POC Sepsis Test Delivers Fast, Accurate and User-Friendly Results in Critical Care Settings
|
By HospiMedica International staff writers Posted on 07 Oct 2024 |

Sepsis is a critical global health issue, impacting 50 million people annually and causing 11 million deaths, which accounts for 20% of all global fatalities. Sepsis is an emergency where time is of the essence, and up to 80% of sepsis-related deaths are preventable. However, it remains notoriously difficult to diagnose due to its non-specific symptoms, which often mimic other common conditions. Early detection is essential for initiating the appropriate treatment protocols and improving patient survival rates. Pancreatic Stone Protein (PSP) has emerged as a promising biomarker for the early detection of sepsis, identifying the condition 24-48 hours earlier than current methods. PSP, produced by the pancreas and immune cells, rises in response to infection and inflammation and has shown significant sensitivity and specificity in detecting sepsis, particularly in critically ill patients. Clinical studies have found that elevated PSP levels are closely linked to the progression of sepsis, enabling healthcare providers to initiate life-saving treatments sooner and improve patient outcomes.
Abionic (Epalinges, Switzerland) has developed the IVD CAPSULE PSP test, which offers rapid, accurate, and easy-to-use results in critical care settings by measuring PSP levels. These measurements are directly tied to a patient's sepsis risk, empowering clinicians to make well-informed decisions and initiate timely treatments. The IVD CAPSULE PSP operates exclusively on Abionic’s abioSCOPE platform, a near-patient rapid diagnostic system that integrates smoothly into clinical workflows. It provides lab-quality results from a drop of blood within minutes, delivering valuable insights and actionable information directly at the point of care.
To perform the test, a trained healthcare provider can obtain 50 μL of capillary blood via a simple finger prick without the need for a phlebotomist to collect venous blood. The sample is placed into an abioMIX tube, shaken 10 times for even mixing, and then applied to the capsule. The biomarker levels are quickly measured with high precision and accuracy, delivering lab-quality results within minutes at the point of care. The IVD CAPSULE PSP test has been granted 510(k) clearance by the U.S. Food and Drug Administration (FDA), expediting the detection of sepsis. Already certified under the EU IVDR as of July 2022, this FDA approval allows Abionic to expand into the U.S. market.
“Achieving FDA 510(k) clearance for IVD CAPSULE PSP marks a significant milestone for Abionic and confirms our ability to meet the need for quick & reliable sepsis testing,” said Patrick Pestalozzi, CEO at Abionic. “This clearance will allow us to deploy our solutions across the United States and provide clinicians in acute care settings with a proven solution to accelerate the Time-To-Detection of sepsis.”
Related Links:
Abionic
Latest Point of Care News
Channels
Critical Care
view channel
Ingestible Smart Capsule for Chemical Sensing in the Gut Moves Closer to Market
Intestinal gases are associated with several health conditions, including colon cancer, irritable bowel syndrome, and inflammatory bowel disease, and they have the potential to serve as crucial biomarkers... Read moreNovel Cannula Delivery System Enables Targeted Delivery of Imaging Agents and Drugs
Multiphoton microscopy has become an invaluable tool in neuroscience, allowing researchers to observe brain activity in real time with high-resolution imaging. A crucial aspect of many multiphoton microscopy... Read more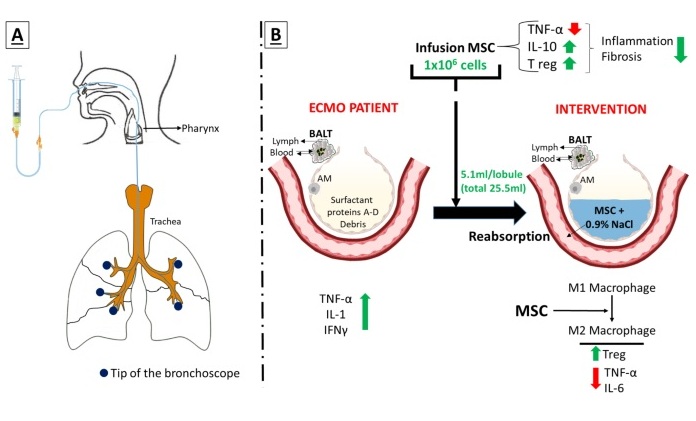
Novel Intrabronchial Method Delivers Cell Therapies in Critically Ill Patients on External Lung Support
Until now, administering cell therapies to patients on extracorporeal membrane oxygenation (ECMO)—a life-support system typically used for severe lung failure—has been nearly impossible.... Read moreSurgical Techniques
view channel
Intravascular Imaging for Guiding Stent Implantation Ensures Safer Stenting Procedures
Patients diagnosed with coronary artery disease, which is caused by plaque accumulation within the arteries leading to chest pain, shortness of breath, and potential heart attacks, frequently undergo percutaneous... Read more
World's First AI Surgical Guidance Platform Allows Surgeons to Measure Success in Real-Time
Surgeons have always faced challenges in measuring their progress toward surgical goals during procedures. Traditionally, obtaining measurements required stepping out of the sterile environment to perform... Read morePatient Care
view channel
Portable Biosensor Platform to Reduce Hospital-Acquired Infections
Approximately 4 million patients in the European Union acquire healthcare-associated infections (HAIs) or nosocomial infections each year, with around 37,000 deaths directly resulting from these infections,... Read moreFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read more
Smartwatches Could Detect Congestive Heart Failure
Diagnosing congestive heart failure (CHF) typically requires expensive and time-consuming imaging techniques like echocardiography, also known as cardiac ultrasound. Previously, detecting CHF by analyzing... Read moreBusiness
view channel
Expanded Collaboration to Transform OR Technology Through AI and Automation
The expansion of an existing collaboration between three leading companies aims to develop artificial intelligence (AI)-driven solutions for smart operating rooms with sophisticated monitoring and automation.... Read more