Functional Microrobots Could Harbor Bioengineering Apps
|
By HospiMedica International staff writers Posted on 22 May 2017 |

Image: A bullet-shape microrobot with a programmed inner cavity, swimming in 5% H2O2 (Photo courtesy of Max Planck Institute).
A new study suggests that untethered micron-scale mobile robots can navigate and non-invasively perform specific tasks inside hard-to-reach body sites.
Currently being designed, fabricated, and tested at the Max Planck Institute for Intelligent Systems and Carnegie Mellon University, the first-generation microrobots will be able to deliver therapeutics and other cargo to targeted body sites, as well as to enclosed organ-on-a-chip microfluidic devices with live cells. A new two-step approach is use to provide the microrobotic devices with desirable functions. The first step uses three-dimensional (3D) laser lithography to crosslink light-responsive polymers.
In the second step, the formed chemically homogenous base structure is functionalized by modifying it at specific geometric sites with chemically compatible small molecules which introduce new chemical groups using selective 3D laser lithography illumination; this causes an unreacted polymer precursor to be removed, and a new precursor with the desired chemical functionality is introduced in its place. The technique allows the microrobots to be fabricated with high versatility.
To prove the concept, the researchers prepared a bullet-shaped micro-swimmer, in which an inner cavity was selectively modified with catalytic platinum nanoparticles. The researchers also designed a microflower structure bearing orthogonal biotin, thiol, and alkyne groups at precisely defined positions. According to the researchers, the new sub-millimeter constructs hold a myriad of applications in various fields besides microrobots, including targeted delivery, tissue engineering, self-organizing systems, programmable matter, and soft microactuators. The study was published on May 8, 2017, in Lab Chip.
“Our key objective is to develop new methods of making miniaturized materials that are performing intelligently in complex and unstable environment,” said lead author postdoctoral researcher Hakan Ceylan, PhD, of the Max Planck Institute. “In the near future, probably in around 10 years, this could have tremendous applications in tissue engineering and regenerative medicine, while in the longer term, it could revolutionize the treatment of genetic diseases by single cell-level protein or nucleic acid delivery.”
Currently being designed, fabricated, and tested at the Max Planck Institute for Intelligent Systems and Carnegie Mellon University, the first-generation microrobots will be able to deliver therapeutics and other cargo to targeted body sites, as well as to enclosed organ-on-a-chip microfluidic devices with live cells. A new two-step approach is use to provide the microrobotic devices with desirable functions. The first step uses three-dimensional (3D) laser lithography to crosslink light-responsive polymers.
In the second step, the formed chemically homogenous base structure is functionalized by modifying it at specific geometric sites with chemically compatible small molecules which introduce new chemical groups using selective 3D laser lithography illumination; this causes an unreacted polymer precursor to be removed, and a new precursor with the desired chemical functionality is introduced in its place. The technique allows the microrobots to be fabricated with high versatility.
To prove the concept, the researchers prepared a bullet-shaped micro-swimmer, in which an inner cavity was selectively modified with catalytic platinum nanoparticles. The researchers also designed a microflower structure bearing orthogonal biotin, thiol, and alkyne groups at precisely defined positions. According to the researchers, the new sub-millimeter constructs hold a myriad of applications in various fields besides microrobots, including targeted delivery, tissue engineering, self-organizing systems, programmable matter, and soft microactuators. The study was published on May 8, 2017, in Lab Chip.
“Our key objective is to develop new methods of making miniaturized materials that are performing intelligently in complex and unstable environment,” said lead author postdoctoral researcher Hakan Ceylan, PhD, of the Max Planck Institute. “In the near future, probably in around 10 years, this could have tremendous applications in tissue engineering and regenerative medicine, while in the longer term, it could revolutionize the treatment of genetic diseases by single cell-level protein or nucleic acid delivery.”
Latest Health IT News
- Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
- Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
- AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
- AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
- First Fully Autonomous Generative AI Personalized Medical Authorizations System Reduces Care Delay
- Electronic Health Records May Be Key to Improving Patient Care, Study Finds
- AI Trained for Specific Vocal Biomarkers Could Accurately Predict Coronary Artery Disease
- First-Ever AI Test for Early Diagnosis of Alzheimer’s to Be Expanded to Diagnosis of Parkinson’s Disease
- New Self-Learning AI-Based Algorithm Reads Electrocardiograms to Spot Unseen Signs of Heart Failure
- Autonomous Robot Performs COVID-19 Nasal Swab Tests

- Statistical Tool Predicts COVID-19 Peaks Worldwide
- Wireless-Controlled Soft Neural Implant Stimulates Brain Cells
- Tiny Polymer Stent Could Treat Pediatric Urethral Strictures
- Human Torso Simulator Helps Design Brace Innovations
- 3D Bioprinting Rebuilds the Human Heart
- Nanodrone Detects Toxic Gases in Hazardous Environments
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreCritical Care
view channel
Wheeze-Counting Wearable Device Monitors Patient's Breathing In Real Time
Lung diseases like asthma, chronic obstructive pulmonary disease (COPD), lung cancer, bronchitis, and infections such as pneumonia, rank among the leading causes of death worldwide. Traditionally, medical... Read more
Wearable Multiplex Biosensors Could Revolutionize COPD Management
Chronic obstructive pulmonary disease (COPD) ranks as the third leading cause of death worldwide. Acute exacerbations of COPD (AECOPD), which are often triggered by lung infections, accelerate the disease's... Read moreSurgical Techniques
view channel
Flexible Microdisplay Visualizes Brain Activity in Real-Time To Guide Neurosurgeons
During brain surgery, neurosurgeons need to identify and preserve regions responsible for critical functions while removing harmful tissue. Traditionally, neurosurgeons rely on a team of electrophysiologists,... Read more.jpg)
Next-Gen Computer Assisted Vacuum Thrombectomy Technology Rapidly Removes Blood Clots
Pulmonary embolism (PE) occurs when a blood clot blocks one of the arteries in the lungs. Often, these clots originate from the leg or another part of the body, a condition known as deep vein thrombosis,... Read more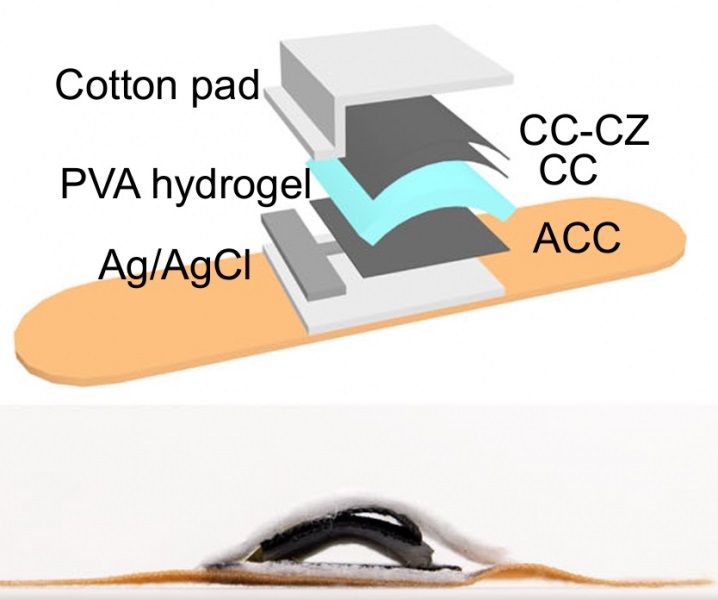
Hydrogel-Based Miniaturized Electric Generators to Power Biomedical Devices
The development of engineered devices that can harvest and convert the mechanical motion of the human body into electricity is essential for powering bioelectronic devices. This mechanoelectrical energy... Read moreWearable Technology Monitors and Analyzes Surgeons' Posture during Long Surgical Procedures
The physical strain associated with the static postures maintained by neurosurgeons during long operations can lead to fatigue and musculoskeletal problems. An objective assessment of surgical ergonomics... Read morePatient Care
view channel
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read more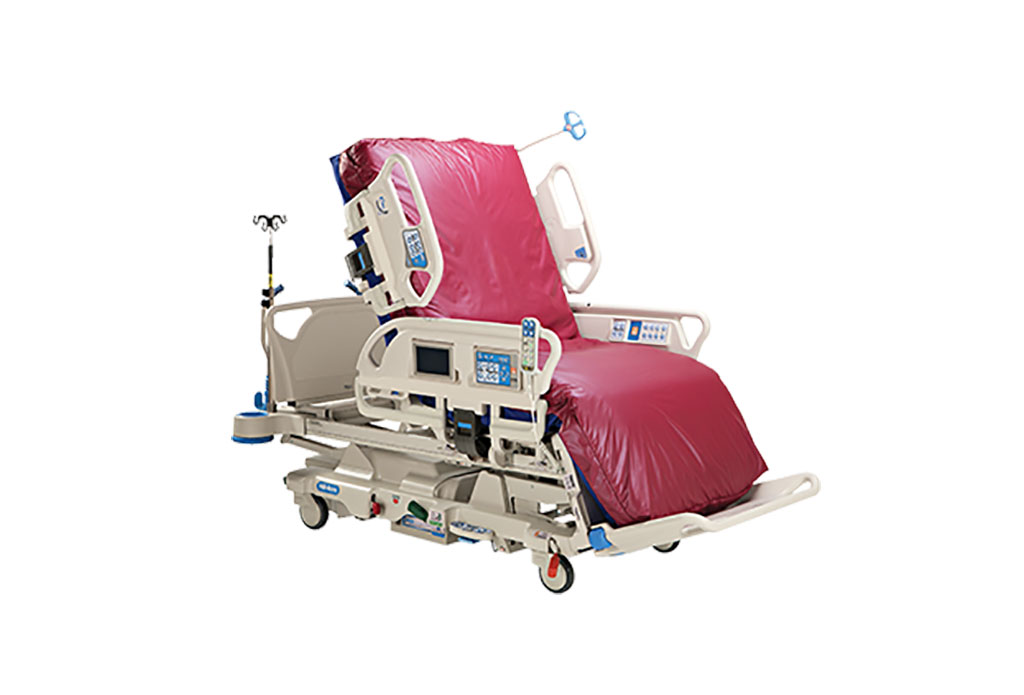
Next Gen ICU Bed to Help Address Complex Critical Care Needs
As the critical care environment becomes increasingly demanding and complex due to evolving hospital needs, there is a pressing requirement for innovations that can facilitate patient recovery.... Read moreGroundbreaking AI-Powered UV-C Disinfection Technology Redefines Infection Control Landscape
Healthcare-associated infection (HCAI) is a widespread complication in healthcare management, posing a significant health risk due to its potential to increase patient morbidity and mortality, prolong... Read morePoint of Care
view channel
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more
New Generation Glucose Hospital Meter System Ensures Accurate, Interference-Free and Safe Use
A new generation glucose hospital meter system now comes with several features that make hospital glucose testing easier and more secure while continuing to offer accuracy, freedom from interference, and... Read moreBusiness
view channel
Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
Johnson & Johnson (New Brunswick, N.J., USA) and Shockwave Medical (Santa Clara, CA, USA) have entered into a definitive agreement under which Johnson & Johnson will acquire all of Shockwave’s... Read more













.jpg)
