The Randox Laboratories Diagnostic Test Distinguishes Coronavirus from Other Respiratory Infections
By HospiMedica International staff writers
Posted on 12 Mar 2020
The global health diagnostics company Randox Laboratories (Crumlin, United Kingdom) has announced the release of a rapid diagnostic test for the COVID-19 strain of coronavirus.Posted on 12 Mar 2020
A novel coronavirus of zoonotic origin, the 2019-nCoV virus (the causative agent of COVID-19 disease) was recently identified in patients with acute respiratory disease. This virus is genetically similar to SARS coronavirus and bat SARS-like coronaviruses. The outbreak was initially detected in Wuhan, a major city of China, but has subsequently been detected in other provinces of China as well as in several other countries. At this time more than 70,000 cases of the disease have been confirmed with over 2,000 fatalities.
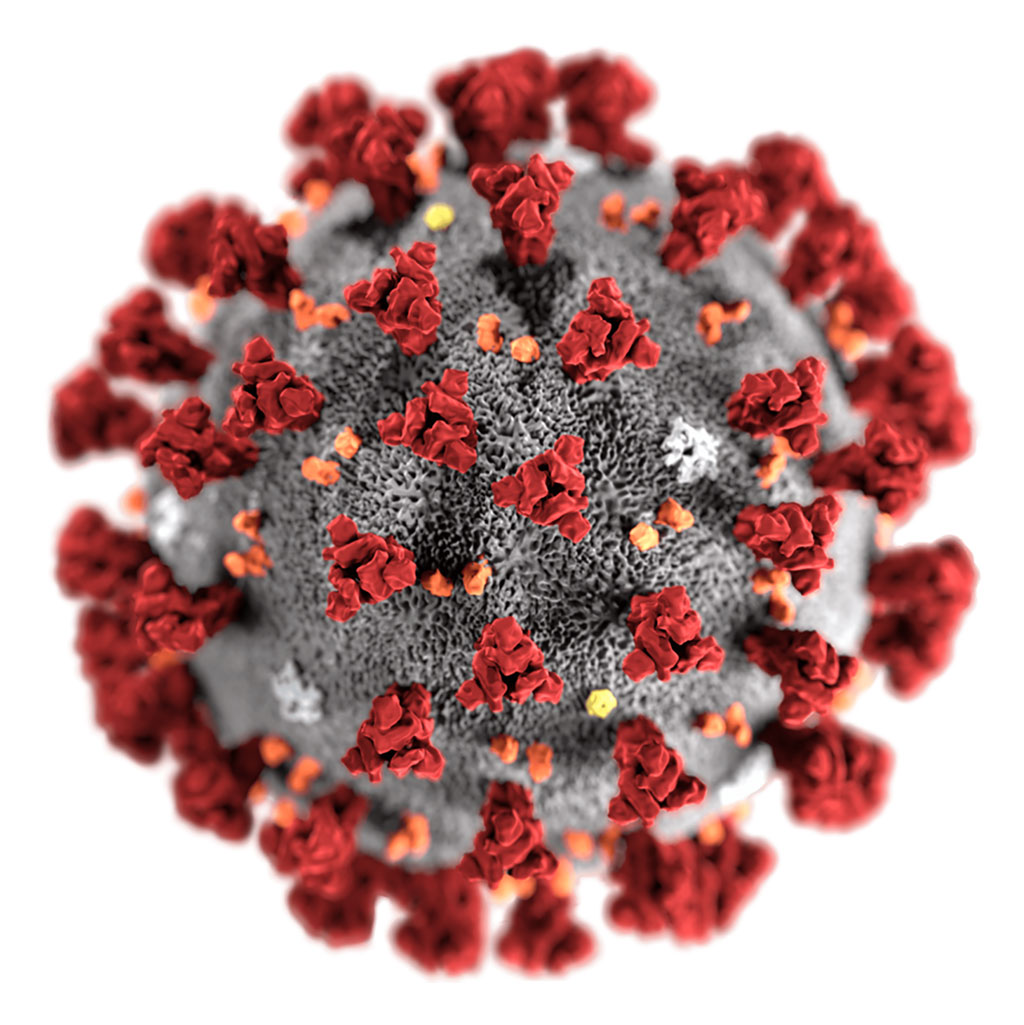
Image: Illustration of a 2019- novel coronavirus (nCoV) virion (Photo courtesy of Alissa Eckert, MS/CDC).
Signs of infection are highly non-specific and these include respiratory symptoms, fever, cough, dyspnea, and viral pneumonia. Currently, experts estimate that only a small percentage of coronavirus cases have actually been diagnosed clinically. Therefore, diagnostic tests specific for this infection are urgently needed for confirming suspected cases, screening patients, and conducting virus surveillance.
In this regard Randox Laboratories, has adapted its patented Biochip Array Technology (BAT) for diagnosis of coronavirus. BAT is an innovative multiplexing technology which is utilized within the cartridge aspect of an autoanalyzer platform. The Biochip allows for the simultaneous detection of multiple analytes from a single sample.
The Randox COVID-19 test is available for the Evidence Investigator autoanalyzer with five hours turnaround time. An additional enhanced multiplex array also includes tests for other respiratory viruses, which display similar symptoms.
This Biochip tests simultaneously, from one patient sample, for the viruses below:
• COVID-19 (previously named 2019-nCoV)
• Coronavirus 229E/NL63
• Coronavirus OC43/HKUI
• Middle East Respiratory Syndrome Coronavirus (MERS-CoV)
• Pan-Coronavirus (SARS, SARS like, COVID-19)
• Adenovirus A/B/C/D/E
• Enterovirus A/B/C
• Influenza A
• Influenza B
• Rhinovirus A/B
Dr. Peter FitzGerald, managing director of Randox Laboratories, said, “Current technologies for the diagnosis of coronavirus are designed simply to detect the presence or lack of COVID-19, and therefore neglect to differentiate between this strain and other respiratory infections. We have therefore developed an extended Viral Respiratory Infection Array that tests simultaneously for COVID-19 and nine other viruses. This will eliminate the need for multiple back-and-forth tests before the root cause of symptoms is found, and empower clinicians to make fast and informed decisions.”
Related Links:
Randox Laboratories