Luminex Receives BARDA Contract to Support Development of Second, Rapid SARS-CoV-2 Test
By HospiMedica International staff writers
Posted on 02 Apr 2020
Luminex Corporation (Austin, TX, USA) has received USD 642,450 in funding from the Biomedical Advanced Research and Development Authority (BARDA) to support the development, testing, and submission or Emergency Use Authorization (EUA) for its ARIES SARS-CoV-2 Assay. This assay will run on the company's sample-to-answer ARIES System, an FDA-cleared, automated molecular diagnostics platform for moderate complexity labs. Luminex had recently received a BARDA contract and an FDA EUA for its new NxTAG CoV Extended Panel, a high-throughput test for detecting SARS-CoV-2 that provides results for up to 96 samples in approximately four hours. The NxTAG CoV Extended Panel runs on Luminex's easy-to-use, compact MAGPIX System.Posted on 02 Apr 2020
Luminex empowers labs to obtain reliable, timely, and actionable answers, ultimately advancing health through its wide range of solutions applicable in diverse markets, including clinical diagnostics, pharmaceutical drug discovery, biomedical research, genomic and proteomic research, biodefense research, and food safety. BARDA is part of the US Department of Health and Human Services and is tasked with protecting the country against emerging infectious diseases and other threats. Through public-private partnerships, BARDA supports the development of vaccines, drugs, and diagnostics.

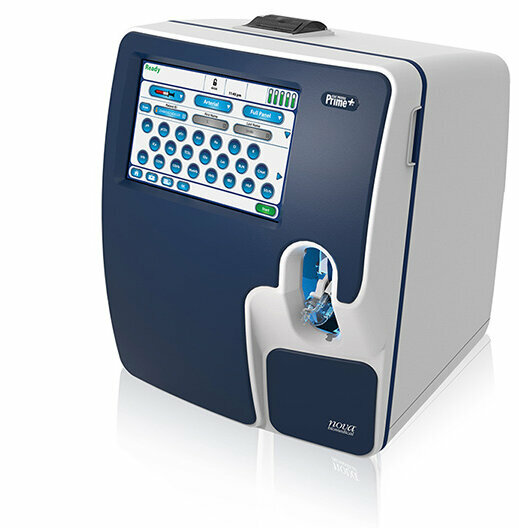
Image: ARIES Systems (Photo courtesy of Luminex Corporation)
Luminex’s ARIES SARS-CoV-2 Assay is designed to provide rapid answers in patients believed to have COVID-19, generating results in approximately two hours. It can be run on 6-unit and 12-unit ARIES Systems for labs seeking a medium-throughput solution with minimal hands-on time required. The assay will improve upon existing laboratory-developed tests (LDTs) for SARS-CoV-2 on the ARIES System by eliminating the need to purchase and incorporate additional reagents, making the test easier to run and allowing labs to start testing immediately upon performance verification. Luminex intends to price the ARIES SARS-CoV-2 Assay below current government reimbursement levels in order to prevent additional financial burden on customers and the healthcare system during the COVID-19 pandemic.
"Throughout this challenging time, the Luminex team has been laser-focused on contributing to the global response to the COVID-19 global outbreak," said Nachum "Homi" Shamir, President and CEO of Luminex. "We thank BARDA for supporting our efforts to develop and quickly launch this new test and our recently EUA-cleared NxTAG CoV Extended Panel. Luminex is now on a run-rate to deliver up to 500,000 tests per month, which are primarily focused on helping our customers fight this global pandemic."
Related Links:
Luminex Corporation