First-Of-Its-Kind Microchannel Drug Delivery Technology to Enable Self-Administrable COVID-19 Vaccines
|
By HospiMedica International staff writers Posted on 20 Aug 2020 |
Illustration
A first-of-its-kind microchannel drug delivery technology could enable self-administrable COVID-19 vaccines, enhancing their potential.
The drug delivery technology has been developed by Aquavit Pharmaceuticals, Inc. (New York, NY, USA) which is working on several innovative products and platform technology IP for personalized medicine. Aquavit has filed its latest patent for its novel drug delivery device through the COVID-19 Prioritized Examination Pilot Program (PEPP) which has been rolled out by the United States Patent and Trademark Office (USPTO) in light of the COVID-19 outbreak to facilitate moving patents for medical devices and services designed to combat the virus to market as soon as possible.
Currently, the majority of existing commercial vaccines are delivered to intramuscular or subcutaneous tissues by needle and syringe. However, in the past few years, there has been a renewed interest in the exploitation of skin vaccination in pursuit of a more potent immune response considering that the skin but not the muscle contains large amounts of antigen-presenting cells (APCs). The major challenge posed by targeting vaccines to the epidermis is to accurately and efficiently access the target tissue. Although the epidermis is superficially located and has a large surface area, access is restricted by the overlying stratum corneum, which forms a physical barrier to molecules > 500 Da molecular weight. Additionally, the epidermal layer is too thin to be reliably accessed by traditional needle and syringe injection.
Aquavit is developing a single-use epidermal drug delivery device that aids in the method of self-administration of biologics, drugs, vaccines, etc. using a microchannel delivery apparatus. The needle features a patented screw thread design that allows the delivery of biologics and vaccines to the epidermis/papillary dermis. Unlike syringe needles and microneedles, the microchannel systems are painless, user-friendly and require a low learning curve.
Aquavit's COVID-19 vaccine device patent covers new devices, compositions and methods that enable the administration of therapeutic vaccine compositions using microchannel delivery into these hard to reach epidermal and dermal layers of the skin rich in Antigen Presenting Cells (APCs) for promoting effective immune responses. The methods and devices are particularly useful during times of epidemic and pandemic crises, such as the current COVID-19 pandemic, when there is no or limited access to health care providers and healthcare systems are strained or limited. Being able to self-administer or have the administration performed by a non-health care provider also reduces the risk that the patient may become infected by visiting a hospital or health care provider where there are many infected patients.
"Multiple studies have proven that intradermal delivery is more effective and cost-efficient than intramuscular delivery. We believe our technology can potentially achieve higher immunogenicity than traditional needle methods, while also having higher patient compliance and accessibility," said Sai Sankar, Aquavit's Chief Biomedical Engineer.
"With the USPTO's fast track program, Aquavit is one step closer to manufacture and commercialize this novel drug delivery device that enhances the potential of any COVID-19 vaccine," said Sobin Chang, MPH, Chief Executive Officer. "As many pharmaceutical companies rush to develop a COVID-19 vaccine, there is an urgent need for integral components and delivery technology. Our universal platform technology can potentially accommodate any vaccines to be self-administered and pandemic distribution ready. We are currently in talks with multiple global scale pharmaceutical companies, vaccine makers, and major universities for collaborations and partnerships."
Related Links:
Aquavit Pharmaceuticals, Inc.
The drug delivery technology has been developed by Aquavit Pharmaceuticals, Inc. (New York, NY, USA) which is working on several innovative products and platform technology IP for personalized medicine. Aquavit has filed its latest patent for its novel drug delivery device through the COVID-19 Prioritized Examination Pilot Program (PEPP) which has been rolled out by the United States Patent and Trademark Office (USPTO) in light of the COVID-19 outbreak to facilitate moving patents for medical devices and services designed to combat the virus to market as soon as possible.
Currently, the majority of existing commercial vaccines are delivered to intramuscular or subcutaneous tissues by needle and syringe. However, in the past few years, there has been a renewed interest in the exploitation of skin vaccination in pursuit of a more potent immune response considering that the skin but not the muscle contains large amounts of antigen-presenting cells (APCs). The major challenge posed by targeting vaccines to the epidermis is to accurately and efficiently access the target tissue. Although the epidermis is superficially located and has a large surface area, access is restricted by the overlying stratum corneum, which forms a physical barrier to molecules > 500 Da molecular weight. Additionally, the epidermal layer is too thin to be reliably accessed by traditional needle and syringe injection.
Aquavit is developing a single-use epidermal drug delivery device that aids in the method of self-administration of biologics, drugs, vaccines, etc. using a microchannel delivery apparatus. The needle features a patented screw thread design that allows the delivery of biologics and vaccines to the epidermis/papillary dermis. Unlike syringe needles and microneedles, the microchannel systems are painless, user-friendly and require a low learning curve.
Aquavit's COVID-19 vaccine device patent covers new devices, compositions and methods that enable the administration of therapeutic vaccine compositions using microchannel delivery into these hard to reach epidermal and dermal layers of the skin rich in Antigen Presenting Cells (APCs) for promoting effective immune responses. The methods and devices are particularly useful during times of epidemic and pandemic crises, such as the current COVID-19 pandemic, when there is no or limited access to health care providers and healthcare systems are strained or limited. Being able to self-administer or have the administration performed by a non-health care provider also reduces the risk that the patient may become infected by visiting a hospital or health care provider where there are many infected patients.
"Multiple studies have proven that intradermal delivery is more effective and cost-efficient than intramuscular delivery. We believe our technology can potentially achieve higher immunogenicity than traditional needle methods, while also having higher patient compliance and accessibility," said Sai Sankar, Aquavit's Chief Biomedical Engineer.
"With the USPTO's fast track program, Aquavit is one step closer to manufacture and commercialize this novel drug delivery device that enhances the potential of any COVID-19 vaccine," said Sobin Chang, MPH, Chief Executive Officer. "As many pharmaceutical companies rush to develop a COVID-19 vaccine, there is an urgent need for integral components and delivery technology. Our universal platform technology can potentially accommodate any vaccines to be self-administered and pandemic distribution ready. We are currently in talks with multiple global scale pharmaceutical companies, vaccine makers, and major universities for collaborations and partnerships."
Related Links:
Aquavit Pharmaceuticals, Inc.
Latest COVID-19 News
- Low-Cost System Detects SARS-CoV-2 Virus in Hospital Air Using High-Tech Bubbles
- World's First Inhalable COVID-19 Vaccine Approved in China
- COVID-19 Vaccine Patch Fights SARS-CoV-2 Variants Better than Needles
- Blood Viscosity Testing Can Predict Risk of Death in Hospitalized COVID-19 Patients
- ‘Covid Computer’ Uses AI to Detect COVID-19 from Chest CT Scans
- MRI Lung-Imaging Technique Shows Cause of Long-COVID Symptoms
- Chest CT Scans of COVID-19 Patients Could Help Distinguish Between SARS-CoV-2 Variants
- Specialized MRI Detects Lung Abnormalities in Non-Hospitalized Long COVID Patients
- AI Algorithm Identifies Hospitalized Patients at Highest Risk of Dying From COVID-19
- Sweat Sensor Detects Key Biomarkers That Provide Early Warning of COVID-19 and Flu
- Study Assesses Impact of COVID-19 on Ventilation/Perfusion Scintigraphy
- CT Imaging Study Finds Vaccination Reduces Risk of COVID-19 Associated Pulmonary Embolism
- Third Day in Hospital a ‘Tipping Point’ in Severity of COVID-19 Pneumonia
- Longer Interval Between COVID-19 Vaccines Generates Up to Nine Times as Many Antibodies
- AI Model for Monitoring COVID-19 Predicts Mortality Within First 30 Days of Admission
- AI Predicts COVID Prognosis at Near-Expert Level Based Off CT Scans
Channels
Critical Care
view channel_1.jpg)
Cutting-Edge Bioelectronic Device Offers Drug-Free Approach to Managing Bacterial Infections
Antibiotic-resistant infections pose an increasing threat to patient safety and healthcare systems worldwide. Recent estimates indicate that drug-resistant infections may rise by 70% by 2050, highlighting... Read more
Sophisticated Machine-Learning Approach Uses Patient EHRs to Predict Pneumonia Outcomes
Pneumonia, an infection that results in difficulty breathing due to fluid accumulation in the lungs, is one of the leading causes of death worldwide. This condition is particularly challenging to treat... Read more
Early TAVR Benefits Patients with Asymptomatic Severe Aortic Stenosis
For patients with asymptomatic severe aortic stenosis (AS) and preserved left-ventricular ejection fraction, current guidelines recommend clinical surveillance every six to twelve months.... Read more
First-Of-Its-Kind Experimental Therapy Enhances Tissue Repair After Heart Attack
Cardiovascular disease remains the leading cause of death worldwide, accounting for one-third of all annual fatalities. Following a heart attack, the heart's natural regenerative ability is limited, resulting... Read moreSurgical Techniques
view channel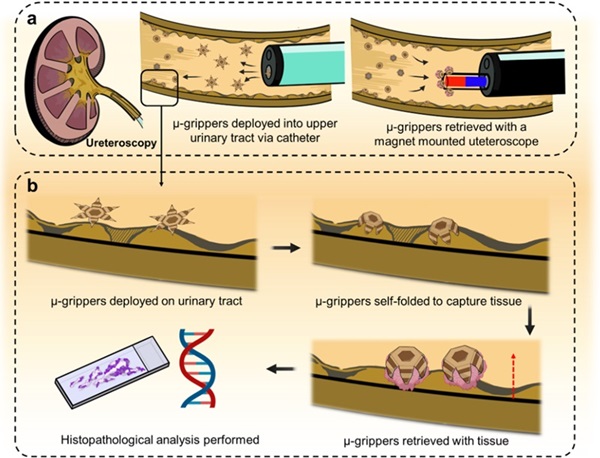
Microgrippers For Miniature Biopsies to Create New Cancer Diagnostic Screening Paradigm
The standard diagnosis of upper urinary tract cancers typically involves the removal of suspicious tissue using forceps, a procedure that is technically challenging and samples only a single region of the organ.... Read moreMiniature Soft Lithium-Ion Battery Could Be Used as Defibrillator During Surgery
The development of tiny smart devices, measuring just a few cubic millimeters, requires equally miniature power sources. For minimally invasive biomedical devices that interact with biological tissues,... Read more.jpg)
TAVI Procedure Supported by Radial Artery Access Reduces Bleeding Complications
The TAVI procedure, or Transcatheter Aortic Valve Implantation, is a minimally invasive technique in which a new aortic valve is inserted through a femoral artery to replace a narrowed old valve.... Read more
Portable Surgical Robot Seamlessly Integrates into Any OR for Performing Cholecystectomy Procedures
The United States represents a significant market with a strong demand for a flexible surgical robotic system applicable across various care settings. However, there has been a lack of sufficient options... Read morePatient Care
view channel
Portable Biosensor Platform to Reduce Hospital-Acquired Infections
Approximately 4 million patients in the European Union acquire healthcare-associated infections (HAIs) or nosocomial infections each year, with around 37,000 deaths directly resulting from these infections,... Read moreFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel
Handheld, Sound-Based Diagnostic System Delivers Bedside Blood Test Results in An Hour
Patients who go to a doctor for a blood test often have to contend with a needle and syringe, followed by a long wait—sometimes hours or even days—for lab results. Scientists have been working hard to... Read more
Smartphone-Enabled, Paper-Based Quantitative Diagnostic Platform Transforms POC Testing
Point-of-care diagnostics are crucial for public health, offering rapid, on-site testing that enables prompt diagnosis and treatment. This is especially valuable in remote or underserved regions where... Read moreBusiness
view channel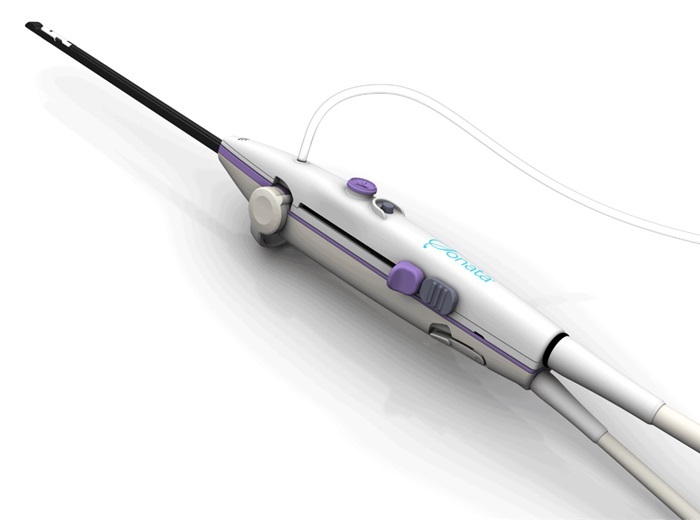
Hologic Acquires Gynesonics to Strengthen Existing Gynecological Surgical Business
Hologic (Marlborough, MA, USA) has signed a definitive agreement to acquire Gynesonics (Redwood City, CA, USA) for approximately USD 350 million, subject to working capital and other customary closing adjustments.... Read more