Hospital Antibiotic Policies Improve Prescription Practices
|
By HospiMedica International staff writers Posted on 21 Feb 2017 |
An updated review of studies identifies effective guidelines and policies to reduce unnecessary use of antibiotics in hospitals.
Researchers at the University of Dundee, University College London, and other institutions searched the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, and Embase for studies examining the effectiveness and safety of interventions designed to improve antibiotic prescribing to hospital inpatients, and to investigate the effect of different intervention functions. In all, the researchers found 221 studies from the United States, Europe, Asia, South America, and Australia.
The researchers found that interventions broadly fell into two categories; 'restrictive' techniques applied rules to make physicians prescribe properly, whilst 'enabling' techniques provided advice or feedback to help physicians make more informed prescribing decisions. In both cases, the aim of the intervention was to increase the number of appropriate prescribing decisions so that patients who were unlikely to benefit from antibiotics did not get them, whilst they were still used for patients who stood to benefit from them.
The results showed that interventions that included enabling or restrictive techniques were consistently more effective than interventions that relied on simple education alone. Moreover, a synergistic effect existed, with enabling techniques increasing the effectiveness of restrictive techniques. In addition, the interventions shortened duration of antibiotic use from 11 to 9 days per patient, and reduced hospital stay from an average of 13 days to 12 per patient. The review was published on February 9, 2017, in The Cochrane Library.
“We do not need more studies to answer the question of whether these interventions reduce unnecessary antibiotic use, but we do need more research to understand why the most effective behavior change techniques are not more widely adopted within hospital settings,” concluded lead author Peter Davey, PhD, of the UD department of population health sciences. “Appropriate antibiotic use in hospitals should ensure effective treatment of patients with infection, and reduce unnecessary prescriptions.”
For the study, the researchers defined restriction as using rules to reduce the opportunity to engage in the target behavior, or increase the target behavior by reducing the opportunity to engage in competing behaviors. Enablement was defined as increasing the means and reducing the barriers in order to increase capability or opportunity.
Researchers at the University of Dundee, University College London, and other institutions searched the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, and Embase for studies examining the effectiveness and safety of interventions designed to improve antibiotic prescribing to hospital inpatients, and to investigate the effect of different intervention functions. In all, the researchers found 221 studies from the United States, Europe, Asia, South America, and Australia.
The researchers found that interventions broadly fell into two categories; 'restrictive' techniques applied rules to make physicians prescribe properly, whilst 'enabling' techniques provided advice or feedback to help physicians make more informed prescribing decisions. In both cases, the aim of the intervention was to increase the number of appropriate prescribing decisions so that patients who were unlikely to benefit from antibiotics did not get them, whilst they were still used for patients who stood to benefit from them.
The results showed that interventions that included enabling or restrictive techniques were consistently more effective than interventions that relied on simple education alone. Moreover, a synergistic effect existed, with enabling techniques increasing the effectiveness of restrictive techniques. In addition, the interventions shortened duration of antibiotic use from 11 to 9 days per patient, and reduced hospital stay from an average of 13 days to 12 per patient. The review was published on February 9, 2017, in The Cochrane Library.
“We do not need more studies to answer the question of whether these interventions reduce unnecessary antibiotic use, but we do need more research to understand why the most effective behavior change techniques are not more widely adopted within hospital settings,” concluded lead author Peter Davey, PhD, of the UD department of population health sciences. “Appropriate antibiotic use in hospitals should ensure effective treatment of patients with infection, and reduce unnecessary prescriptions.”
For the study, the researchers defined restriction as using rules to reduce the opportunity to engage in the target behavior, or increase the target behavior by reducing the opportunity to engage in competing behaviors. Enablement was defined as increasing the means and reducing the barriers in order to increase capability or opportunity.
Latest Hospital News News
- Nurse Tracking System Improves Hospital Workflow
- New Children’s Hospital Transforms California Healthcare
- Noisy Hospitals Face Threat of Decreased Federal Compensation
- Orthopedics Centre of Excellence Planned for Guy’s Hospital
- Research Suggests Avoidance of Low-Value Surgical Procedures
- U.S. Federal Readmission Fines Linked to Higher Mortality
- Columbia China to Build New Hospital in Jiaxing
- Dubai Debuts Second Robotic Pharmacy Service
- Seattle Hospital Network Shifts Away from Overlapping Surgeries
- ACC to Launch Valvular Heart Disease Program in China
- Mortality Rates Lower at Major Teaching Hospitals
- South Australia to Inaugurate Upscale Hospital
- Raffles to Launch Second Hospital Project in China
- Research Center Tackles Antimicrobial Drugs Challenge
- Miami Cardiac & Vascular Institute Completes Expansion Project
- USC Virtual Care Clinic to Employ Avatar Doctors
Channels
Artificial Intelligence
view channel
AI Platform Supports Noninvasive Remote Hemodynamic Monitoring in Heart Failure
Heart failure remains a leading cause of hospitalization in adults over 65, affecting more than 6.7 million people in the U.S. Clinicians often lose visibility into hemodynamic deterioration once patients... Read more
AI Tool Predicts Unplanned Care and Symptom Burden in Cancer Survivors
Unplanned emergency visits and hospitalizations remain common in cancer survivorship, when routine clinical contact often tapers while new symptoms emerge. These events reflect unmet needs and disrupt... Read moreCritical Care
view channel
Smartphone Heart Rhythm App Reduces Unnecessary Cardioversion Procedures
Atrial fibrillation, an irregular and often rapid heart rhythm, is the most common arrhythmia in adults. Elective electrical cardioversion is frequently canceled on the day of treatment when patients revert... Read more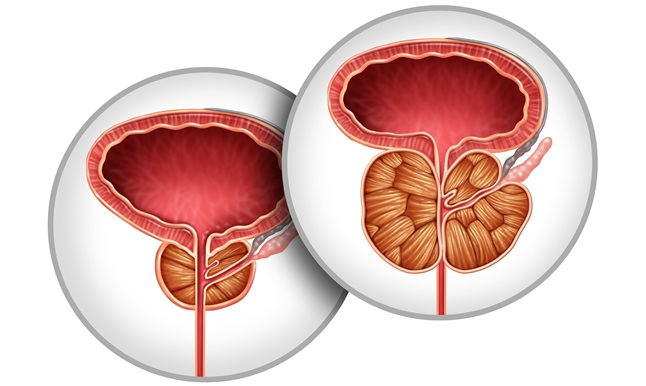
New Practice Guidance Supports Prostatic Artery Embolization for BPH Symptoms
Benign prostatic hyperplasia is a noncancerous enlargement of the prostate that can cause lower urinary tract symptoms and impair daily functioning. These symptoms erode sleep, productivity, and sexual... Read more
Bedside CSF Monitor Detects Early Infection in Fluid Drains
External drainage of cerebrospinal fluid (CSF) after traumatic brain injury, hydrocephalus, or hemorrhage carries a significant risk of infection. These infections can prolong intensive care, cause severe... Read more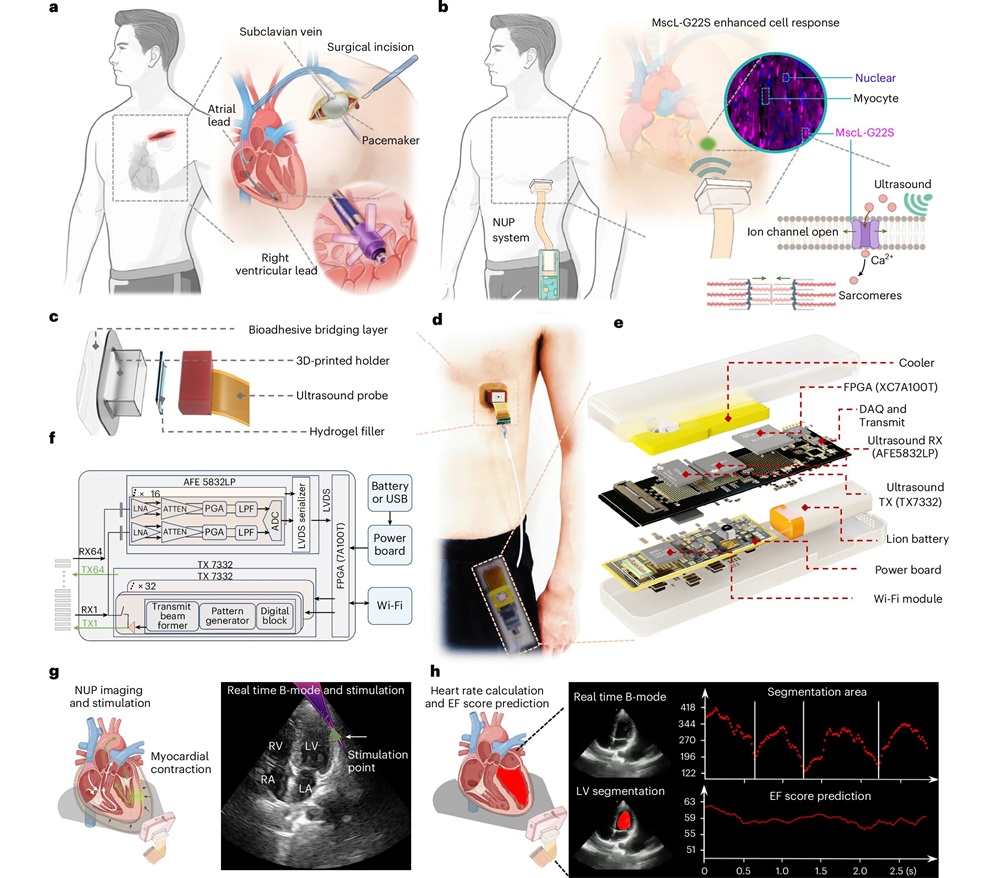
Wearable Ultrasound Patch Noninvasively Paces Heart to Stabilize Arrhythmias
Cardiac arrhythmias, including slow and irregular heart rhythms, often require pacemakers that are surgically implanted. While effective, implants carry procedural risks and long-term device maintenance burdens.... Read moreSurgical Techniques
view channel
CE-Marked Ultrasonic Shears Streamline Breast and Thyroid Surgery
Thyroid and breast surgeries are often performed in confined anatomical spaces near critical structures, making precise dissection and controlled thermal management essential. As the global disease burden... Read more
3D Map of Heart Electrical Wiring Aims to Guide Congenital Heart Repair
Tetralogy of Fallot is one of the most common congenital heart problems and often requires surgery in infancy. Many survivors later develop conduction abnormalities because the cardiac electrical system... Read morePatient Care
view channel
AI Avatar Doctor Improves Patient Understanding Before Radiotherapy
Radiation oncology consultations require patients to grasp complex concepts quickly, yet anxiety and information overload often undermine understanding and informed consent. Poor comprehension can also... Read more
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read moreHealth IT
view channel
AI-Native EHR Achieves EU Medical Device Certification
InterSystems (Boston, MA, USA) announced that its IntelliCare electronic health record (EHR) solutions have been certified as Class IIa medical devices under the European Union Medical Device Regulation... Read more
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read morePoint of Care
view channel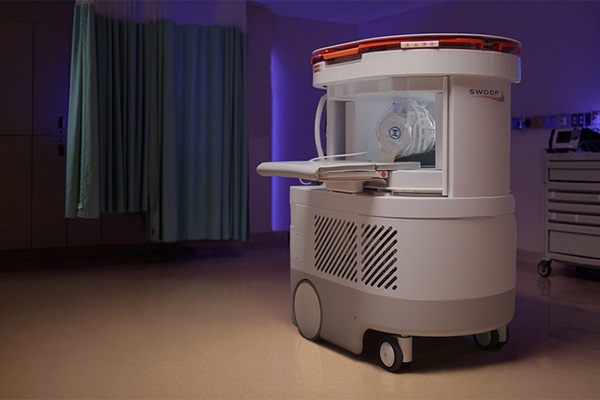
Portable MRI System Accelerates Emergency Brain Imaging and Triage
Emergency departments frequently face delays accessing conventional magnetic resonance imaging (MRI) for patients with suspected neurological emergencies. Such waits can slow triage, prolong boarding,... Read more