Novel SMP Responds Directly to Enzymatic Activity
|
By HospiMedica International staff writers Posted on 14 Mar 2019 |

Image: A shape memory polymer (SMP) that responds to enzymes could promote healing (Photo courtesy of Syracuse University).
A new study shows how a novel shape memory polymer (SMP) that responds to exposure to enzymes could soon be used to treat open wounds, infections, and cancer.
Developed by researchers at Syracuse University (NY, USA) and Bucknell University (Lewisburg, PA, USA), the new SMP includes a shape fixing component, poly(ε-caprolactone) (PCL), that is vulnerable to enzymatic degradation, and a shape memory component, Pellethane, that is enzymatically stable. As the shape fixing PCL component undergoes enzymatically-catalyzed degradation, the SMP can return to its original, programmed shape. It requires no additional trigger, such as a change in temperature, and could thus respond to cellular activity.
The researchers also analyzed material properties, shape memory performance, and cytocompatibility of the enzymatically-catalyzed shape memory response. The results demonstrate enzymatic recovery, as contraction of tensile specimens, and that shape recovery is achieved by degradation of the PCL shape-fixing phase. In addition, both the materials composing the SMP and the process of enzymatic shape recovery were cytocompatible. The study describing the SMP was published on January 15, 2019, in Acta Biomaterialia.
“We anticipate that the materials we're developing could have broad application in health care. For example, our SMPs could be used in drugs that only activate when the target cells or organ are in the desired physiological state, in scaffolds that guide tissue regeneration in response to the behavior of the regenerating tissue itself, and in decision-making biosensors that guide patient treatment more effectively,” said senior author Professor James Henderson, PhD, of Syracuse University. “We're very excited to have achieved these first enzymatically responsive SMPs.”
“The enzymatic sensitivity of the material allows it to respond directly to cell behavior. For instance, you could place it over a wound, and as the tissue remodeled and degraded it, the SMP would slowly pull the wound closed,” said lead author candidate Shelby Buffington, MSc, a biomedical engineering PhD candidate at Syracuse University. “It could be adapted to play a role in treating infections and cancer by adjusting the material's chemistry.”
SMPs include foams, scaffolds, meshes, and other polymeric substrates that have the ability to return from a deformed state (temporary shape) to their original (permanent) shape induced by an external stimulus, such as a temperature change, an electric or magnetic field, light, or a chemical solution. As well as polymers in general, SMPs also cover a wide property-range from stable to biodegradable, from soft to hard, and from elastic to rigid, depending on the structural units that constitute the SMP.
Related Links:
Syracuse University
Bucknell University
Developed by researchers at Syracuse University (NY, USA) and Bucknell University (Lewisburg, PA, USA), the new SMP includes a shape fixing component, poly(ε-caprolactone) (PCL), that is vulnerable to enzymatic degradation, and a shape memory component, Pellethane, that is enzymatically stable. As the shape fixing PCL component undergoes enzymatically-catalyzed degradation, the SMP can return to its original, programmed shape. It requires no additional trigger, such as a change in temperature, and could thus respond to cellular activity.
The researchers also analyzed material properties, shape memory performance, and cytocompatibility of the enzymatically-catalyzed shape memory response. The results demonstrate enzymatic recovery, as contraction of tensile specimens, and that shape recovery is achieved by degradation of the PCL shape-fixing phase. In addition, both the materials composing the SMP and the process of enzymatic shape recovery were cytocompatible. The study describing the SMP was published on January 15, 2019, in Acta Biomaterialia.
“We anticipate that the materials we're developing could have broad application in health care. For example, our SMPs could be used in drugs that only activate when the target cells or organ are in the desired physiological state, in scaffolds that guide tissue regeneration in response to the behavior of the regenerating tissue itself, and in decision-making biosensors that guide patient treatment more effectively,” said senior author Professor James Henderson, PhD, of Syracuse University. “We're very excited to have achieved these first enzymatically responsive SMPs.”
“The enzymatic sensitivity of the material allows it to respond directly to cell behavior. For instance, you could place it over a wound, and as the tissue remodeled and degraded it, the SMP would slowly pull the wound closed,” said lead author candidate Shelby Buffington, MSc, a biomedical engineering PhD candidate at Syracuse University. “It could be adapted to play a role in treating infections and cancer by adjusting the material's chemistry.”
SMPs include foams, scaffolds, meshes, and other polymeric substrates that have the ability to return from a deformed state (temporary shape) to their original (permanent) shape induced by an external stimulus, such as a temperature change, an electric or magnetic field, light, or a chemical solution. As well as polymers in general, SMPs also cover a wide property-range from stable to biodegradable, from soft to hard, and from elastic to rigid, depending on the structural units that constitute the SMP.
Related Links:
Syracuse University
Bucknell University
Latest Critical Care News
- Wheeze-Counting Wearable Device Monitors Patient's Breathing In Real Time
- Wearable Multiplex Biosensors Could Revolutionize COPD Management
- New Low-Energy Defibrillation Method Controls Cardiac Arrhythmias
- New Machine Learning Models Help Predict Heart Disease Risk in Women
- Deep-Learning Model Predicts Arrhythmia 30 Minutes before Onset
- Breakthrough Technology Combines Detection and Treatment of Nerve-Related Disorders in Single Procedure
- Plasma Irradiation Promotes Faster Bone Healing
- New Device Treats Acute Kidney Injury from Sepsis
- Study Confirms Safety of DCB-Only Strategy for Treating De Novo Left Main Coronary Artery Disease
- Revascularization Improves Quality of Life for Patients with Chronic Limb Threatening Ischemia
- AI-Driven Prediction Models Accurately Predict Critical Care Patient Deterioration
- Preventive PCI for High-Risk Coronary Plaques Reduces Cardiac Events
- AI Diagnostic Tool Guides Rapid Diagnosis and Prediction of Sepsis
- World's First AI-Powered Sepsis Alert System Detects Sepsis in One Minute
- Smartphone Magnetometer Uses Magnetized Hydrogel to Measure Biomarkers for Disease Diagnosis
- New Technology to Revolutionize Valvular Heart Disease Care
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreSurgical Techniques
view channel
Flexible Microdisplay Visualizes Brain Activity in Real-Time To Guide Neurosurgeons
During brain surgery, neurosurgeons need to identify and preserve regions responsible for critical functions while removing harmful tissue. Traditionally, neurosurgeons rely on a team of electrophysiologists,... Read more.jpg)
Next-Gen Computer Assisted Vacuum Thrombectomy Technology Rapidly Removes Blood Clots
Pulmonary embolism (PE) occurs when a blood clot blocks one of the arteries in the lungs. Often, these clots originate from the leg or another part of the body, a condition known as deep vein thrombosis,... Read more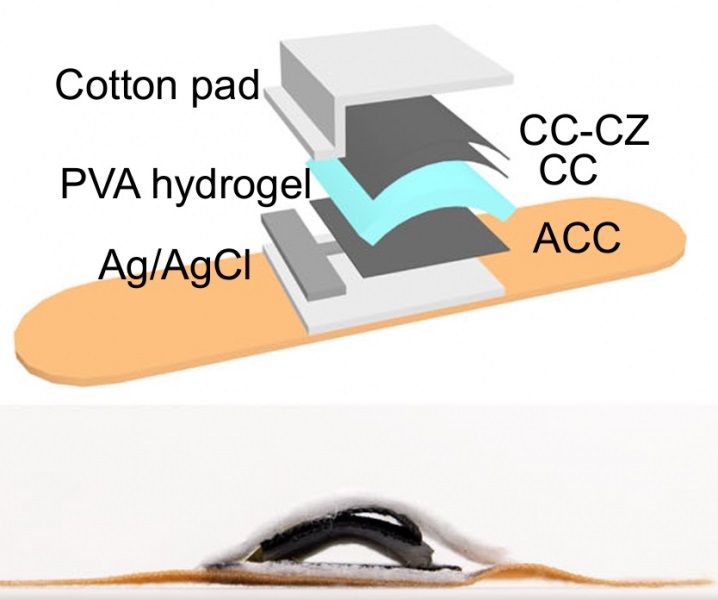
Hydrogel-Based Miniaturized Electric Generators to Power Biomedical Devices
The development of engineered devices that can harvest and convert the mechanical motion of the human body into electricity is essential for powering bioelectronic devices. This mechanoelectrical energy... Read moreWearable Technology Monitors and Analyzes Surgeons' Posture during Long Surgical Procedures
The physical strain associated with the static postures maintained by neurosurgeons during long operations can lead to fatigue and musculoskeletal problems. An objective assessment of surgical ergonomics... Read morePatient Care
view channel
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read more
Next Gen ICU Bed to Help Address Complex Critical Care Needs
As the critical care environment becomes increasingly demanding and complex due to evolving hospital needs, there is a pressing requirement for innovations that can facilitate patient recovery.... Read moreGroundbreaking AI-Powered UV-C Disinfection Technology Redefines Infection Control Landscape
Healthcare-associated infection (HCAI) is a widespread complication in healthcare management, posing a significant health risk due to its potential to increase patient morbidity and mortality, prolong... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more
New Generation Glucose Hospital Meter System Ensures Accurate, Interference-Free and Safe Use
A new generation glucose hospital meter system now comes with several features that make hospital glucose testing easier and more secure while continuing to offer accuracy, freedom from interference, and... Read moreBusiness
view channel
Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
Johnson & Johnson (New Brunswick, N.J., USA) and Shockwave Medical (Santa Clara, CA, USA) have entered into a definitive agreement under which Johnson & Johnson will acquire all of Shockwave’s... Read more