HPV Vaccine Appears Safe in Early Pregnancy
|
By HospiMedica International staff writers Posted on 14 Apr 2017 |
A new study suggests that vaccination against human papillomavirus (HPV) during pregnancy does not increase the risk of maternal or fetal complications.
Researchers at the Danish Statens Serum Institut conducted a retrospective study of all women in Denmark who had a pregnancy that ended between October 1, 2006, and November 30, 2013. Using nationwide registers, the researchers linked data on quadrivalent HPV vaccination, adverse pregnancy outcomes, and potential confounders. Vaccinated women were matched on a 1:4 ratio with women who were not. Study outcomes included a range of gestational and newborn factors.
The results revealed 65 birth defects in vaccine-exposed pregnancies, compared to 220 in unexposed pregnancies; 20 spontaneous abortions with vaccine exposure versus 131 without; 116 preterm births versus 407; 76 cases of low birth weight versus 277; 171 cases of small size for gestational age versus 783; and two stillbirths versus four. Upon analysis, exposure to HPV vaccine was not associated with significantly higher risks for major birth defect, spontaneous abortion, preterm birth, low birth weight, small size for gestational age, or stillbirth. The study was published on March 30, 2017, in the New England Journal of Medicine (NEJM).
“Our results are consistent with other evidence that does not indicate that the vaccination of pregnant women with inactivated virus, bacterial, or toxoid vaccines generally confers a higher risk of adverse pregnancy outcomes than no such vaccination,” concluded lead author Nikolai Scheller, MD, and colleagues. “Our results also confirm and considerably expand on results from previous studies of the quadrivalent HPV vaccine.”
Introduced in 2006, the quadrivalent HPV vaccine covers the four viral strains most closely associated with gynecologic cancer (HPV-6, 11, 16, and 18). HPV types 6 and 11 also cause genital warts. As of 2014, 58 countries include HPV in their routine vaccinations for all girls around the ages of 9-13, and some countries vaccinate boys as well; the vaccines provide protection for at least eight years. Denmark also vaccinates women up to 26 years of age, and as a result some women are inadvertently exposed to HPV vaccination during early pregnancy.
Researchers at the Danish Statens Serum Institut conducted a retrospective study of all women in Denmark who had a pregnancy that ended between October 1, 2006, and November 30, 2013. Using nationwide registers, the researchers linked data on quadrivalent HPV vaccination, adverse pregnancy outcomes, and potential confounders. Vaccinated women were matched on a 1:4 ratio with women who were not. Study outcomes included a range of gestational and newborn factors.
The results revealed 65 birth defects in vaccine-exposed pregnancies, compared to 220 in unexposed pregnancies; 20 spontaneous abortions with vaccine exposure versus 131 without; 116 preterm births versus 407; 76 cases of low birth weight versus 277; 171 cases of small size for gestational age versus 783; and two stillbirths versus four. Upon analysis, exposure to HPV vaccine was not associated with significantly higher risks for major birth defect, spontaneous abortion, preterm birth, low birth weight, small size for gestational age, or stillbirth. The study was published on March 30, 2017, in the New England Journal of Medicine (NEJM).
“Our results are consistent with other evidence that does not indicate that the vaccination of pregnant women with inactivated virus, bacterial, or toxoid vaccines generally confers a higher risk of adverse pregnancy outcomes than no such vaccination,” concluded lead author Nikolai Scheller, MD, and colleagues. “Our results also confirm and considerably expand on results from previous studies of the quadrivalent HPV vaccine.”
Introduced in 2006, the quadrivalent HPV vaccine covers the four viral strains most closely associated with gynecologic cancer (HPV-6, 11, 16, and 18). HPV types 6 and 11 also cause genital warts. As of 2014, 58 countries include HPV in their routine vaccinations for all girls around the ages of 9-13, and some countries vaccinate boys as well; the vaccines provide protection for at least eight years. Denmark also vaccinates women up to 26 years of age, and as a result some women are inadvertently exposed to HPV vaccination during early pregnancy.
Latest Patient Care News
- AI Avatar Doctor Improves Patient Understanding Before Radiotherapy
- Wearable Sleep Data Predict Adherence to Pulmonary Rehabilitation
- Revolutionary Automatic IV-Line Flushing Device to Enhance Infusion Care
- VR Training Tool Combats Contamination of Portable Medical Equipment
- Portable Biosensor Platform to Reduce Hospital-Acquired Infections
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization

- Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
- Next Gen ICU Bed to Help Address Complex Critical Care Needs
- Groundbreaking AI-Powered UV-C Disinfection Technology Redefines Infection Control Landscape
- Clean Hospitals Can Reduce Antibiotic Resistance, Save Lives
- Smart Hospital Beds Improve Accuracy of Medical Diagnosis
- New Fast Endoscope Drying System Improves Productivity and Traceability
- World’s First Automated Endoscope Cleaner Fights Antimicrobial Resistance
- Portable High-Capacity Digital Stretcher Scales Provide Precision Weighing for Patients in ER
- Portable Clinical Scale with Remote Indicator Allows for Flexible Patient Weighing Use
Channels
Artificial Intelligence
view channel
AI Platform Supports Noninvasive Remote Hemodynamic Monitoring in Heart Failure
Heart failure remains a leading cause of hospitalization in adults over 65, affecting more than 6.7 million people in the U.S. Clinicians often lose visibility into hemodynamic deterioration once patients... Read more
AI Tool Predicts Unplanned Care and Symptom Burden in Cancer Survivors
Unplanned emergency visits and hospitalizations remain common in cancer survivorship, when routine clinical contact often tapers while new symptoms emerge. These events reflect unmet needs and disrupt... Read moreCritical Care
view channel
Bedside CSF Monitor Detects Early Infection in Fluid Drains
External drainage of cerebrospinal fluid (CSF) after traumatic brain injury, hydrocephalus, or hemorrhage carries a significant risk of infection. These infections can prolong intensive care, cause severe... Read more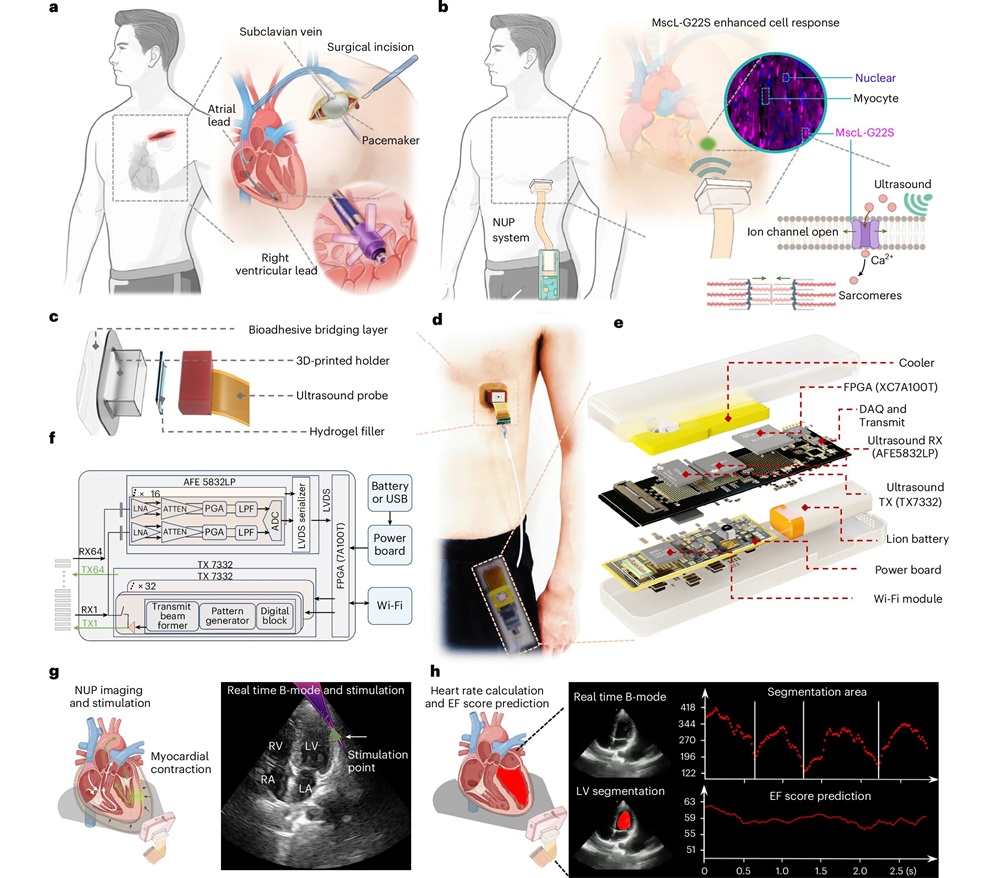
Wearable Ultrasound Patch Noninvasively Paces Heart to Stabilize Arrhythmias
Cardiac arrhythmias, including slow and irregular heart rhythms, often require pacemakers that are surgically implanted. While effective, implants carry procedural risks and long-term device maintenance burdens.... Read more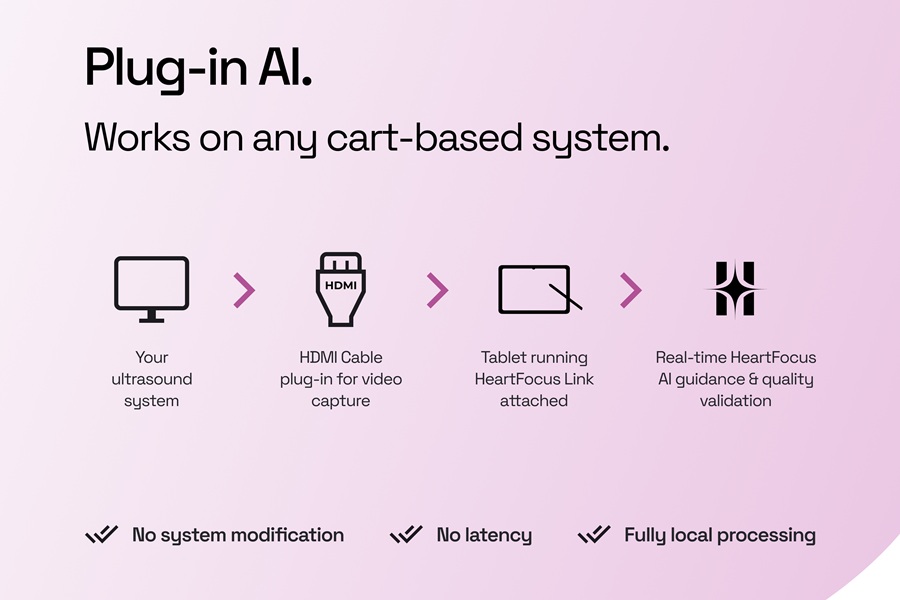
AI-Guided System Supports Cardiac Ultrasound Training on Cart-Based Systems
Cardiac ultrasound adoption in frontline settings often lags despite widespread investment in ultrasound equipment. Many systems sit underused for cardiac imaging because acquiring standard views requires... Read more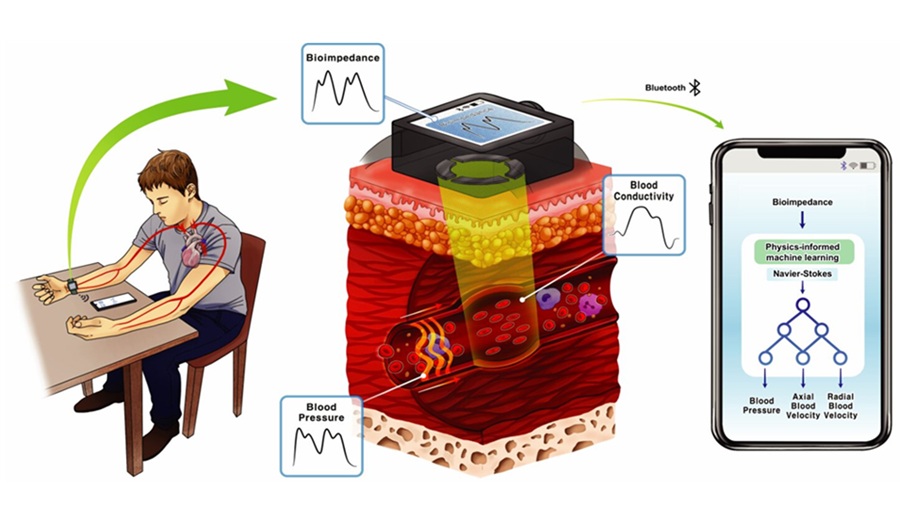
Cuffless Wearable Enables Continuous Blood Pressure Monitoring for Hypertension Care
Hypertension, or chronically elevated blood pressure, drives major risks for heart attack and stroke yet is typically assessed with intermittent cuff readings. These snapshots miss rapid physiologic changes... Read moreSurgical Techniques
view channel
3D Map of Heart Electrical Wiring Aims to Guide Congenital Heart Repair
Tetralogy of Fallot is one of the most common congenital heart problems and often requires surgery in infancy. Many survivors later develop conduction abnormalities because the cardiac electrical system... Read more
Small Cryoprobe Outperforms Forceps in Lung Biopsy Trial
Accurate diagnosis of pulmonary nodules, post-transplant allograft dysfunction, and diffuse parenchymal lung disease depends on high-quality tissue obtained during bronchoscopy. Conventional forceps biopsy... Read moreHealth IT
view channel
AI-Native EHR Achieves EU Medical Device Certification
InterSystems (Boston, MA, USA) announced that its IntelliCare electronic health record (EHR) solutions have been certified as Class IIa medical devices under the European Union Medical Device Regulation... Read more
EHR-Integrated Screening Workflow Detects Cognitive Impairment at Admission
Cognitive impairment involves difficulties with thinking, learning, memory, and decision-making, and is more common in older adults. In U.S. hospitals, more than 40% of admitted older adults have dementia,... Read morePoint of Care
view channel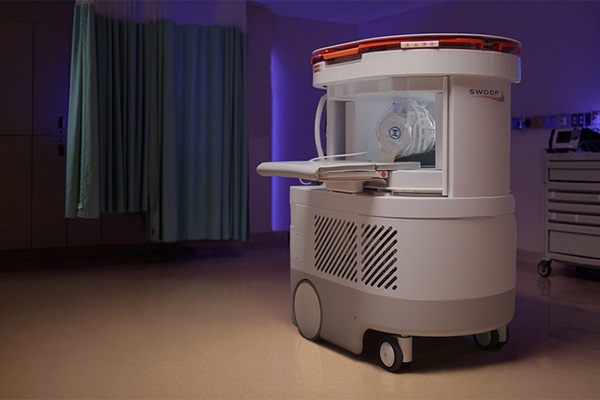
Portable MRI System Accelerates Emergency Brain Imaging and Triage
Emergency departments frequently face delays accessing conventional magnetic resonance imaging (MRI) for patients with suspected neurological emergencies. Such waits can slow triage, prolong boarding,... Read more