Valve-in-Valve Replacement Obviates Second Heart Surgery
|
By HospiMedica International staff writers Posted on 22 Apr 2015 |

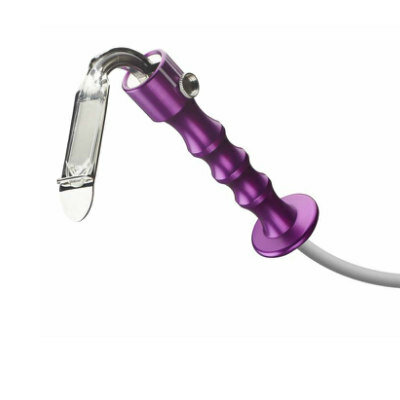
Image: The Medtronic CoreValve system (Photo courtesy of Medtronic).
An aortic “valve-in-valve” replacement system helps treat patients who have previously had a tissue aortic valve replacement and are in need of a second one.
The CoreValve artificial heart valve can now be used in patients in need of replacement of a failed tissue aortic valve due to severe native calcific aortic stenosis or failure of a surgical bioprosthetic aortic valve, and who are at high risk for complications associated with traditional open-heart surgery. CoreValve is made of tissue obtained from the heart of a pig; for support, it is attached to a flexible, self-expanding metal frame made of nickel-titanium alloy. To implant the device, it is delivered compressed to the implantation site via transcatheter aortic valve replacement (TAVR).
The procedure involves a catheter threaded through the blood vessels until it reaches the failed tissue aortic valve. It is then released from the end of the catheter, expanding on its own and anchoring itself to the old failed valve. Once the device is in place, it opens and closes properly, restoring the aortic valve function. The CoreValve system, a product of Medtronic (Minneapolis, MN, USA), was approved by the US Food and Drug Administration (FDA) as a replacement valve in patients who are infection-free; do not have a mechanical aortic heart valve; and who can tolerate blood thinners.
“The CoreValve System offers a less invasive treatment option for a significant number of patients with failed tissue aortic valves whose medical teams determine that the risks associated with repeat open-heart surgery are high or extremely high,” said William Maisel, MD, deputy center director for science and chief scientist in the FDA Center for Devices and Radiological Health (CDRH). “The approval is an important expansion of the authorized use of the transcatheter aortic valve replacement technology.”
TAVR aortic valve replacement, without the need for open heart surgery or cardiopulmonary bypass, is faster and less invasive than current open-heart procedures, and has so far been proven effective in high-risk and inoperable patients, and could soon become the standard of care, even in moderate and low surgical risk patients.
Related Links:
Medtronic
The CoreValve artificial heart valve can now be used in patients in need of replacement of a failed tissue aortic valve due to severe native calcific aortic stenosis or failure of a surgical bioprosthetic aortic valve, and who are at high risk for complications associated with traditional open-heart surgery. CoreValve is made of tissue obtained from the heart of a pig; for support, it is attached to a flexible, self-expanding metal frame made of nickel-titanium alloy. To implant the device, it is delivered compressed to the implantation site via transcatheter aortic valve replacement (TAVR).
The procedure involves a catheter threaded through the blood vessels until it reaches the failed tissue aortic valve. It is then released from the end of the catheter, expanding on its own and anchoring itself to the old failed valve. Once the device is in place, it opens and closes properly, restoring the aortic valve function. The CoreValve system, a product of Medtronic (Minneapolis, MN, USA), was approved by the US Food and Drug Administration (FDA) as a replacement valve in patients who are infection-free; do not have a mechanical aortic heart valve; and who can tolerate blood thinners.
“The CoreValve System offers a less invasive treatment option for a significant number of patients with failed tissue aortic valves whose medical teams determine that the risks associated with repeat open-heart surgery are high or extremely high,” said William Maisel, MD, deputy center director for science and chief scientist in the FDA Center for Devices and Radiological Health (CDRH). “The approval is an important expansion of the authorized use of the transcatheter aortic valve replacement technology.”
TAVR aortic valve replacement, without the need for open heart surgery or cardiopulmonary bypass, is faster and less invasive than current open-heart procedures, and has so far been proven effective in high-risk and inoperable patients, and could soon become the standard of care, even in moderate and low surgical risk patients.
Related Links:
Medtronic
Latest Surgical Techniques News
- AR Surgical Technology Translates Complex 2D Medical Imaging to Enhance Accuracy
- Miniaturized Snake-Like Probe Images Cerebral Arteries From Within
- Miniaturized Implantable Multi-Sensors Device to Monitor Vessels Health
- Tiny Robots Made Out Of Carbon Could Conduct Colonoscopy, Pelvic Exam or Blood Test
- Miniaturized Ultrasonic Scalpel Enables Faster and Safer Robotic-Assisted Surgery
- AI Assisted Reading Tool for Small Bowel Video Capsule Endoscopy Detects More Lesions
- First-Ever Contact Force Pulsed Field Ablation System to Transform Treatment of Ventricular Arrhythmias
- Caterpillar Robot with Built-In Steering System Crawls Easily Through Loops and Bends
- Tiny Wraparound Electronic Implants to Revolutionize Treatment of Spinal Cord Injuries
- Small, Implantable Cardiac Pump to Help Children Awaiting Heart Transplant
- Gastrointestinal Imaging Capsule a Game-Changer in Esophagus Surveillance and Treatment
- World’s Smallest Laser Probe for Brain Procedures Facilitates Ablation of Full Range of Targets
- Artificial Intelligence Broadens Diagnostic Abilities of Conventional Coronary Angiography
- AI-Powered Surgical Visualization Tool Supports Surgeons' Visual Recognition in Real Time
- Cutting-Edge Robotic Bronchial Endoscopic System Provides Prompt Intervention during Emergencies
- Handheld Device for Fluorescence-Guided Surgery a Game Changer for Removal of High-Grade Glioma Brain Tumors
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreCritical Care
view channel.jpeg)
Transcatheter Valve Replacement Outcomes Similar To Surgery, Finds Study
A new study has shown that a minimally invasive procedure for replacing the aortic valve in the heart—known as transcatheter aortic valve replacement (TAVR)—is on par with the more traditional surgical... Read more
Revascularization Improves Life Quality in Chronic Limb-Threatening Ischemia, Finds Study
Researchers have undertaken a detailed study to evaluate the effects of revascularization strategies on the health-related quality of life (HRQoL) of patients with chronic limb-threatening ischemia.... Read morePatient Care
view channelFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more
New Generation Glucose Hospital Meter System Ensures Accurate, Interference-Free and Safe Use
A new generation glucose hospital meter system now comes with several features that make hospital glucose testing easier and more secure while continuing to offer accuracy, freedom from interference, and... Read moreBusiness
view channel
Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
Johnson & Johnson (New Brunswick, N.J., USA) and Shockwave Medical (Santa Clara, CA, USA) have entered into a definitive agreement under which Johnson & Johnson will acquire all of Shockwave’s... Read more