New Rapid COVID-19 Test Beats ELISA Method in Identifying SARS-CoV-2 Antibodies in Blood Within Just 12 Minutes
|
By HospiMedica International staff writers Posted on 10 Feb 2021 |
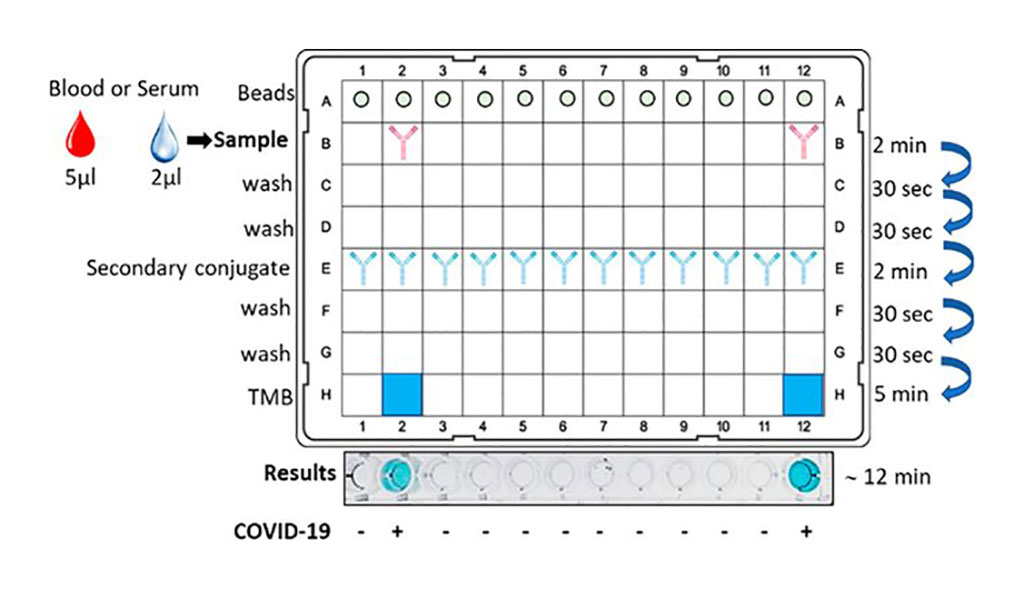
Image: New Rapid COVID-19 Test Beats ELISA Method in Identifying SARS-CoV-2 Antibodies in Blood within Just 12 Minutes (Photo courtesy of American Chemical Society)
A new rapid COVID-19 test to detect coronavirus antibodies beats the enzyme linked immunosorbent assay (ELISA) method in delivering results within just 12 minutes, marking a milestone in the development of immunological diagnostics.
An international research team involving the University of Paraná (Paraná, Brazil) and the University of Tübingen (Tübingen, Germany) has developed the new rapid test that can reliably identify COVID-19 antibodies in the blood within minutes. The new process is based on a simple measuring principle, making it easy to carry out without expensive instruments and is therefore, suitable for use at mobile testing centers or by laboratories in less economically developed regions. The new diagnostic method is also far faster than the ELISA procedure, which for decades has been seen as the gold standard for laboratory diagnosis of antibodies.
The new test is based on magnetic nanoparticles that are coated with viral antigens. To conduct the test, blood serum or blood is applied to the test surface. After roughly two minutes, the nanoparticles are washed and treated with a developer reagent. If the blood sample displays antibodies to coronavirus, a color change occurs. While the traditional ELISA test produces results after about three hours, study results show that the new method only takes 12 minutes. Positive and negative results can be established simply with the naked eye, and by using additional instruments, such as a microplate reader, the precision of the test can be further increased. The total response time is 15 times shorter than that of the classic ELISA test, which means that hundreds of samples can be tested in just a few hours.
In addition, the study shows that the new technology can also be applied to the serological diagnosis of other diseases. There are no reports in research literature on an immunology test for COVID-19 that delivers data as quickly, as precisely and above all as cheaply. The researchers believe that it will be possible to offer the new test at a comparable price to the ELISA test in future.
“Only a small sample is needed for the test: a single drop that contains two microliters of serum is sufficient,” said the study’s lead author, Professor Luciano F. Huergo from the University of Paraná: “It’s also possible to use whole blood, in other words the separation of soluble blood components that is normally necessary can be omitted. It isn’t absolutely necessary to have a fully-equipped laboratory or use special equipment to carry it out.”
“Another advantage over the ELISA procedure is that the color result of our new procedure is directly proportional to the concentration of antibodies,” added Huergo. “In other words, the new method delivers data on the quantity of antibodies and not just whether any are present.”
Related Links:
University of Paraná
University of Tübingen
An international research team involving the University of Paraná (Paraná, Brazil) and the University of Tübingen (Tübingen, Germany) has developed the new rapid test that can reliably identify COVID-19 antibodies in the blood within minutes. The new process is based on a simple measuring principle, making it easy to carry out without expensive instruments and is therefore, suitable for use at mobile testing centers or by laboratories in less economically developed regions. The new diagnostic method is also far faster than the ELISA procedure, which for decades has been seen as the gold standard for laboratory diagnosis of antibodies.
The new test is based on magnetic nanoparticles that are coated with viral antigens. To conduct the test, blood serum or blood is applied to the test surface. After roughly two minutes, the nanoparticles are washed and treated with a developer reagent. If the blood sample displays antibodies to coronavirus, a color change occurs. While the traditional ELISA test produces results after about three hours, study results show that the new method only takes 12 minutes. Positive and negative results can be established simply with the naked eye, and by using additional instruments, such as a microplate reader, the precision of the test can be further increased. The total response time is 15 times shorter than that of the classic ELISA test, which means that hundreds of samples can be tested in just a few hours.
In addition, the study shows that the new technology can also be applied to the serological diagnosis of other diseases. There are no reports in research literature on an immunology test for COVID-19 that delivers data as quickly, as precisely and above all as cheaply. The researchers believe that it will be possible to offer the new test at a comparable price to the ELISA test in future.
“Only a small sample is needed for the test: a single drop that contains two microliters of serum is sufficient,” said the study’s lead author, Professor Luciano F. Huergo from the University of Paraná: “It’s also possible to use whole blood, in other words the separation of soluble blood components that is normally necessary can be omitted. It isn’t absolutely necessary to have a fully-equipped laboratory or use special equipment to carry it out.”
“Another advantage over the ELISA procedure is that the color result of our new procedure is directly proportional to the concentration of antibodies,” added Huergo. “In other words, the new method delivers data on the quantity of antibodies and not just whether any are present.”
Related Links:
University of Paraná
University of Tübingen
Latest COVID-19 News
- Low-Cost System Detects SARS-CoV-2 Virus in Hospital Air Using High-Tech Bubbles
- World's First Inhalable COVID-19 Vaccine Approved in China
- COVID-19 Vaccine Patch Fights SARS-CoV-2 Variants Better than Needles
- Blood Viscosity Testing Can Predict Risk of Death in Hospitalized COVID-19 Patients
- ‘Covid Computer’ Uses AI to Detect COVID-19 from Chest CT Scans
- MRI Lung-Imaging Technique Shows Cause of Long-COVID Symptoms
- Chest CT Scans of COVID-19 Patients Could Help Distinguish Between SARS-CoV-2 Variants
- Specialized MRI Detects Lung Abnormalities in Non-Hospitalized Long COVID Patients
- AI Algorithm Identifies Hospitalized Patients at Highest Risk of Dying From COVID-19
- Sweat Sensor Detects Key Biomarkers That Provide Early Warning of COVID-19 and Flu
- Study Assesses Impact of COVID-19 on Ventilation/Perfusion Scintigraphy
- CT Imaging Study Finds Vaccination Reduces Risk of COVID-19 Associated Pulmonary Embolism
- Third Day in Hospital a ‘Tipping Point’ in Severity of COVID-19 Pneumonia
- Longer Interval Between COVID-19 Vaccines Generates Up to Nine Times as Many Antibodies
- AI Model for Monitoring COVID-19 Predicts Mortality Within First 30 Days of Admission
- AI Predicts COVID Prognosis at Near-Expert Level Based Off CT Scans
Channels
Artificial Intelligence
view channel
FDA-Cleared AI System Detects Sepsis Earlier and Reduces Mortality
Sepsis remains one of the deadliest complications for hospitalized patients, in part because its early signs overlap with other conditions. Each hour of delayed recognition measurably decreases survival,... Read moreFacial Image Analysis Tracks Biological Aging, Predicts Cancer Outcomes
Biological aging is the progressive loss of physiological function that may diverge from chronological age. In cancer care, clinicians need simple tools that reflect dynamic changes in patient resilience... Read moreCritical Care
view channel
High-Frequency Ultrasound Disables Viruses While Sparing Human Cells
Respiratory viral infections such as influenza A and COVID-19 continue to challenge patient management. Antiviral drugs are generally difficult to develop, and safe, broadly applicable options remain limited.... Read more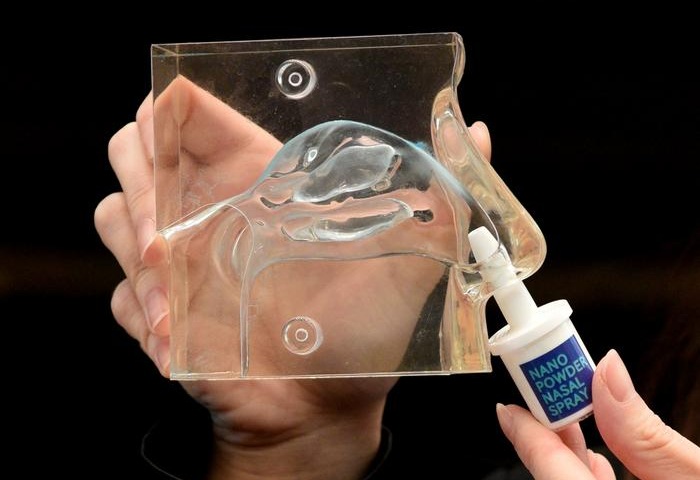
New Nasal Spray Enables Prehospital Neuroprotection in Ischemic Stroke
Ischemic stroke, caused by arterial occlusion in the brain, requires time‑critical intervention to limit neuronal loss. Many patients arrive outside the narrow window for thrombolysis or thrombectomy,... Read more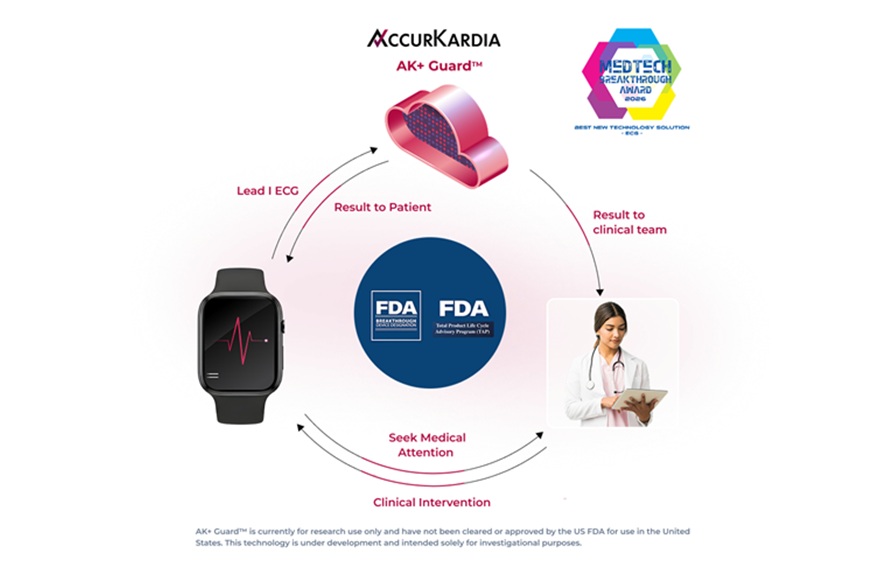
Single-Lead AI ECG Tool Detects Moderate-to-Severe Hyperkalemia Outside Clinic
Hyperkalemia can escalate quickly and is commonly monitored through laboratory blood draws, leaving risk between clinic visits. Clinicians need timely, accessible tools to identify moderate to severe elevations... Read moreSurgical Techniques
view channel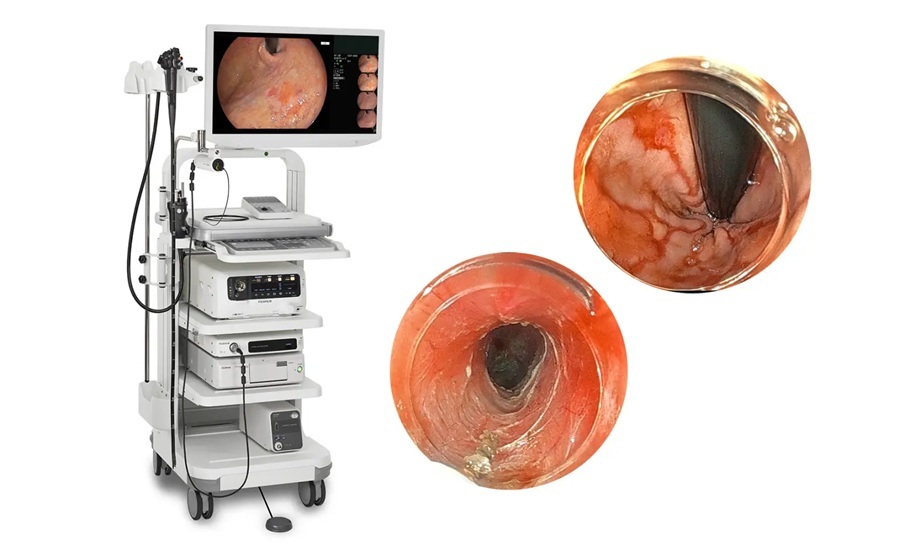
Advanced Endoscopy Platform Targets Challenging Upper GI Procedures
Fujifilm’s ELUXEO 8000 Endoscopic Imaging System, used with the EG-840TP ultra‑slim therapeutic gastroscope, was named “Best New Endoscopy Technology Solution” in the 10th annual MedTech Breakthrough Awards program.... Read more
Photoacoustic Imaging System Maps Hidden Nerves and Vessels in Robotic Laparoscopy
Hidden neurovascular structures can be difficult to visualize during robot-assisted laparoscopy, increasing the risk of iatrogenic injury. Such injuries can cause hemorrhage, neurological deficits, and... Read morePatient Care
view channel
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read more
Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
More than 80% of in-hospital patients receive intravenous (IV) therapy. Every dose of IV medicine delivered in a small volume (<250 mL) infusion bag should be followed by subsequent flushing to ensure... Read moreHealth IT
view channel
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read more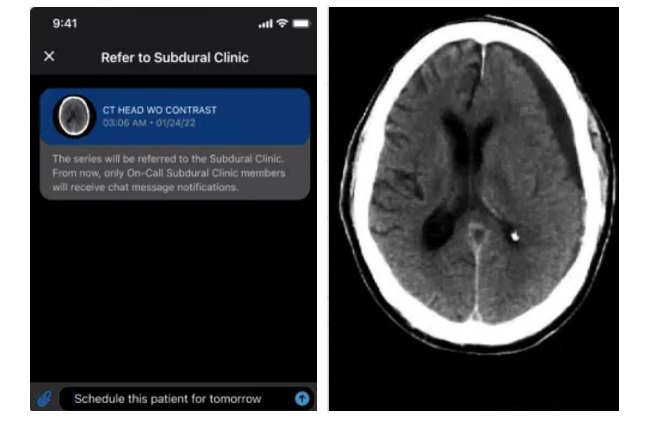
AI System Detects and Quantifies Chronic Subdural Hematoma
Viz.ai (San Francisco, CA, USA) announced a strategic commercialization collaboration with Johnson & Johnson (New Brunswick, NJ, USA) to expand access in the United States to the Viz Subdural solution... Read more
Continuous Monitoring Platform Detects Infection Risk Across Care Transitions
Patients leaving skilled nursing facilities often lose continuous physiologic monitoring, increasing the risk of undetected infection and delayed intervention. Nursing home residents are seven times more... Read more
Automated System Classifies and Tracks Cardiogenic Shock Across Hospital Settings
Cardiogenic shock remains a difficult, time-sensitive emergency, with delayed identification driving poor outcomes and persistently high mortality. Many cases go undocumented even at advanced stages, hindering... Read morePoint of Care
view channel
Point-of-Care Viscoelastic Testing System Supports Obstetric Bleeding Management
HemoSonics (Durham, NC, USA) announced on May 5, 2026 that the company's Quantra Hemostasis System for Obstetric Procedures won Silver in the 2026 Edison Awards in the Women’s Health and Reproductive Innovations... Read moreBusiness
view channel
Olympus Partnership Aims to Expand Access to Robot-Assisted Endoscopic Therapy
Olympus has signed an exclusive global distribution agreement with EndoRobotics Co., Ltd., under which robot-assisted technologies developed by EndoRobotics will be distributed worldwide as part of the... Read more