First-Of-Its-Kind 4K Endoscope for Arthroscopic Surgical Procedures Launched Globally
|
By HospiMedica International staff writers Posted on 07 Nov 2022 |

A first-of-its-kind 4K endoscope designed for use in arthroscopic surgical procedures is revolutionizing the market by delivering 4K optics in a single-use device – providing first-time quality for every procedure.
Integrated Endoscopy (Irvine, CA, USA) has launched its second generation NUVIS (pronounced “nu-vee”) Single-Use Arthroscope, a first-of-its-kind 4K endoscope designed for use in arthroscopic surgical procedures. The release of the Gen II NUVIS features a patent pending simplified optical train design which reduces the number of optical elements by over 60% while boosting image quality to 4K. Not only is the image quality superior, but the Gen II NUVIS will also be substantially faster and less expensive to manufacture and amenable to high-volume automation.
As part of the Gen II launch, Integrated Endoscopy is releasing multiple versions of NUVIS to make the technology compatible with most cannulas on the market. NUVIS is also compatible with most installed video towers and will support a 4K camera system without requiring additional capital equipment. NUVIS has a built-in battery powered LED, which eliminates the light box and bulky light cord. Integrated Endoscopy’s Gen II NUVIS single-use, 4K, battery operated cordless arthroscope is FDA 501(k) cleared, CE marked, and is being registered in a number of markets around the world. As manufacturing capacity increases, the company will continue to expand its global distribution network to provide this unique technology to the arthroscopic surgical community.
“The release of the Gen II NUVIS is a huge milestone for the company in terms of image quality and compatibility,” said Brad Sharp, Integrated Endoscopy’s CEO. “Our next-gen arthroscope also provides a substantial decrease in manufacturing costs while simultaneously increasing ease of manufacturability. The demand we have seen for NUVIS far exceeds our current manufacturing capacity, so the efficiencies the Gen II NUVIS affords will greatly aid in shortening production time and costs. With a global market of over 1.5 million units per month, increasing our manufacturing capacity will be a major focus for the company moving forward.”
Related Links:
Latest Surgical Techniques News
- First-Ever Contact Force Pulsed Field Ablation System to Transform Treatment of Ventricular Arrhythmias
- Caterpillar Robot with Built-In Steering System Crawls Easily Through Loops and Bends
- Tiny Wraparound Electronic Implants to Revolutionize Treatment of Spinal Cord Injuries
- Small, Implantable Cardiac Pump to Help Children Awaiting Heart Transplant
- Gastrointestinal Imaging Capsule a Game-Changer in Esophagus Surveillance and Treatment
- World’s Smallest Laser Probe for Brain Procedures Facilitates Ablation of Full Range of Targets
- Artificial Intelligence Broadens Diagnostic Abilities of Conventional Coronary Angiography
- AI-Powered Surgical Visualization Tool Supports Surgeons' Visual Recognition in Real Time
- Cutting-Edge Robotic Bronchial Endoscopic System Provides Prompt Intervention during Emergencies
- Handheld Device for Fluorescence-Guided Surgery a Game Changer for Removal of High-Grade Glioma Brain Tumors
- Porous Gel Sponge Facilitates Rapid Hemostasis and Wound Healing
- Novel Rigid Endoscope System Enables Deep Tissue Imaging During Surgery
- Robotic Nerve ‘Cuffs’ Could Treat Various Neurological Conditions
- Flexible Microdisplay Visualizes Brain Activity in Real-Time To Guide Neurosurgeons

- Next-Gen Computer Assisted Vacuum Thrombectomy Technology Rapidly Removes Blood Clots
- Hydrogel-Based Miniaturized Electric Generators to Power Biomedical Devices
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreCritical Care
view channel
One-Hour Endoscopic Procedure Could Eliminate Need for Insulin for Type 2 Diabetes
Over 37 million Americans are diagnosed with diabetes, and more than 90% of these cases are Type 2 diabetes. This form of diabetes is most commonly seen in individuals over 45, though an increasing number... Read moreAI Can Prioritize Emergecny Department Patients Requiring Urgent Treatment
Emergency departments across the world are facing severe overcrowding and excessive demands, but a new study indicates that artificial intelligence (AI) might soon assist in prioritizing patients who require... Read more
AI to Improve Diagnosis of Atrial Fibrillation
Abnormal heart rhythms frequently arise from—and contribute to—structural abnormalities in the heart. Atrial fibrillation is a specific type of abnormal rhythm that may not be consistently present, often... Read more
Stretchable Microneedles to Help In Accurate Tracking of Abnormalities and Identifying Rapid Treatment
The field of personalized medicine is transforming rapidly, with advancements like wearable devices and home testing kits making it increasingly easy to monitor a wide range of health metrics, from heart... Read morePatient Care
view channelFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel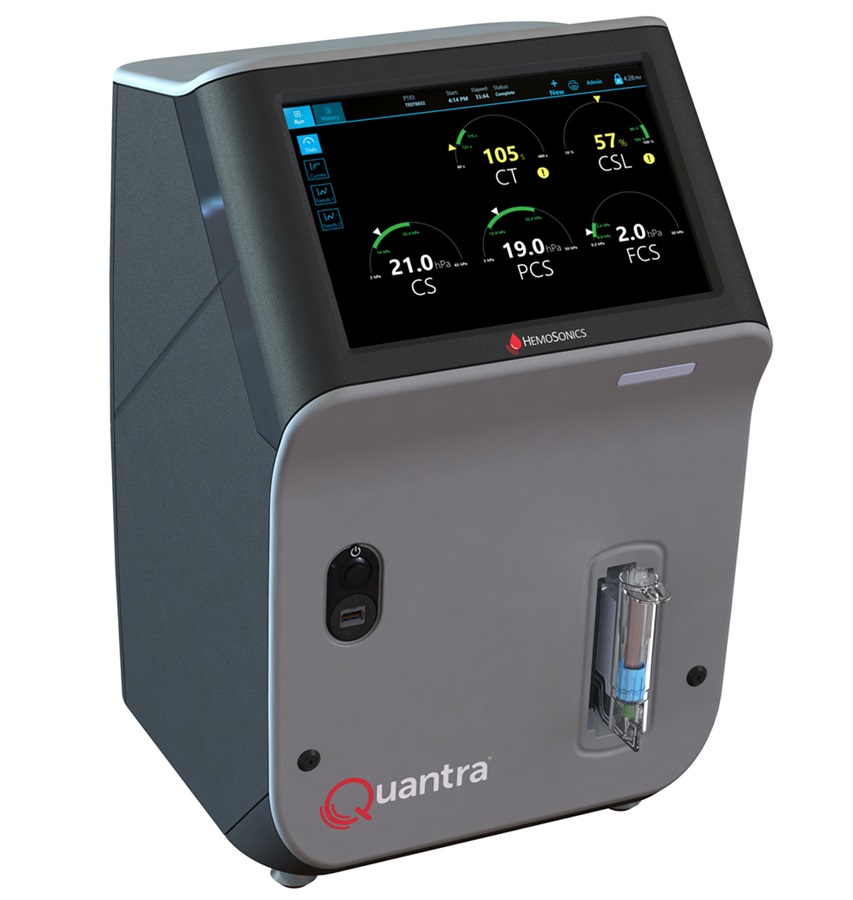
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more
New Generation Glucose Hospital Meter System Ensures Accurate, Interference-Free and Safe Use
A new generation glucose hospital meter system now comes with several features that make hospital glucose testing easier and more secure while continuing to offer accuracy, freedom from interference, and... Read moreBusiness
view channel
Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
Johnson & Johnson (New Brunswick, N.J., USA) and Shockwave Medical (Santa Clara, CA, USA) have entered into a definitive agreement under which Johnson & Johnson will acquire all of Shockwave’s... Read more