Merck and Ridgeback’s Experimental Oral COVID-19 Antiviral Halves Risk of Hospitalization or Death
|
By HospiMedica International staff writers Posted on 01 Oct 2021 |
Merck’s (Kenilworth, NJ, USA) experimental oral COVID-19 antiviral drug, molnupiravir, developed in collaboration with Ridgeback Biotherapeutics’ (Miami, FL, USA), significantly reduced the risk of hospitalization or death at a planned interim analysis of the Phase 3 MOVe-OUT trial in at risk, non-hospitalized adult patients with mild-to-moderate COVID-19.
The planned interim analysis evaluated data from 775 patients who were initially enrolled in the Phase 3 MOVe-OUT trial on or prior to Aug. 5, 2021. Eligibility criteria required that all patients had laboratory-confirmed mild-to-moderate COVID-19, with symptom onset within five days of study randomization. All patients were required to have at least one risk factor associated with poor disease outcome at study entry.
At the interim analysis, molnupiravir reduced the risk of hospitalization or death by approximately 50%; 7.3% of patients who received molnupiravir were either hospitalized or died through Day 29 following randomization (28/385), compared with 14.1% of placebo-treated patients (53/377). Through Day 29, no deaths were reported in patients who received molnupiravir, as compared to 8 deaths in patients who received placebo.
At the recommendation of an independent Data Monitoring Committee and in consultation with the U.S. Food and Drug Administration (FDA), recruitment into the study has been stopped early due to these positive results. Merck plans to submit an application for Emergency Use Authorization (EUA) to the U.S. FDA as soon as possible based on these findings and plans to submit marketing applications to other regulatory bodies worldwide.
“With these compelling results, we are optimistic that molnupiravir can become an important medicine as part of the global effort to fight the pandemic and will add to Merck’s unique legacy of bringing forward breakthroughs in infectious diseases when they are needed most,” said Robert M. Davis, chief executive officer and president, Merck. “Consistent with Merck’s unwavering commitment to save and improve lives, we will continue to work with regulatory agencies on our applications and do everything we can to bring molnupiravir to patients as quickly as possible.”
“With the virus continuing to circulate widely, and because therapeutic options currently available are infused and/or require access to a healthcare facility, antiviral treatments that can be taken at home to keep people with COVID-19 out of the hospital are critically needed,” added Wendy Holman, chief executive officer of Ridgeback Biotherapeutics. “We are very encouraged by the results from the interim analysis and hope molnupiravir, if authorized for use, can make a profound impact in controlling the pandemic.”
Related Links:
Ridgeback Biotherapeutics
Merck
Latest COVID-19 News
- Low-Cost System Detects SARS-CoV-2 Virus in Hospital Air Using High-Tech Bubbles
- World's First Inhalable COVID-19 Vaccine Approved in China
- COVID-19 Vaccine Patch Fights SARS-CoV-2 Variants Better than Needles
- Blood Viscosity Testing Can Predict Risk of Death in Hospitalized COVID-19 Patients
- ‘Covid Computer’ Uses AI to Detect COVID-19 from Chest CT Scans
- MRI Lung-Imaging Technique Shows Cause of Long-COVID Symptoms
- Chest CT Scans of COVID-19 Patients Could Help Distinguish Between SARS-CoV-2 Variants
- Specialized MRI Detects Lung Abnormalities in Non-Hospitalized Long COVID Patients
- AI Algorithm Identifies Hospitalized Patients at Highest Risk of Dying From COVID-19
- Sweat Sensor Detects Key Biomarkers That Provide Early Warning of COVID-19 and Flu
- Study Assesses Impact of COVID-19 on Ventilation/Perfusion Scintigraphy
- CT Imaging Study Finds Vaccination Reduces Risk of COVID-19 Associated Pulmonary Embolism
- Third Day in Hospital a ‘Tipping Point’ in Severity of COVID-19 Pneumonia
- Longer Interval Between COVID-19 Vaccines Generates Up to Nine Times as Many Antibodies
- AI Model for Monitoring COVID-19 Predicts Mortality Within First 30 Days of Admission
- AI Predicts COVID Prognosis at Near-Expert Level Based Off CT Scans
Channels
Critical Care
view channel
Mechanosensing-Based Approach Offers Promising Strategy to Treat Cardiovascular Fibrosis
Cardiac fibrosis, which involves the stiffening and scarring of heart tissue, is a fundamental feature of nearly every type of heart disease, from acute ischemic injuries to genetic cardiomyopathies.... Read more
AI Interpretability Tool for Photographed ECG Images Offers Pixel-Level Precision
The electrocardiogram (ECG) is a crucial diagnostic tool in modern medicine, used to detect heart conditions such as arrhythmias and structural abnormalities. Every year, millions of ECGs are performed... Read moreSurgical Techniques
view channel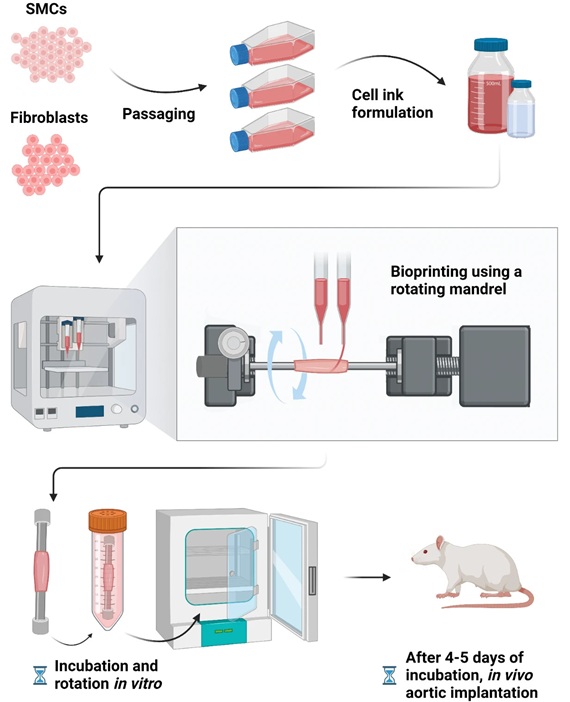
Bioprinted Aortas Offer New Hope for Vascular Repair
Current treatment options for severe cardiovascular diseases include using grafts made from a patient's own tissue (autologous) or synthetic materials. However, autologous grafts require invasive surgery... Read more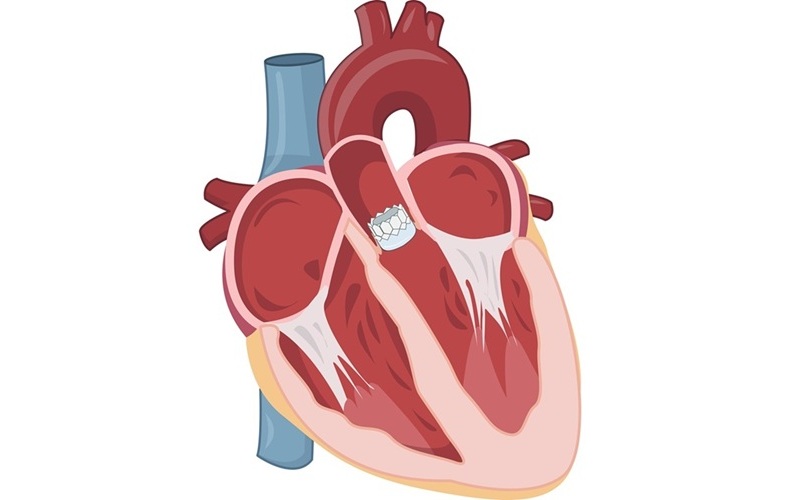
Early TAVR Intervention Reduces Cardiovascular Events in Asymptomatic Aortic Stenosis Patients
Each year, approximately 300,000 Americans are diagnosed with aortic stenosis (AS), a serious condition that results from the narrowing or blockage of the aortic valve in the heart. Two common treatments... Read more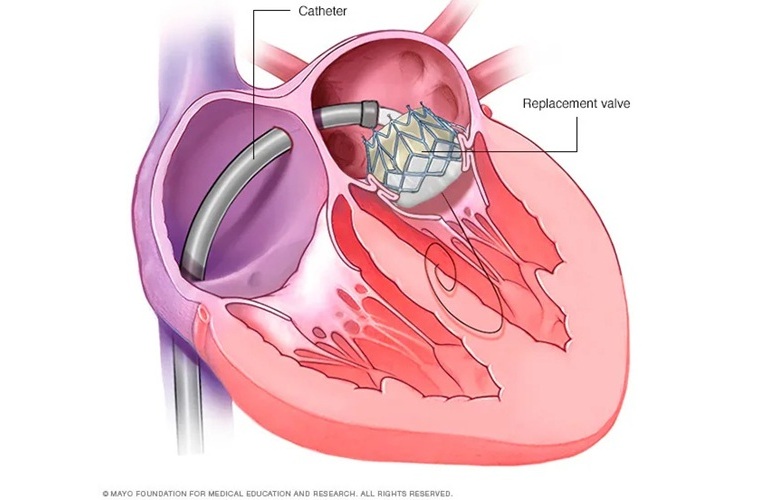
New Procedure Found Safe and Effective for Patients Undergoing Transcatheter Mitral Valve Replacement
In the United States, approximately four million people suffer from mitral valve regurgitation, the most common type of heart valve disease. As an alternative to open-heart surgery, transcatheter mitral... Read morePatient Care
view channel
Portable Biosensor Platform to Reduce Hospital-Acquired Infections
Approximately 4 million patients in the European Union acquire healthcare-associated infections (HAIs) or nosocomial infections each year, with around 37,000 deaths directly resulting from these infections,... Read moreFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read more
Smartwatches Could Detect Congestive Heart Failure
Diagnosing congestive heart failure (CHF) typically requires expensive and time-consuming imaging techniques like echocardiography, also known as cardiac ultrasound. Previously, detecting CHF by analyzing... Read moreBusiness
view channel
Expanded Collaboration to Transform OR Technology Through AI and Automation
The expansion of an existing collaboration between three leading companies aims to develop artificial intelligence (AI)-driven solutions for smart operating rooms with sophisticated monitoring and automation.... Read more