3D-Printed Spinal Implant Promotes Biological Fixation
|
By HospiMedica International staff writers Posted on 30 Oct 2017 |

Image: The Tritanium C anterior cervical cage implant (Photo courtesy of Stryker).
A new anterior cervical cage fabricated via additive manufacturing provides a favorable environment for cell attachment and proliferation.
The Stryker Corporation (Kalamazoo, MI, USA) Tritanium C Anterior Cervical Cage is indicated for use in cervical interbody fusion procedures in skeletally mature patients with degenerative disc disease. The C Anterior Cervical Cage is manufactured using Tritanium, a highly porous titanium material that consists of a random interconnected architecture with rugged, irregular pore sizes and shapes that are designed to mimic cancellous bone. Tritanium encourages bone in-growth and biological fixation, and can also wick and retain fluid.
The C Anterior Cervical Cage implant is fabricated via Stryker AMagine, an approach to implant creation that is based on additive three-dimensional (3D) printing. Features include an open central graft window and lateral windows to help reduce stiffness of the cage and minimize subsidence; in addition, the large graft window allows for bone graft containment. Serrations on the superior and inferior surfaces allow bidirectional fixation and maximize surface area for endplate contact with the cage, while smooth posterior edges facilitate insertion and protect soft tissue and anatomy.
“Stryker’s Tritanium Technology is engineered for bone,” said Bradley Paddock, president of the Stryker Spine division. “Our growing line of Tritanium implants reflects our commitment to being at the forefront of technology advances in spinal surgery, and to providing our surgeon customers with a range of options to address their preferences and meet the needs of their patients.”
An interbody cage device is a prosthesis used in spinal fusion procedures to maintain foraminal height and decompression. Once in place, the cage resists flexion and extension of the spine, as well as axial forces across the ventral and middle columns. The Tritanium C Anterior Cervical Cage is offered in a number of footprints, heights, and lordotic angles to adapt to a variety of patient anatomies.
The Stryker Corporation (Kalamazoo, MI, USA) Tritanium C Anterior Cervical Cage is indicated for use in cervical interbody fusion procedures in skeletally mature patients with degenerative disc disease. The C Anterior Cervical Cage is manufactured using Tritanium, a highly porous titanium material that consists of a random interconnected architecture with rugged, irregular pore sizes and shapes that are designed to mimic cancellous bone. Tritanium encourages bone in-growth and biological fixation, and can also wick and retain fluid.
The C Anterior Cervical Cage implant is fabricated via Stryker AMagine, an approach to implant creation that is based on additive three-dimensional (3D) printing. Features include an open central graft window and lateral windows to help reduce stiffness of the cage and minimize subsidence; in addition, the large graft window allows for bone graft containment. Serrations on the superior and inferior surfaces allow bidirectional fixation and maximize surface area for endplate contact with the cage, while smooth posterior edges facilitate insertion and protect soft tissue and anatomy.
“Stryker’s Tritanium Technology is engineered for bone,” said Bradley Paddock, president of the Stryker Spine division. “Our growing line of Tritanium implants reflects our commitment to being at the forefront of technology advances in spinal surgery, and to providing our surgeon customers with a range of options to address their preferences and meet the needs of their patients.”
An interbody cage device is a prosthesis used in spinal fusion procedures to maintain foraminal height and decompression. Once in place, the cage resists flexion and extension of the spine, as well as axial forces across the ventral and middle columns. The Tritanium C Anterior Cervical Cage is offered in a number of footprints, heights, and lordotic angles to adapt to a variety of patient anatomies.
Latest Surgical Techniques News
- Intravascular Imaging Guidance System Optimizes Stenting Procedures for Improved Patient Outcomes
- Better-Designed Operating Room Shortens Surgical Procedure Time and Produces Better Outcomes
- Spatial Computing Technology Could Revolutionize Operating Room Environment
- Innovative Catheter Guidance Technology Aims for Zero Malpositioning
- Breakthrough Heart Valve Combines Best of Mechanical and Tissue Replacement Technology
- Ultraportable Battery Powered Medical Device Revolutionizes Concept of Portable Surgical Care
- Groundbreaking Tubular Scaffolds Enhance Bone Regeneration of Critical-Sized Skull Defects
- AI-Powered Precision Surgery Platform to Make Procedures Smarter and Safer
- Novel Magnet Compression Anastomosis Technology Ensures Fewer Complications, Bleeds or Leaks
- Cloud-Based AI Endoscopy System Assists Gastroenterologists in Detecting Suspected Colorectal Polyps
- Sperm-Like “Micro-Robots” Navigate Through Complex Environments for Minimally Invasive Surgery

- Stent Implantation Found Preferable for Previously Untreated Non-Complex Coronary Artery Disease
- Neuroendoscopy System Improves Outcome of Minimally Invasive Neurosurgical Procedures
- AI Platform Leverages Robotic Bronchoscopy Under 3D Fluoroscopic Guidance for Precision Biopsy
- New Implant Powers Healing After Spinal Cord Injury
- New Method Combines EEG, MRI, and ML to Identify Seizure-Prone Brain Regions Before Surgery
Channels
Critical Care
view channel
Specialized Stent that Expands as Child Grows to Result in Fewer Heart Surgeries for Babies
Approximately 40,000 babies are born each year in the United States with a heart defect, according to the U.S. Centers for Disease Control and Prevention. Many of these infants have narrowing in either... Read moreNext-Gen Wearable Continuous Glucose Monitoring System to Revolutionize Diabetes Management
Continuous glucose monitoring systems (CGMs) play a vital role in the closed-loop management of diabetes. With advances in the field, the demand for next-generation CGMs that offer improved noise resistance,... Read morePatient Care
view channelFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel
New 8-Minute Blood Test to Diagnose or Rule Out Heart Attack Shortens ED Stay
Emergency department overcrowding is a significant global issue that leads to increased mortality and morbidity, with chest pain being one of the most common reasons for hospital admissions.... Read more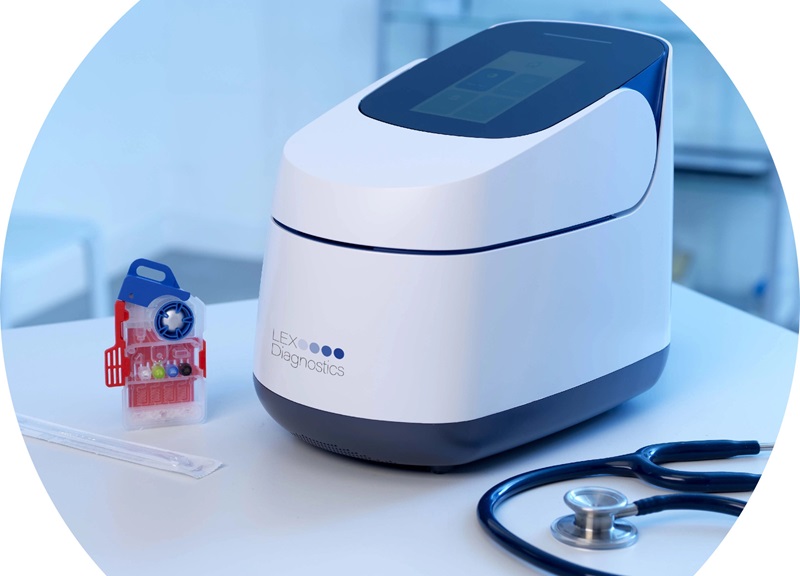
5-Minute Multiplex PCR Testing System to Redefine Point-Of-Care Diagnostics
Point of care (POC) testing provides significant clinical benefits, particularly when results can be obtained quickly enough to inform clinical decisions during a single consultation, eliminating the need... Read moreBusiness
view channel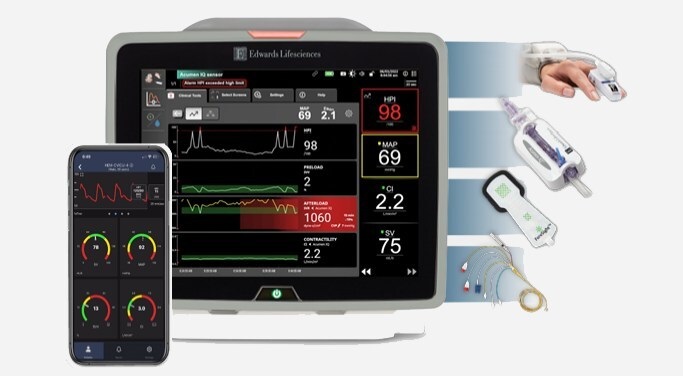
BD Completes Acquisition of Critical Care from Edwards Lifesciences
BD (Becton, Dickinson and Company, Franklin Lakes, J, USA) has completed the acquisition of Edwards Lifesciences' (Irvine, CA, USA) Critical Care product group, which will be renamed as BD Advanced... Read more
ZOLL to Acquire Vyaire Medical’s Ventilator Business
ZOLL (Chelmsford, MA, USA), an Asahi Kasei (Tokyo, Japan) company that manufactures medical devices and related software solutions, has become the winning bidder in an auction to acquire Vyaire Medical’s... Read more