BioMérieux BIOFIRE Respiratory 2.1 (RP2.1) Panel with SARS-CoV-2 Becomes First COVID-19 Diagnostic Test to Obtain De Novo FDA Authorization
|
By HospiMedica International staff writers Posted on 19 Mar 2021 |

Image: BIOFIRE RP2.1 Panel (Photo courtesy of BioMérieux)
BioMérieux (Marcy-l'Étoile, France) has received US Food and Drug Administration (FDA) De Novo authorization for the BIOFIRE RP2.1 Panel, making it the first SARS-CoV-2 diagnostic test of any kind to be granted De Novo status by US FDA.
The panel allows for the detection of 22 viral and bacterial pathogens responsible for respiratory infections, including SARS-CoV-2 (the cause of COVID-19 disease). The panel was granted De Novo status by US FDA after having gone through the normal US FDA review pathway outside of the Emergency Use Authorization (EUA) track. The De Novo authorization will be concurrent with the revocation of the US FDA EUA that was obtained on May 1, 2020 for the panel. The BIOFIRE RP2.1 Panel EUA and De Novo kits are identical with the exception of changes to the labeling.
The BIOFIRE RP2.1 Panel allows healthcare providers to quickly identify common respiratory pathogens found in patients presenting with acute respiratory tract infection, using one simple test. The BIOFIRE RP2.1 Panel yields results in approximately 45 minutes using nasopharyngeal swab (NPS) samples in transport media or saline. It runs on the fully automated BIOFIRE FILMARRAY 2.0 and BIOFIRE Torch Systems with only two minutes of sample preparation time.
The De Novo application was supported by a multicenter prospective clinical study in which the performance of the BIOFIRE RP2.1 Panel SARS-CoV-2 assay was evaluated in over 500 specimens against a combined reference of three independent molecular SARS-CoV-2 assays, each with U.S. FDA EUA designation. The BIOFIRE RP2.1 Panel SARS-CoV-2 assay demonstrated positive percent agreement (PPA) of 98.4% and negative percent agreement (NPA) of 98.9%.
“The De Novo authorization of the BIOFIRE RP2.1 Panel demonstrates how BioFire is dedicated to responding to a rapidly-evolving global pandemic with urgency and accuracy. This is the first US FDA De Novo authorized COVID-19 test,” said Pierre Boulud, Chief Operating Officer, Clinical Operations of bioMérieux.
Related Links:
BioMérieux
The panel allows for the detection of 22 viral and bacterial pathogens responsible for respiratory infections, including SARS-CoV-2 (the cause of COVID-19 disease). The panel was granted De Novo status by US FDA after having gone through the normal US FDA review pathway outside of the Emergency Use Authorization (EUA) track. The De Novo authorization will be concurrent with the revocation of the US FDA EUA that was obtained on May 1, 2020 for the panel. The BIOFIRE RP2.1 Panel EUA and De Novo kits are identical with the exception of changes to the labeling.
The BIOFIRE RP2.1 Panel allows healthcare providers to quickly identify common respiratory pathogens found in patients presenting with acute respiratory tract infection, using one simple test. The BIOFIRE RP2.1 Panel yields results in approximately 45 minutes using nasopharyngeal swab (NPS) samples in transport media or saline. It runs on the fully automated BIOFIRE FILMARRAY 2.0 and BIOFIRE Torch Systems with only two minutes of sample preparation time.
The De Novo application was supported by a multicenter prospective clinical study in which the performance of the BIOFIRE RP2.1 Panel SARS-CoV-2 assay was evaluated in over 500 specimens against a combined reference of three independent molecular SARS-CoV-2 assays, each with U.S. FDA EUA designation. The BIOFIRE RP2.1 Panel SARS-CoV-2 assay demonstrated positive percent agreement (PPA) of 98.4% and negative percent agreement (NPA) of 98.9%.
“The De Novo authorization of the BIOFIRE RP2.1 Panel demonstrates how BioFire is dedicated to responding to a rapidly-evolving global pandemic with urgency and accuracy. This is the first US FDA De Novo authorized COVID-19 test,” said Pierre Boulud, Chief Operating Officer, Clinical Operations of bioMérieux.
Related Links:
BioMérieux
Latest COVID-19 News
- Low-Cost System Detects SARS-CoV-2 Virus in Hospital Air Using High-Tech Bubbles
- World's First Inhalable COVID-19 Vaccine Approved in China
- COVID-19 Vaccine Patch Fights SARS-CoV-2 Variants Better than Needles
- Blood Viscosity Testing Can Predict Risk of Death in Hospitalized COVID-19 Patients
- ‘Covid Computer’ Uses AI to Detect COVID-19 from Chest CT Scans
- MRI Lung-Imaging Technique Shows Cause of Long-COVID Symptoms
- Chest CT Scans of COVID-19 Patients Could Help Distinguish Between SARS-CoV-2 Variants
- Specialized MRI Detects Lung Abnormalities in Non-Hospitalized Long COVID Patients
- AI Algorithm Identifies Hospitalized Patients at Highest Risk of Dying From COVID-19
- Sweat Sensor Detects Key Biomarkers That Provide Early Warning of COVID-19 and Flu
- Study Assesses Impact of COVID-19 on Ventilation/Perfusion Scintigraphy
- CT Imaging Study Finds Vaccination Reduces Risk of COVID-19 Associated Pulmonary Embolism
- Third Day in Hospital a ‘Tipping Point’ in Severity of COVID-19 Pneumonia
- Longer Interval Between COVID-19 Vaccines Generates Up to Nine Times as Many Antibodies
- AI Model for Monitoring COVID-19 Predicts Mortality Within First 30 Days of Admission
- AI Predicts COVID Prognosis at Near-Expert Level Based Off CT Scans
Channels
Artificial Intelligence
view channel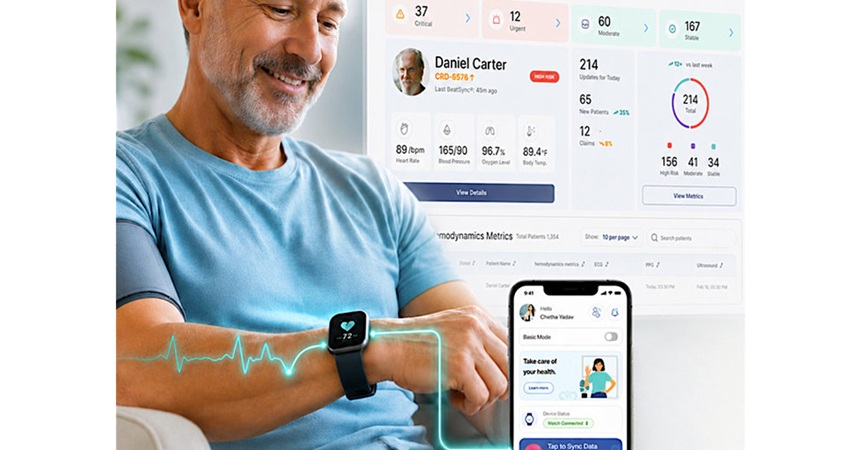
AI Platform Supports Noninvasive Remote Hemodynamic Monitoring in Heart Failure
Heart failure remains a leading cause of hospitalization in adults over 65, affecting more than 6.7 million people in the U.S. Clinicians often lose visibility into hemodynamic deterioration once patients... Read more
AI Tool Predicts Unplanned Care and Symptom Burden in Cancer Survivors
Unplanned emergency visits and hospitalizations remain common in cancer survivorship, when routine clinical contact often tapers while new symptoms emerge. These events reflect unmet needs and disrupt... Read moreCritical Care
view channel
Bedside CSF Monitor Detects Early Infection in Fluid Drains
External drainage of cerebrospinal fluid (CSF) after traumatic brain injury, hydrocephalus, or hemorrhage carries a significant risk of infection. These infections can prolong intensive care, cause severe... Read more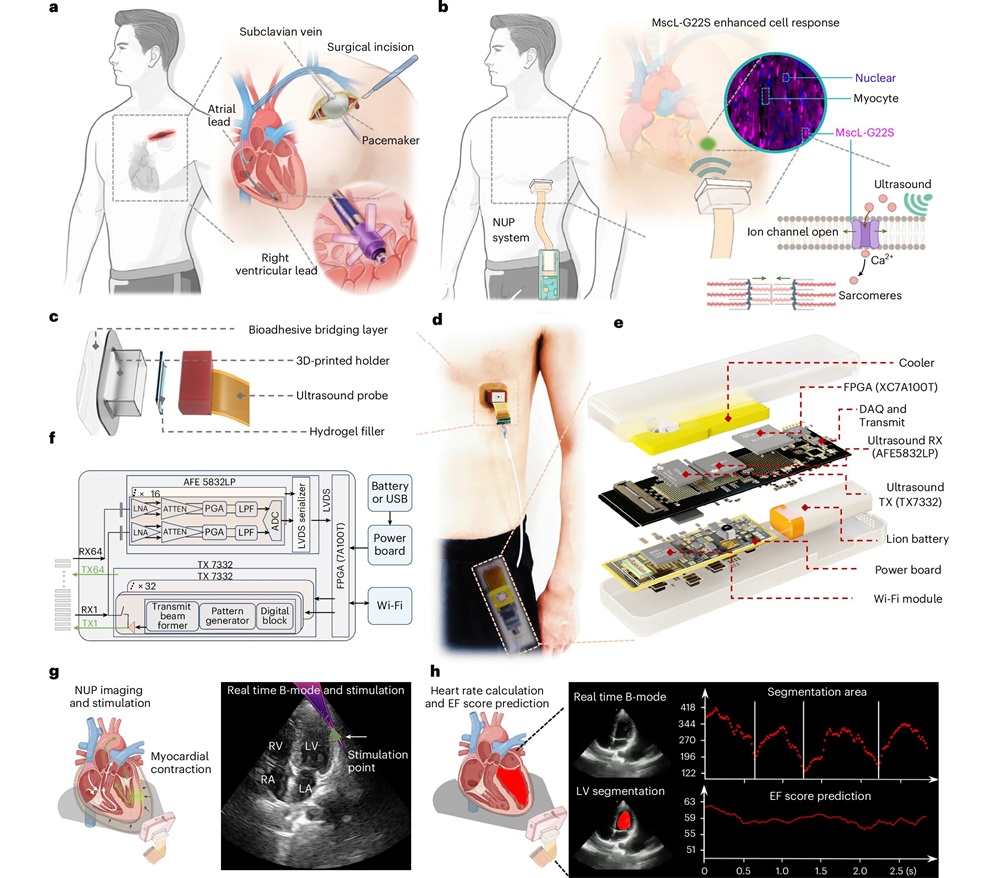
Wearable Ultrasound Patch Noninvasively Paces Heart to Stabilize Arrhythmias
Cardiac arrhythmias, including slow and irregular heart rhythms, often require pacemakers that are surgically implanted. While effective, implants carry procedural risks and long-term device maintenance burdens.... Read more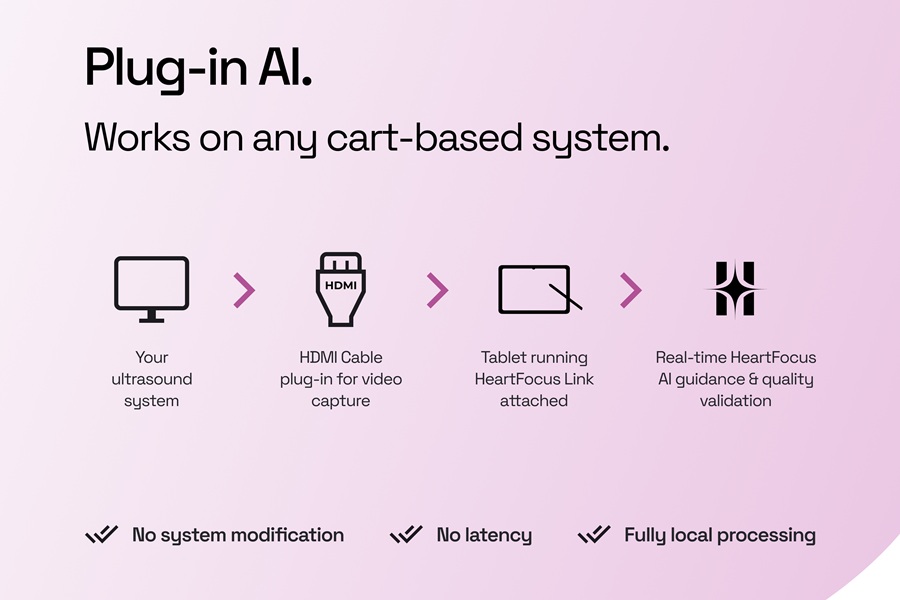
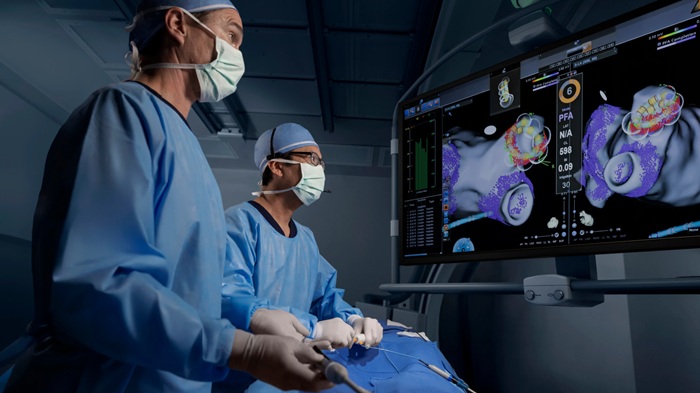
AI-Guided System Supports Cardiac Ultrasound Training on Cart-Based Systems
Cardiac ultrasound adoption in frontline settings often lags despite widespread investment in ultrasound equipment. Many systems sit underused for cardiac imaging because acquiring standard views requires... Read more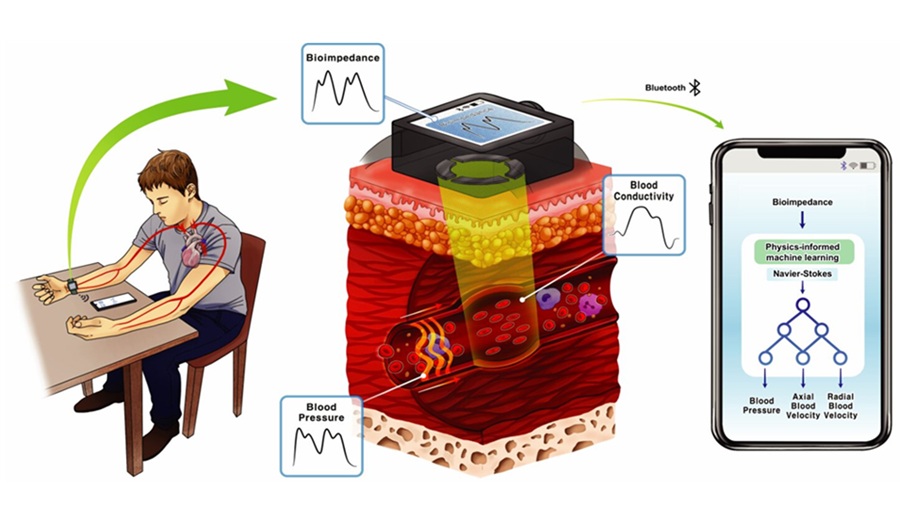
Cuffless Wearable Enables Continuous Blood Pressure Monitoring for Hypertension Care
Hypertension, or chronically elevated blood pressure, drives major risks for heart attack and stroke yet is typically assessed with intermittent cuff readings. These snapshots miss rapid physiologic changes... Read moreSurgical Techniques
view channel
3D Map of Heart Electrical Wiring Aims to Guide Congenital Heart Repair
Tetralogy of Fallot is one of the most common congenital heart problems and often requires surgery in infancy. Many survivors later develop conduction abnormalities because the cardiac electrical system... Read more
Small Cryoprobe Outperforms Forceps in Lung Biopsy Trial
Accurate diagnosis of pulmonary nodules, post-transplant allograft dysfunction, and diffuse parenchymal lung disease depends on high-quality tissue obtained during bronchoscopy. Conventional forceps biopsy... Read morePatient Care
view channel
AI Avatar Doctor Improves Patient Understanding Before Radiotherapy
Radiation oncology consultations require patients to grasp complex concepts quickly, yet anxiety and information overload often undermine understanding and informed consent. Poor comprehension can also... Read more
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read moreHealth IT
view channel
AI-Native EHR Achieves EU Medical Device Certification
InterSystems (Boston, MA, USA) announced that its IntelliCare electronic health record (EHR) solutions have been certified as Class IIa medical devices under the European Union Medical Device Regulation... Read more
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read morePoint of Care
view channel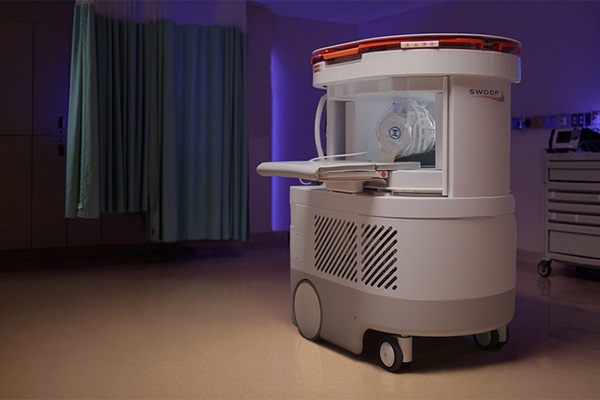
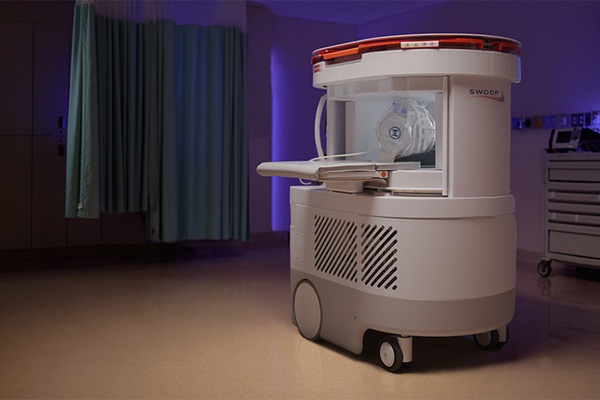
Portable MRI System Accelerates Emergency Brain Imaging and Triage
Emergency departments frequently face delays accessing conventional magnetic resonance imaging (MRI) for patients with suspected neurological emergencies. Such waits can slow triage, prolong boarding,... Read more