FDA Urges Hospitals to Check Mattresses
|
By HospiMedica International staff writers Posted on 16 May 2013 |
The US Food and Drug Administration (FDA, Silver Spring, MD, USA) urge healthcare professionals to replace worn medical bed mattress covers to prevent the spread of contamination.
The warning follows 458 reports submitted to the FDA of medical bed mattress covers failing to prevent blood and body fluid ingress between January 2011 and January 2013. Fluid ingress may occur if mattress covers become worn or damaged from small holes or rips in the fabric or from incorrect cleaning, allowing effusions from one patient to penetrate the cover and leak out later, when another patient is placed on the same bed. Damaged covers can also lead to the mattress itself retaining seeping fluids, and the zipper on the mattress cover may also allow fluid to penetrate the mattress.
The FDA therefore recommend that healthcare professionals regularly inspect the mattress covers for visible signs of damage, removing them and inspecting their inside surface (as well as the mattress itself), for any visible signs of damage or wear such as cuts, tears, cracks, pinholes, snags or stains. Once the mattress cover is removed, the mattress should also be inspected for wet spots, staining, or signs of damage or wear on all sides, including the bottom of the mattress. Any medical bed mattress and/or cover with visible signs of damage or wear should be replaced immediately.
“Inspection may prove challenging because most mattress covers are a dark color,” cautioned the FDA. “Healthcare professionals should also create an inspection plan for all mattress covers used in the facility, and any damaged or leaking mattress covers should be replaced immediately.”
Medical bed mattress covers protect mattresses by keeping blood and body fluids from getting into the core of the mattress through water-resistance, waterproofing, or water repelling. These effects may be diluted over time and may vary from mattress cover to mattress cover. Healthcare staff may be largely unaware of the hazards posed by damaged protective coverings on medical beds and risks associated with them, including widespread contamination during infection outbreaks.
Related Links:
US Food and Drug Administration
The warning follows 458 reports submitted to the FDA of medical bed mattress covers failing to prevent blood and body fluid ingress between January 2011 and January 2013. Fluid ingress may occur if mattress covers become worn or damaged from small holes or rips in the fabric or from incorrect cleaning, allowing effusions from one patient to penetrate the cover and leak out later, when another patient is placed on the same bed. Damaged covers can also lead to the mattress itself retaining seeping fluids, and the zipper on the mattress cover may also allow fluid to penetrate the mattress.
The FDA therefore recommend that healthcare professionals regularly inspect the mattress covers for visible signs of damage, removing them and inspecting their inside surface (as well as the mattress itself), for any visible signs of damage or wear such as cuts, tears, cracks, pinholes, snags or stains. Once the mattress cover is removed, the mattress should also be inspected for wet spots, staining, or signs of damage or wear on all sides, including the bottom of the mattress. Any medical bed mattress and/or cover with visible signs of damage or wear should be replaced immediately.
“Inspection may prove challenging because most mattress covers are a dark color,” cautioned the FDA. “Healthcare professionals should also create an inspection plan for all mattress covers used in the facility, and any damaged or leaking mattress covers should be replaced immediately.”
Medical bed mattress covers protect mattresses by keeping blood and body fluids from getting into the core of the mattress through water-resistance, waterproofing, or water repelling. These effects may be diluted over time and may vary from mattress cover to mattress cover. Healthcare staff may be largely unaware of the hazards posed by damaged protective coverings on medical beds and risks associated with them, including widespread contamination during infection outbreaks.
Related Links:
US Food and Drug Administration
Latest Patient Care News
- AI Avatar Doctor Improves Patient Understanding Before Radiotherapy
- Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization

- Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
- Next Gen ICU Bed to Help Address Complex Critical Care Needs
- Groundbreaking AI-Powered UV-C Disinfection Technology Redefines Infection Control Landscape
- Clean Hospitals Can Reduce Antibiotic Resistance, Save Lives
- Smart Hospital Beds Improve Accuracy of Medical Diagnosis
- New Fast Endoscope Drying System Improves Productivity and Traceability
- World’s First Automated Endoscope Cleaner Fights Antimicrobial Resistance
- Portable High-Capacity Digital Stretcher Scales Provide Precision Weighing for Patients in ER
- Portable Clinical Scale with Remote Indicator Allows for Flexible Patient Weighing Use
Channels
Artificial Intelligence
view channel
AI Platform Supports Noninvasive Remote Hemodynamic Monitoring in Heart Failure
Heart failure remains a leading cause of hospitalization in adults over 65, affecting more than 6.7 million people in the U.S. Clinicians often lose visibility into hemodynamic deterioration once patients... Read more
AI Tool Predicts Unplanned Care and Symptom Burden in Cancer Survivors
Unplanned emergency visits and hospitalizations remain common in cancer survivorship, when routine clinical contact often tapers while new symptoms emerge. These events reflect unmet needs and disrupt... Read moreSurgical Techniques
view channel
3D Map of Heart Electrical Wiring Aims to Guide Congenital Heart Repair
Tetralogy of Fallot is one of the most common congenital heart problems and often requires surgery in infancy. Many survivors later develop conduction abnormalities because the cardiac electrical system... Read more
Small Cryoprobe Outperforms Forceps in Lung Biopsy Trial
Accurate diagnosis of pulmonary nodules, post-transplant allograft dysfunction, and diffuse parenchymal lung disease depends on high-quality tissue obtained during bronchoscopy. Conventional forceps biopsy... Read morePatient Care
view channel
AI Avatar Doctor Improves Patient Understanding Before Radiotherapy
Radiation oncology consultations require patients to grasp complex concepts quickly, yet anxiety and information overload often undermine understanding and informed consent. Poor comprehension can also... Read more
Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
Chronic obstructive pulmonary disease (COPD) is a long-term lung disorder that makes breathing difficult and often disturbs sleep, reducing energy for daily activities. Limited engagement in pulmonary... Read moreHealth IT
view channel
AI-Native EHR Achieves EU Medical Device Certification
InterSystems (Boston, MA, USA) announced that its IntelliCare electronic health record (EHR) solutions have been certified as Class IIa medical devices under the European Union Medical Device Regulation... Read more
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read morePoint of Care
view channel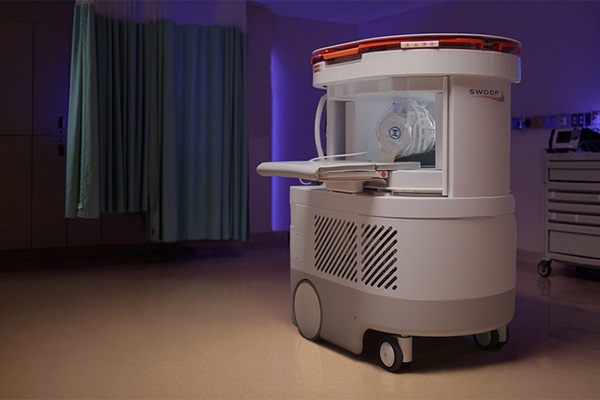
Portable MRI System Accelerates Emergency Brain Imaging and Triage
Emergency departments frequently face delays accessing conventional magnetic resonance imaging (MRI) for patients with suspected neurological emergencies. Such waits can slow triage, prolong boarding,... Read more