AI Technology to Assess Breathing Tube Placements Through Chest X-Rays Receives FDA Clearance
|
By HospiMedica International staff writers Posted on 21 Jan 2022 |
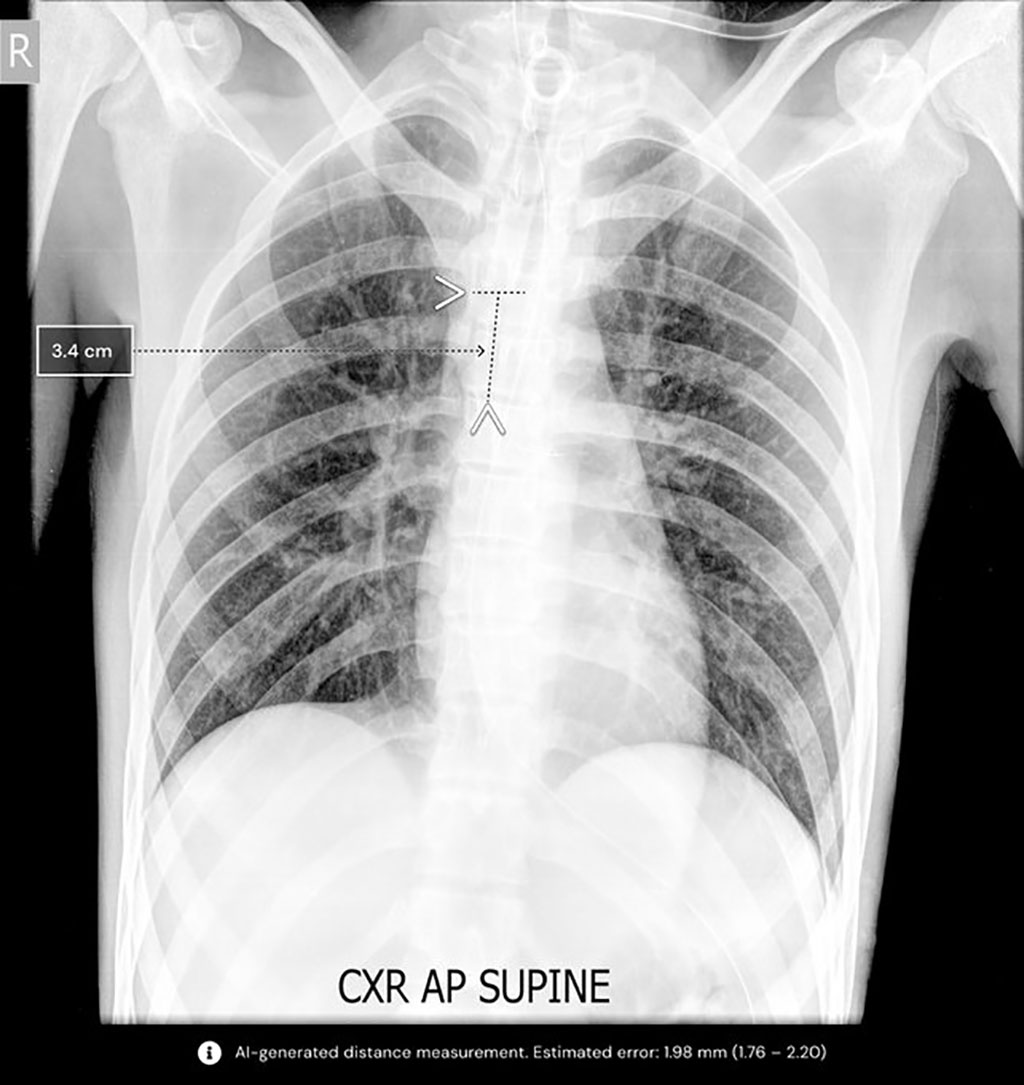
Qure.ai Technologies (Mumbai, India) has gained 510(k) clearance from the Food and Drug Administration (FDA) for an artificial intelligence (AI) algorithm called qXR-BT that will help doctors in assessing Breathing Tube (BT) placements.
Through chest X-rays, the algorithm assists clinicians for intubated patients in locating the BT placement and automating measurements. This is the first solution of its kind to automate the manual measurement process for both endotracheal and tracheostomy tubes. Verification of endotracheal tube (ETT) placement is imperative for the oxygenation, ventilation, and airway protection of patients. While a common procedure done in hospitals daily, rates for incorrect ET tube placement have been noted to be up to 25%. Mistakes during the intubation process pose a threat to the lives of hospital intensive care unit patients. Even if an expert clinical team is present for inserting and securing the tube after the initial placement, ETT migration is an inevitable consequence of coughing, suctioning, transport, and patient movement.
Qure's qXT-BT algorithm analyzes the tube position, automates measurement, and gives the physician a report on the tube’s positional accuracy in less than a minute. This enables clinicians to rapidly identify if the tube is properly positioned or whether extra attention is required. The algorithm is vendor-agnostic and is designed to work on both portable and stationary X-ray machines. qXR-BT is expected to become a standard feature of any critical care framework, giving residents and junior clinicians more confidence in reliably measuring breathing tube placement in intubated patients.
“Daily monitoring of Tubes is critical for all intubated inpatients, and sometimes an arduous task on the portable exam with either the carina obscured or the tip not visible. An accurate AI solution could be a valuable aid for reporting on these chest X-rays- especially with the measurement,” said Dr. Mannudeep Kalra, attending thoracic radiologist, Massachusetts General Hospital and Professor of Radiology, Harvard Medical School, who was involved in a research collaboration evaluating the technology.
“We are pleased to have received FDA clearance for qXR-BT. In the last two years, we have seen the need to decrease processing times and solve workflow delays,” said Prashant Warier, CEO and Co-Founder, Qure.ai. “Especially in the wake of the COVID-19 pandemic and the need for mechanical ventilation in affected patients, the need for prompt assistance to an overburdened healthcare workforce is paramount.”
Related Links:
Qure.ai Technologies
Latest Critical Care News
- Breakthrough Computational Method Predicts Sudden Cardiac Death
- Ingestible Microbiome Sampling Pill to Help Diagnose Wide Range of Health Conditions
- GPS-Like Smart Pills with AI Provide Real-Time 3D Monitoring Of Gastrointestinal Health
- Bioengineering Breakthrough to Improve Bone Regeneration Treatments
- Soft Robots with Electronic Skins and Artificial Muscles to Provide Medical Treatment
- AI Camera Technology Helps Doctors Quickly Assess Severity of Infections
- Machine Learning Delivers Personalized Oxygenation for Patients on Ventilators
- New AI Algorithm Detects Rare Epileptic Seizures from EEG Data
- Facial Thermal Imaging Combined with AI Predicts Coronary Artery Disease
- AI Technology Predicts Cardiac Events 10 Years in Advance
- Recyclable ‘Smart Skin’ Monitors Biological Signals on Demand
- Novel Catheter to Reduce Risk for Associated Urinary Tract Infections and Sepsis
- AI Doubles Medical Professionals’ Accuracy in Reading EEG Charts of ICU Patients
- Flexible Device Enables Sweat Gland Stimulation and Simultaneous Biosensing
- WHO Publishes First Global Guidelines to Reduce Bloodstream Infections from Catheter Use
- Innovative Material Paves Way for Next-Generation Wearable Devices
Channels
Surgical Techniques
view channel
Total Robotic Metabolic and Bariatric Surgery Proves More Beneficial than Conventional Laparoscopy
According to the U.S. Centers for Disease Control and Prevention (CDC), 42.4% of Americans are affected by obesity. Research indicates that obesity can compromise the immune system, trigger chronic inflammation,... Read moreWirelessly Activated Robotic Device Aids Digestion in Patients with Compromised Organs
The transport of fluids and solids is essential in the human body, driven by a wave-like movement in the lumen known as peristalsis. However, peristalsis can be disrupted in patients who have obstructions... Read morePatient Care
view channelFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel
POCT for Infectious Diseases Delivers Laboratory Equivalent Pathology Results
On-site pathology tests for infectious diseases in rural and remote locations can achieve the same level of reliability and accuracy as those conducted in hospital laboratories, a recent study suggests.... Read more
Cartridge-Based Hemostasis Analyzer System Enables Faster Coagulation Testing
Quickly assessing a patient's total hemostasis status can be critical to influencing clinical outcomes and using blood products. Haemonetics Corporation (Boston, MA, USA) has now obtained 510(k) clearance... Read more
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read moreBusiness
view channel
BD Acquires Edwards Lifesciences' Critical Care Product Group for USD 4.2 Billion
BD (Becton, Dickinson and Company, Franklin Lakes, NJ, USA) and Edwards Lifesciences (Irvine, CA, USA) have entered into a definitive agreement under which BD will acquire Edwards' Critical Care product... Read more