Vaginal Insert Offers Alternative Treatment for Fecal Incontinence 
|
By HospiMedica International staff writers Posted on 02 Feb 2016 |
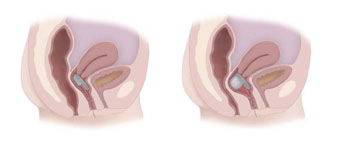
Image: The Eclipse vaginal insert in deflated (L) and Inflated (R) states (Phoyo courtesy of Pelvalon).
A novel vaginal insert for bowel control eliminates the need for surgery or an in-office procedure for the treatment of female fecal incontinence (FI).
The second-generation Eclipse System is comprised of a vaginal insert in the form of an inflatable balloon that is placed in the same location as a tampon or a diaphragm. The insert is composed of a base portion, an inflatable balloon portion, an inflation tube, and a self-closing Luer valve connection. The base portion positions the balloon and helps maintain the placement of the insert in the vagina. The balloon itself is made of thin walled silicone, with an enclosed, non-body contacting polyurethane liner to minimize air loss. The silicone inflation tube terminates in the self-closing Luer valve that connects to the patient pump.
The pump itself is fitted with a removable regulating valve that controls the amount of air introduced to the inflatable balloon. During inflation, the pump is squeezed seven to ten times to adequately fill the balloon, with any excess air vented out by the regulator. When inflated, the balloon exerts pressure through the vaginal wall, occluding the rectal area and providing immediate bowel control. The patient can inflate and deflate the device at home when needed, thus reducing the number of FI episodes and protecting from unwanted stool passage. The device is removed periodically for cleaning.
The insert is initially fitted using a sizing tool to assist with customizing insert size, and is inflated to the correct volume by a clinician to determine the correct regulator valve. A trial insert is available to allow patients to evaluate the therapy before deciding whether it works for them. The system is available in three base sizes and two balloon sizes, and is intended for women 18–75 years old who have had four or more FI episodes during a two-week period. The Eclipse System is a product of Pelvalon (Sunnyvale, CA, USA), and has been approved by the US Food and Drug Administration (FDA).
“With this FDA clearance for our next-generation Eclipse, we are excited to initiate the first phase of our commercial launch in select centers of excellence,” said Miles Rosen, CEO and co-founder of Pelvalon. “We believe that this early phase of partnership with thought and practice leaders in the field of pelvic floor disorders will ensure a smooth expansion down the road.”
“Eclipse is a nonsurgical therapy offering immediate bowel control that can be used early in the treatment pathway,” said Holly Richter, MD, director of the division of urogynecology and pelvic reconstructive surgery at the University of Alabama (Birmingham, USA). “Like the first-generation device, the next-generation Eclipse is an easily tried approach that fills a gap between lifestyle changes in diet and exercise and more invasive treatments. I'm excited that this new option will soon be available to my patients.”
FI is the inability to control bowel movements and is a common problem, especially among older adults. It affects women about twice as often as men, most probably due to childbirth, nerve or muscle damage in the pelvic region, or gastrointestinal disorders such as irritable bowel syndrome (IBS). First-line treatments include dietary changes, exercise, and medication; if the issue persists, patients may need to move on to more invasive and costly treatments, such as surgery, surgical implants, or injections.
Related Links:
Pelvalon
The second-generation Eclipse System is comprised of a vaginal insert in the form of an inflatable balloon that is placed in the same location as a tampon or a diaphragm. The insert is composed of a base portion, an inflatable balloon portion, an inflation tube, and a self-closing Luer valve connection. The base portion positions the balloon and helps maintain the placement of the insert in the vagina. The balloon itself is made of thin walled silicone, with an enclosed, non-body contacting polyurethane liner to minimize air loss. The silicone inflation tube terminates in the self-closing Luer valve that connects to the patient pump.
The pump itself is fitted with a removable regulating valve that controls the amount of air introduced to the inflatable balloon. During inflation, the pump is squeezed seven to ten times to adequately fill the balloon, with any excess air vented out by the regulator. When inflated, the balloon exerts pressure through the vaginal wall, occluding the rectal area and providing immediate bowel control. The patient can inflate and deflate the device at home when needed, thus reducing the number of FI episodes and protecting from unwanted stool passage. The device is removed periodically for cleaning.
The insert is initially fitted using a sizing tool to assist with customizing insert size, and is inflated to the correct volume by a clinician to determine the correct regulator valve. A trial insert is available to allow patients to evaluate the therapy before deciding whether it works for them. The system is available in three base sizes and two balloon sizes, and is intended for women 18–75 years old who have had four or more FI episodes during a two-week period. The Eclipse System is a product of Pelvalon (Sunnyvale, CA, USA), and has been approved by the US Food and Drug Administration (FDA).
“With this FDA clearance for our next-generation Eclipse, we are excited to initiate the first phase of our commercial launch in select centers of excellence,” said Miles Rosen, CEO and co-founder of Pelvalon. “We believe that this early phase of partnership with thought and practice leaders in the field of pelvic floor disorders will ensure a smooth expansion down the road.”
“Eclipse is a nonsurgical therapy offering immediate bowel control that can be used early in the treatment pathway,” said Holly Richter, MD, director of the division of urogynecology and pelvic reconstructive surgery at the University of Alabama (Birmingham, USA). “Like the first-generation device, the next-generation Eclipse is an easily tried approach that fills a gap between lifestyle changes in diet and exercise and more invasive treatments. I'm excited that this new option will soon be available to my patients.”
FI is the inability to control bowel movements and is a common problem, especially among older adults. It affects women about twice as often as men, most probably due to childbirth, nerve or muscle damage in the pelvic region, or gastrointestinal disorders such as irritable bowel syndrome (IBS). First-line treatments include dietary changes, exercise, and medication; if the issue persists, patients may need to move on to more invasive and costly treatments, such as surgery, surgical implants, or injections.
Related Links:
Pelvalon
Latest Patient Care News
- Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization

- Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
- Next Gen ICU Bed to Help Address Complex Critical Care Needs
- Groundbreaking AI-Powered UV-C Disinfection Technology Redefines Infection Control Landscape
- Clean Hospitals Can Reduce Antibiotic Resistance, Save Lives
- Smart Hospital Beds Improve Accuracy of Medical Diagnosis
- New Fast Endoscope Drying System Improves Productivity and Traceability
- World’s First Automated Endoscope Cleaner Fights Antimicrobial Resistance
- Portable High-Capacity Digital Stretcher Scales Provide Precision Weighing for Patients in ER
- Portable Clinical Scale with Remote Indicator Allows for Flexible Patient Weighing Use
- Innovative and Highly Customizable Medical Carts Offer Unlimited Configuration Possibilities
Channels
Artificial Intelligence
view channel
Machine Learning Approach Enhances Liver Cancer Risk Stratification
Hepatocellular carcinoma, the most common form of primary liver cancer, is often detected late despite targeted surveillance programs. Current screening guidelines emphasize patients with known cirrhosis,... Read more
New AI Approach Monitors Brain Health Using Passive Wearable Data
Brain health spans cognitive and emotional functions and can fluctuate even in adults without diagnosed disease. Detecting early changes remains difficult in routine care and burdens specialty services... Read moreCritical Care
view channel
Noninvasive Monitoring Device Enables Earlier Intervention in Heart Failure
Hospitalizations for heart failure with preserved ejection fraction (HFpEF) remain common because lung congestion often worsens before symptoms prompt treatment changes. Missed early decompensation... Read more
Automated IV Labeling Solution Improves Infusion Safety and Efficiency
Medication administration in high-acuity settings is often complicated by multiple concurrent infusions, making accurate line identification essential. In a 10-hospital intensive care unit study, 60% of... Read moreSurgical Techniques
view channel
Continuous Monitoring with Wearables Enhances Postoperative Patient Safety
Postoperative hypoxemia on general surgical wards is common and often missed by intermittent vital sign checks. Undetected low oxygen levels can delay recovery and raise the risk of complications that... Read more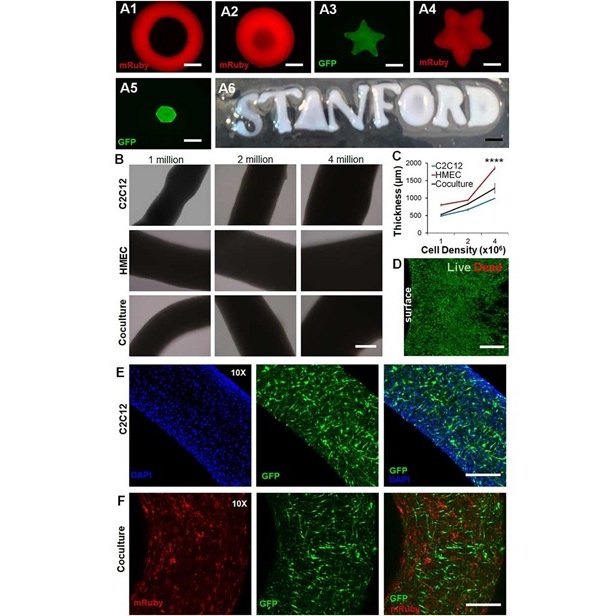
New Approach Enables Customized Muscle Tissue Without Biomaterial Scaffolds
Volumetric muscle loss is a traumatic loss of skeletal muscle that often leads to permanent functional impairment and limited reconstructive options. Current experimental strategies struggle to deliver... Read moreHealth IT
view channel
EMR-Based Tool Predicts Graft Failure After Kidney Transplant
Kidney transplantation offers patients with end-stage kidney disease longer survival and better quality of life than dialysis, yet graft failure remains a major challenge. Although a successful transplant... Read more
Printable Molecule-Selective Nanoparticles Enable Mass Production of Wearable Biosensors
The future of medicine is likely to focus on the personalization of healthcare—understanding exactly what an individual requires and delivering the appropriate combination of nutrients, metabolites, and... Read moreBusiness
view channel