Non-Eluting Coating Actively Kills Bacteria Contaminating Surface of Medical Devices
|
By HospiMedica International staff writers Posted on 28 Jun 2024 |

Millions of medical devices are used or implanted in patients worldwide annually. Despite rigorous sterilization protocols, device-related infections are increasingly problematic. Research indicates that 70-100% of devices removed from joint implants due to failure show contamination from various sources. Operating rooms, despite strict cleanliness standards, often harbor bacteria, leading to high risks of implant failures and infections. These infections can result in pain, reduced mobility, costly additional surgeries, and even fatalities. Now, a novel antibacterial treatment can be applied to any implantable device for robust protection against microbes responsible for most orthopedic device-related issues.
Orthobond Corporation (Monmouth Junction, NJ, USA) has pioneered a technique to covalently-bond its antibacterial molecule to implant surfaces. Using advanced covalent linker technology, the Ostaguard treatment delivers effective antimicrobial protection directly on the device surface, combating a broad spectrum of pathogens including S. aureus, MRSA, S. epidermidis, P. acnes, P. aeruginosa, E. coli, and C. albicans. Ostaguard has been rigorously tested on multiple surfaces against 12 microbes that account for nearly all device-related infections, demonstrating high effectiveness in eliminating several bacteria strains likely to be encountered in surgical settings while remaining harmless to mammalian cells.
The Ostaguard antibacterial surface treatment is versatile enough for application on various device surfaces including those used in joint reconstruction, neuromodulation, oncology, sports medicine, plastic surgery, and cardiovascular procedures. This proprietary technology is integrated during the manufacturing phase before the device is packaged and sterilized. The U.S. Food and Drug Administration (FDA) has approved the De Novo marketing request for Ostaguard to be used on permanent medical devices, marking this as the first FDA-approved non-eluting coating that actively kills bacteria on a medical device surface. The initial application approved is for orthopedic spinal implants.
“We are proud of our evidence-based submission to the FDA and look forward to making our innovative technology available to a wide variety of customers. We believe our surface coating has the potential to be the standard of care for implantable devices to protect patients from pathogens, and that this will be transformative to the field of surgery and beyond,” said David Nichols, CEO of Orthobond. “This marks a huge moment in the history of Orthobond and brings to fruition the vision of our co-founders, Gregory Lutz, M.D. and Jeffrey Schwartz, Ph.D., who were committed to helping patients and physicians by providing a much needed and easy-to-implement antibacterial option to protect implants. This milestone also provides further testament to Gregory’s legacy and we are proud to be able to carry it on into this next chapter.”
Related Links:
Orthobond Corporation
Latest Surgical Techniques News
- Easy-To-Apply Gel Could Prevent Formation of Post-Surgical Abdominal Adhesions
- Groundbreaking Leadless Pacemaker to Prevent Invasive Surgeries for Children
- Spectroscopy Technique Improves Surgery for Pediatric Epilepsy Patients
- Bioengineered Arteries Show Promise for Cardiovascular Surgery
- Online Tool Guides Surgical Decisions for Gallbladder Cancer
- Innovative Technology Enables Rapid Life-Saving Surgical Leak Detection
- First-Of-Its-Kind Bioresorbable Implant to Help Children with Rare Respiratory Disease
- Screw-Shaped Magnetic Microrobots to Transform Treatment for Patients with Inoperable Blood Clots
- "Ultra-Rapid" Testing in the OR Could Enable Accurate Removal of Brain Tumors
- Automated Endoscopic Device Obtains Improved Biopsy Results in Single Pass
- World's First Machine Learning Model Combats Wrong-Site Surgery
- Novel Method Combining Heart Biopsy and Device Implantation Reduces Complications Risk
- New Surface Coating Could Prevent Blood Clotting in Medical Devices and Implants

- Dumbbell-Shaped Thrombectomy Device Offers Novel Approach to Cerebral Venous Sinus Thrombosis Treatment
- Novel Catheter Mimics Snake Teeth to Grab Blood Clots
- New Laparoscopic Imaging Technique Accurately Maps Biological Tissue for Minimally Invasive Surgery
Channels
Artificial Intelligence
view channel
Innovative Risk Score Predicts Heart Attack or Stroke in Kidney Transplant Candidates
Heart researchers have utilized an innovative risk assessment score to accurately predict whether patients being evaluated for kidney transplants are at risk for future major cardiac events, such as a... Read more
AI Algorithm Detects Early-Stage Metabolic-Associated Steatotic Liver Disease Using EHRs
Liver disease, which is treatable when detected early, often goes unnoticed until it reaches advanced stages. Metabolic-associated steatotic liver disease (MASLD), the most prevalent form of liver disease,... Read moreCritical Care
view channel
New IV Pole Improves Safety and Ease of Administering IV Medications at Hospital Bedside
Preventable medication errors affect around 500,000 hospitalized patients in the U.S. every year. A significant portion of these errors occur with intravenous (IV) smart pumps, which require a precise... Read more
Battery-Powered Wearable Device Monitors Joint Pain
Ankle injuries are common among active Americans. Each day, approximately 25,000 people experience ankle sprains, and 25% of Americans regularly deal with knee pain. However, physical therapy can be both... Read more
Wireless Pacifier Monitors Vitals of NICU Babies Without Need for Painful Blood Draws
Newborns require frequent monitoring of their vital signs, with electrolyte levels being one of the most important indicators of their health. Currently, the only method to monitor these levels is through... Read morePatient Care
view channel
Portable Biosensor Platform to Reduce Hospital-Acquired Infections
Approximately 4 million patients in the European Union acquire healthcare-associated infections (HAIs) or nosocomial infections each year, with around 37,000 deaths directly resulting from these infections,... Read moreFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read more
Smartwatches Could Detect Congestive Heart Failure
Diagnosing congestive heart failure (CHF) typically requires expensive and time-consuming imaging techniques like echocardiography, also known as cardiac ultrasound. Previously, detecting CHF by analyzing... Read morePoint of Care
view channel
Handheld, Sound-Based Diagnostic System Delivers Bedside Blood Test Results in An Hour
Patients who go to a doctor for a blood test often have to contend with a needle and syringe, followed by a long wait—sometimes hours or even days—for lab results. Scientists have been working hard to... Read more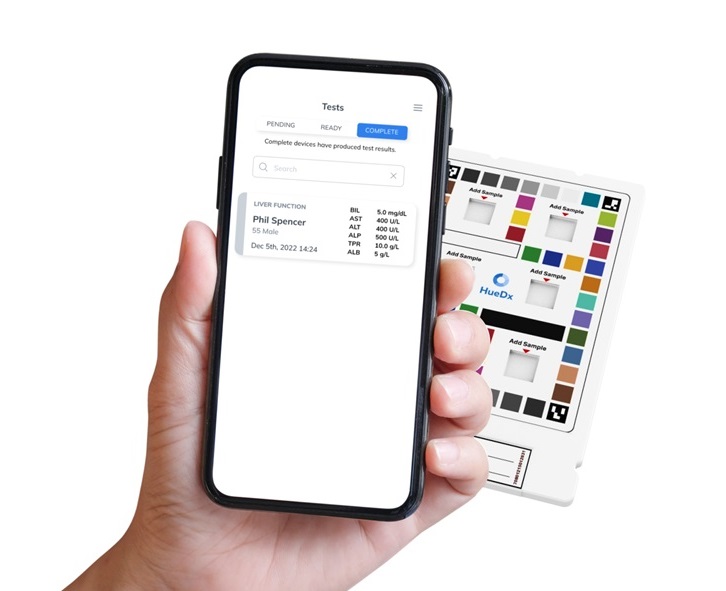
Smartphone-Enabled, Paper-Based Quantitative Diagnostic Platform Transforms POC Testing
Point-of-care diagnostics are crucial for public health, offering rapid, on-site testing that enables prompt diagnosis and treatment. This is especially valuable in remote or underserved regions where... Read moreBusiness
view channel
Becton Dickinson to Spin Out Biosciences and Diagnostic Solutions Business
Becton, Dickinson and Company (BD, Franklin Lakes, NJ, USA), has announced that its board of directors has unanimously authorized BD management to pursue a plan to separate BD's Biosciences and Diagnostic... Read more