balance any potential risk.”
The proposed rule does not require the antibacterial soap products to be removed from the market at this time. When the proposed rule is finalized, either companies will have provided data to support an antibacterial claim, or if not, they will have to reformulate or re-label these products in order to continue marketing.
Related Links:
US Food and Drug Administration
Latest Critical Care News
- Smart Capsule Offers Real-Time Profiling Across GI Tract
- Ultra-Thin Implant Helps Patients with Spinal Cord Injury Recover Lost Functions
- Portable Cell Therapy Device to Enable Rapid On-Demand Modification of RBCs at POC
- Monitoring Airborne Fungal Spores Could Help Predict COVID-19 & Flu Surges
- New System Measures Blood Sodium Without Needles
- Sleep Data from Wearable Device May Help Predict Preterm Birth
- AI Tool Interprets Echocardiograms in Minutes
- Electrochemical Catheter Hub Prevents Bloodstream Infections
- Noninvasive Double Microbubble Delivery Approach Marks Breakthrough in Brain Cancer Treatment
- Self-Healing Skin-Like Material to Find Applications in Health Monitoring, Surgery and Implants
- Highly-Sensitive Electronic Skin Allows Robots to Feel Heat, Pain and Pressure

- AI-Powered Wearable Sensor Predicts Labor Onset in Pregnant Women
- Implantable Device to Redefine Continuous Glucose Monitoring
- Smart Microgel Could Repair and Replace Damaged Organs
- Smart Breath Tracker Wristband to Revolutionize Respiratory Care
- Stronger Blood Clot Prevention Measures Needed After Leg Artery Procedures in High-Risk Patients
Channels
Surgical Techniques
view channel
First-Ever Technology Makes Blood Translucent During Surgery
No matter the discipline or scale, bleeding is a regular part of any surgery and can create several challenges. In operating room imaging, seeing through blood in real-time during a surgery has been a... Read more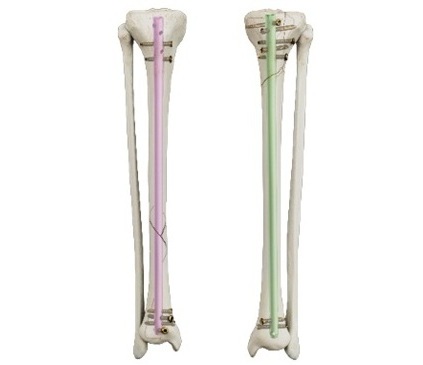
Tibia Nailing System with Novel Side-Specific Nails to Revolutionize Fracture Surgery
Smith+Nephew (Hull, UK;) has launched its new TRIGEN MAX Tibia Nailing System for stable and unstable fractures of the tibia, including the shaft. It is the only system to now offer trauma surgeons the... Read morePatient Care
view channel
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read more
VR Training Tool Combats Contamination of Portable Medical Equipment
Healthcare-associated infections (HAIs) impact one in every 31 patients, cause nearly 100,000 deaths each year, and cost USD 28.4 billion in direct medical expenses. Notably, up to 75% of these infections... Read more
Portable Biosensor Platform to Reduce Hospital-Acquired Infections
Approximately 4 million patients in the European Union acquire healthcare-associated infections (HAIs) or nosocomial infections each year, with around 37,000 deaths directly resulting from these infections,... Read moreFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read moreHealth IT
view channel
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read more
Smartwatches Could Detect Congestive Heart Failure
Diagnosing congestive heart failure (CHF) typically requires expensive and time-consuming imaging techniques like echocardiography, also known as cardiac ultrasound. Previously, detecting CHF by analyzing... Read moreBusiness
view channel
Bayer and Broad Institute Extend Research Collaboration to Develop New Cardiovascular Therapies
A research collaboration will focus on the joint discovery of novel therapeutic approaches based on findings in human genomics research related to cardiovascular diseases. Bayer (Berlin, Germany) and... Read more