Pfizer Evaluates Oral COVID-19 Antiviral Therapy for Prescribing at First Sign of Infection Without Requiring Hospitalization
|
By HospiMedica International staff writers Posted on 25 Mar 2021 |
Illustration
Pfizer Inc. (New York, NY, USA) is progressing to multiple ascending doses after completing the dosing of single ascending doses in a Phase 1 study in healthy adults to evaluate the safety and tolerability of an investigational, novel oral antiviral therapeutic for SARS-CoV-2, the virus that causes COVID-19.
The oral antiviral clinical candidate PF-07321332, a SARS-CoV2-3CL protease inhibitor (link is external), has demonstrated potent in vitro anti-viral activity against SARS-CoV-2, as well as activity against other coronaviruses, suggesting potential for use in the treatment of COVID-19 as well as potential use to address future coronavirus threats.
Protease inhibitors bind to a viral enzyme (called a protease), preventing the virus from replicating in the cell. Protease inhibitors have been effective at treating other viral pathogens such as HIV and hepatitis C virus, both alone and in combination with other antivirals. Currently marketed therapeutics that target viral proteases are not generally associated with toxicity and as such, this class of molecules may potentially provide well-tolerated treatments against COVID-19.
The Phase 1 trial is a randomized, double-blind, sponsor-open, placebo-controlled, single- and multiple-dose escalation study in healthy adults evaluating the safety, tolerability and pharmacokinetics of PF-07321332. Initiation of this study is supported by preclinical studies that demonstrated the antiviral activity of this potential first-in-class SARS-CoV-2 therapeutic designed specifically to inhibit replication of the SARS-CoV2 virus. The structure of PF-07321332, together with the pre-clinical data, will be shared in a COVID-19 session of the Spring American Chemical Society meeting on April 6. Pfizer is also investigating an intravenously administered investigational protease inhibitor, PF-07304814, which is currently in a Phase 1b multi-dose trial in hospitalized clinical trial participants with COVID-19.
“Tackling the COVID-19 pandemic requires both prevention via vaccine and targeted treatment for those who contract the virus. Given the way that SARS-CoV-2 is mutating and the continued global impact of COVID-19, it appears likely that it will be critical to have access to therapeutic options both now and beyond the pandemic,” said Mikael Dolsten, MD, PhD., Chief Scientific Officer and President, Worldwide Research, Development and Medical of Pfizer. “We have designed PF-07321332 as a potential oral therapy that could be prescribed at the first sign of infection, without requiring that patients are hospitalized or in critical care. At the same time, Pfizer’s intravenous antiviral candidate is a potential novel treatment option for hospitalized patients. Together, the two have the potential to create an end to end treatment paradigm that complements vaccination in cases where disease still occurs.”
Related Links:
Pfizer Inc.
The oral antiviral clinical candidate PF-07321332, a SARS-CoV2-3CL protease inhibitor (link is external), has demonstrated potent in vitro anti-viral activity against SARS-CoV-2, as well as activity against other coronaviruses, suggesting potential for use in the treatment of COVID-19 as well as potential use to address future coronavirus threats.
Protease inhibitors bind to a viral enzyme (called a protease), preventing the virus from replicating in the cell. Protease inhibitors have been effective at treating other viral pathogens such as HIV and hepatitis C virus, both alone and in combination with other antivirals. Currently marketed therapeutics that target viral proteases are not generally associated with toxicity and as such, this class of molecules may potentially provide well-tolerated treatments against COVID-19.
The Phase 1 trial is a randomized, double-blind, sponsor-open, placebo-controlled, single- and multiple-dose escalation study in healthy adults evaluating the safety, tolerability and pharmacokinetics of PF-07321332. Initiation of this study is supported by preclinical studies that demonstrated the antiviral activity of this potential first-in-class SARS-CoV-2 therapeutic designed specifically to inhibit replication of the SARS-CoV2 virus. The structure of PF-07321332, together with the pre-clinical data, will be shared in a COVID-19 session of the Spring American Chemical Society meeting on April 6. Pfizer is also investigating an intravenously administered investigational protease inhibitor, PF-07304814, which is currently in a Phase 1b multi-dose trial in hospitalized clinical trial participants with COVID-19.
“Tackling the COVID-19 pandemic requires both prevention via vaccine and targeted treatment for those who contract the virus. Given the way that SARS-CoV-2 is mutating and the continued global impact of COVID-19, it appears likely that it will be critical to have access to therapeutic options both now and beyond the pandemic,” said Mikael Dolsten, MD, PhD., Chief Scientific Officer and President, Worldwide Research, Development and Medical of Pfizer. “We have designed PF-07321332 as a potential oral therapy that could be prescribed at the first sign of infection, without requiring that patients are hospitalized or in critical care. At the same time, Pfizer’s intravenous antiviral candidate is a potential novel treatment option for hospitalized patients. Together, the two have the potential to create an end to end treatment paradigm that complements vaccination in cases where disease still occurs.”
Related Links:
Pfizer Inc.
Latest COVID-19 News
- Low-Cost System Detects SARS-CoV-2 Virus in Hospital Air Using High-Tech Bubbles
- World's First Inhalable COVID-19 Vaccine Approved in China
- COVID-19 Vaccine Patch Fights SARS-CoV-2 Variants Better than Needles
- Blood Viscosity Testing Can Predict Risk of Death in Hospitalized COVID-19 Patients
- ‘Covid Computer’ Uses AI to Detect COVID-19 from Chest CT Scans
- MRI Lung-Imaging Technique Shows Cause of Long-COVID Symptoms
- Chest CT Scans of COVID-19 Patients Could Help Distinguish Between SARS-CoV-2 Variants
- Specialized MRI Detects Lung Abnormalities in Non-Hospitalized Long COVID Patients
- AI Algorithm Identifies Hospitalized Patients at Highest Risk of Dying From COVID-19
- Sweat Sensor Detects Key Biomarkers That Provide Early Warning of COVID-19 and Flu
- Study Assesses Impact of COVID-19 on Ventilation/Perfusion Scintigraphy
- CT Imaging Study Finds Vaccination Reduces Risk of COVID-19 Associated Pulmonary Embolism
- Third Day in Hospital a ‘Tipping Point’ in Severity of COVID-19 Pneumonia
- Longer Interval Between COVID-19 Vaccines Generates Up to Nine Times as Many Antibodies
- AI Model for Monitoring COVID-19 Predicts Mortality Within First 30 Days of Admission
- AI Predicts COVID Prognosis at Near-Expert Level Based Off CT Scans
Channels
Artificial Intelligence
view channel
AI Tool Predicts Unplanned Care and Symptom Burden in Cancer Survivors
Unplanned emergency visits and hospitalizations remain common in cancer survivorship, when routine clinical contact often tapers while new symptoms emerge. These events reflect unmet needs and disrupt... Read more
Automated Phone Speech Test Identifies Alzheimer’s Pathology for Prescreening
Alzheimer’s disease assessment and trial recruitment often rely on costly, invasive biomarker testing and clinic-based cognitive evaluations, limiting scalability as populations age. Providers and trial... Read moreCritical Care
view channel
Ring-Type Cuffless Monitor Becomes First Added to Official Hypertension Guidelines
Detecting nocturnal and morning hypertension often requires out-of-office assessment, but conventional cuff-type monitors can disrupt sleep. New national guidance in South Korea expands 24-hour monitoring... Read more
“Intelligent Tattoo” Method Detects Early Melanoma Signals
Melanoma is a lethal skin cancer in which earlier detection drives better outcomes. Current evaluation relies on visual inspection followed by biopsy, which can miss nascent lesions and lead to unnecessary... Read more
Reusable Intermittent Catheters Reduce Antibiotic Use Without Increasing Urinary Tract Infections
Intermittent self-catheterization, used to empty the bladder several times a day, can leave patients vulnerable to recurrent urinary tract infections and repeated antibiotic use. Reliance on single-use... Read moreSurgical Techniques
view channel
Fracture Plating System Combines Anatomical Fit with Streamlined Instrumentation
Fracture management involves diverse anatomies and injury patterns that can make plate fit and fixation strategy challenging. Surgeons need both intraoperative efficiency and flexibility, while hospitals... Read more
Pink Noise Stimulation Approach Could Support Safer Anesthesia
Maintaining stable unconsciousness during general anesthesia while minimizing drug exposure remains a core perioperative challenge. Sudden nociceptive surges can disrupt anesthetic depth, increasing neurological... Read morePatient Care
view channel
AI Avatar Doctor Improves Patient Understanding Before Radiotherapy
Radiation oncology consultations require patients to grasp complex concepts quickly, yet anxiety and information overload often undermine understanding and informed consent. Poor comprehension can also... Read more
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read moreHealth IT
view channel
AI-Native EHR Achieves EU Medical Device Certification
InterSystems (Boston, MA, USA) announced that its IntelliCare electronic health record (EHR) solutions have been certified as Class IIa medical devices under the European Union Medical Device Regulation... Read more
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read morePoint of Care
view channel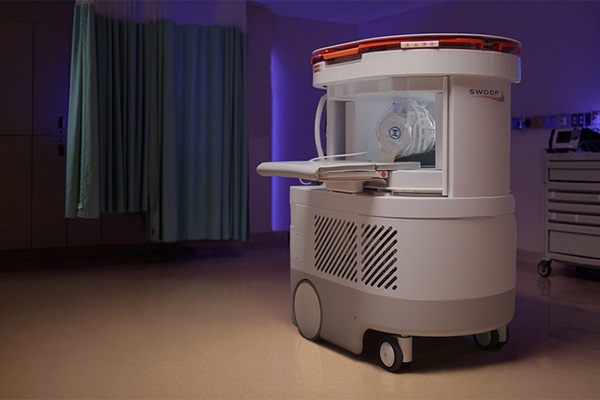
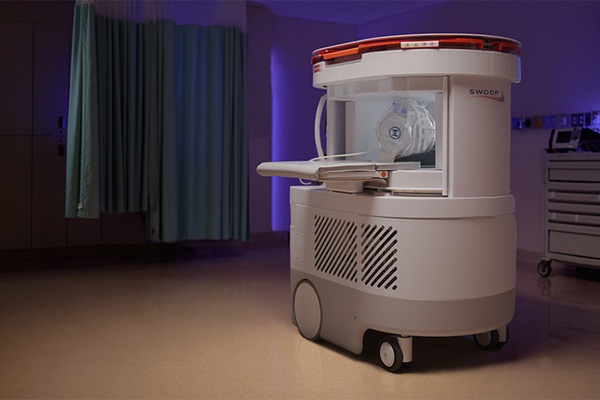
Portable MRI System Accelerates Emergency Brain Imaging and Triage
Emergency departments frequently face delays accessing conventional magnetic resonance imaging (MRI) for patients with suspected neurological emergencies. Such waits can slow triage, prolong boarding,... Read more