World’s First COVID-19 Drug Based on Human Immunoglobulin Developed from Plasma of Recovered Patients
|
By HospiMedica International staff writers Posted on 14 Sep 2021 |

Illustration
The world’s first drug based on human immunoglobulin being developed from plasma of recovered patients could be effective on COVID-19 patients with mild conditions.
China National Pharmaceutical Group Co., Ltd. (Sinopharm Beijing, China) is developing two medicines for the treatment of COVID-19 based on human immunoglobulin and monoclonal antibody. Both the drugs could reduce the impact of the novel coronavirus in infected people and will be effective on patients with mild COVID-19, according to a report by the Global Times.
The first drug being developed from plasma of recovered COVID-19 patients or vaccinated uninfected people contains high-potency SARS-COV-2 neutralizing antibodies. The drug has demonstrated significant relief of symptoms and damage caused by SARS-COV-2 in pre-clinical studies and animal tests. The new drug containing a high level of neutralizing antibodies to the novel coronavirus is all set to soon enter Phase 3 clinical trials.
The second COVID-19 drug under development is based on a potent monoclonal antibody against the Delta variant discovered by researchers at Sinopharm. The monoclonal antibody is capable of effectively blocking the binding of the novel coronavirus to the Angiotensin-converting enzyme 2 which is attached to the membranes of cells located in the intestines, kidney, testes, gallbladder and heart. According to Sinopharm, the antibody can prevent the SARS-COV-2 virus from infecting human cells.
Related Links:
China National Pharmaceutical Group Co., Ltd.
China National Pharmaceutical Group Co., Ltd. (Sinopharm Beijing, China) is developing two medicines for the treatment of COVID-19 based on human immunoglobulin and monoclonal antibody. Both the drugs could reduce the impact of the novel coronavirus in infected people and will be effective on patients with mild COVID-19, according to a report by the Global Times.
The first drug being developed from plasma of recovered COVID-19 patients or vaccinated uninfected people contains high-potency SARS-COV-2 neutralizing antibodies. The drug has demonstrated significant relief of symptoms and damage caused by SARS-COV-2 in pre-clinical studies and animal tests. The new drug containing a high level of neutralizing antibodies to the novel coronavirus is all set to soon enter Phase 3 clinical trials.
The second COVID-19 drug under development is based on a potent monoclonal antibody against the Delta variant discovered by researchers at Sinopharm. The monoclonal antibody is capable of effectively blocking the binding of the novel coronavirus to the Angiotensin-converting enzyme 2 which is attached to the membranes of cells located in the intestines, kidney, testes, gallbladder and heart. According to Sinopharm, the antibody can prevent the SARS-COV-2 virus from infecting human cells.
Related Links:
China National Pharmaceutical Group Co., Ltd.
Latest COVID-19 News
- Low-Cost System Detects SARS-CoV-2 Virus in Hospital Air Using High-Tech Bubbles
- World's First Inhalable COVID-19 Vaccine Approved in China
- COVID-19 Vaccine Patch Fights SARS-CoV-2 Variants Better than Needles
- Blood Viscosity Testing Can Predict Risk of Death in Hospitalized COVID-19 Patients
- ‘Covid Computer’ Uses AI to Detect COVID-19 from Chest CT Scans
- MRI Lung-Imaging Technique Shows Cause of Long-COVID Symptoms
- Chest CT Scans of COVID-19 Patients Could Help Distinguish Between SARS-CoV-2 Variants
- Specialized MRI Detects Lung Abnormalities in Non-Hospitalized Long COVID Patients
- AI Algorithm Identifies Hospitalized Patients at Highest Risk of Dying From COVID-19
- Sweat Sensor Detects Key Biomarkers That Provide Early Warning of COVID-19 and Flu
- Study Assesses Impact of COVID-19 on Ventilation/Perfusion Scintigraphy
- CT Imaging Study Finds Vaccination Reduces Risk of COVID-19 Associated Pulmonary Embolism
- Third Day in Hospital a ‘Tipping Point’ in Severity of COVID-19 Pneumonia
- Longer Interval Between COVID-19 Vaccines Generates Up to Nine Times as Many Antibodies
- AI Model for Monitoring COVID-19 Predicts Mortality Within First 30 Days of Admission
- AI Predicts COVID Prognosis at Near-Expert Level Based Off CT Scans
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreCritical Care
view channel
Stretchable Microneedles to Help In Accurate Tracking of Abnormalities and Identifying Rapid Treatment
The field of personalized medicine is transforming rapidly, with advancements like wearable devices and home testing kits making it increasingly easy to monitor a wide range of health metrics, from heart... Read more
Machine Learning Tool Identifies Rare, Undiagnosed Immune Disorders from Patient EHRs
Patients suffering from rare diseases often endure extensive delays in receiving accurate diagnoses and treatments, which can lead to unnecessary tests, worsening health, psychological strain, and significant... Read more
On-Skin Wearable Bioelectronic Device Paves Way for Intelligent Implants
A team of researchers at the University of Missouri (Columbia, MO, USA) has achieved a milestone in developing a state-of-the-art on-skin wearable bioelectronic device. This development comes from a lab... Read more
First-Of-Its-Kind Dissolvable Stent to Improve Outcomes for Patients with Severe PAD
Peripheral artery disease (PAD) affects millions and presents serious health risks, particularly its severe form, chronic limb-threatening ischemia (CLTI). CLTI develops when arteries are blocked by plaque,... Read moreSurgical Techniques
view channel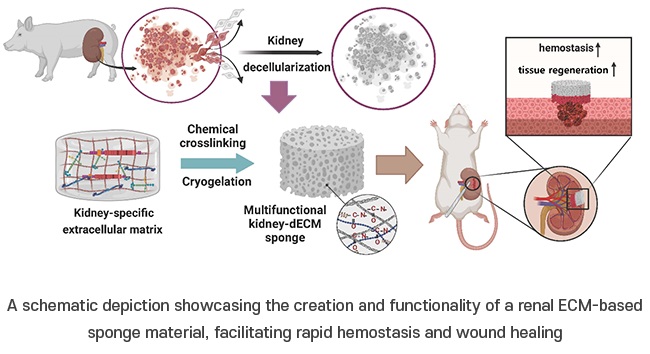
Porous Gel Sponge Facilitates Rapid Hemostasis and Wound Healing
The kidneys are essential organs that handle critical bodily functions, including waste elimination and blood pressure regulation. Often referred to as the silent organ because they typically do not manifest... Read more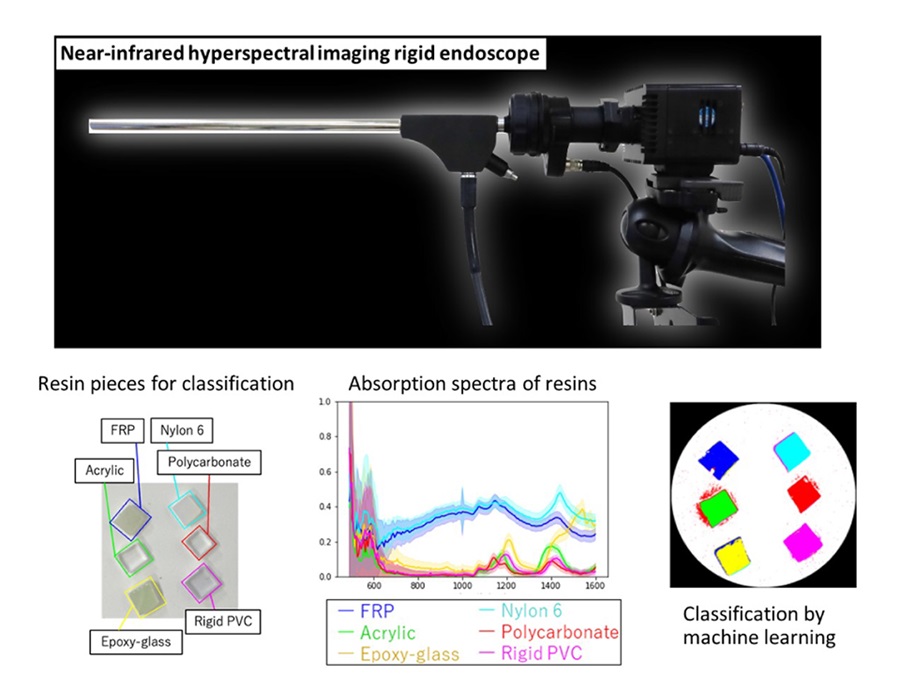
Novel Rigid Endoscope System Enables Deep Tissue Imaging During Surgery
Hyperspectral imaging (HSI) is an advanced technique that captures and processes information across a given electromagnetic spectrum. Near-infrared hyperspectral imaging (NIR-HSI) has particularly gained... Read more
Robotic Nerve ‘Cuffs’ Could Treat Various Neurological Conditions
Electric nerve implants serve dual functions: they can either stimulate or block signals in specific nerves. For example, they may alleviate pain by inhibiting pain signals or restore movement in paralyzed... Read more
Flexible Microdisplay Visualizes Brain Activity in Real-Time To Guide Neurosurgeons
During brain surgery, neurosurgeons need to identify and preserve regions responsible for critical functions while removing harmful tissue. Traditionally, neurosurgeons rely on a team of electrophysiologists,... Read morePatient Care
view channelFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel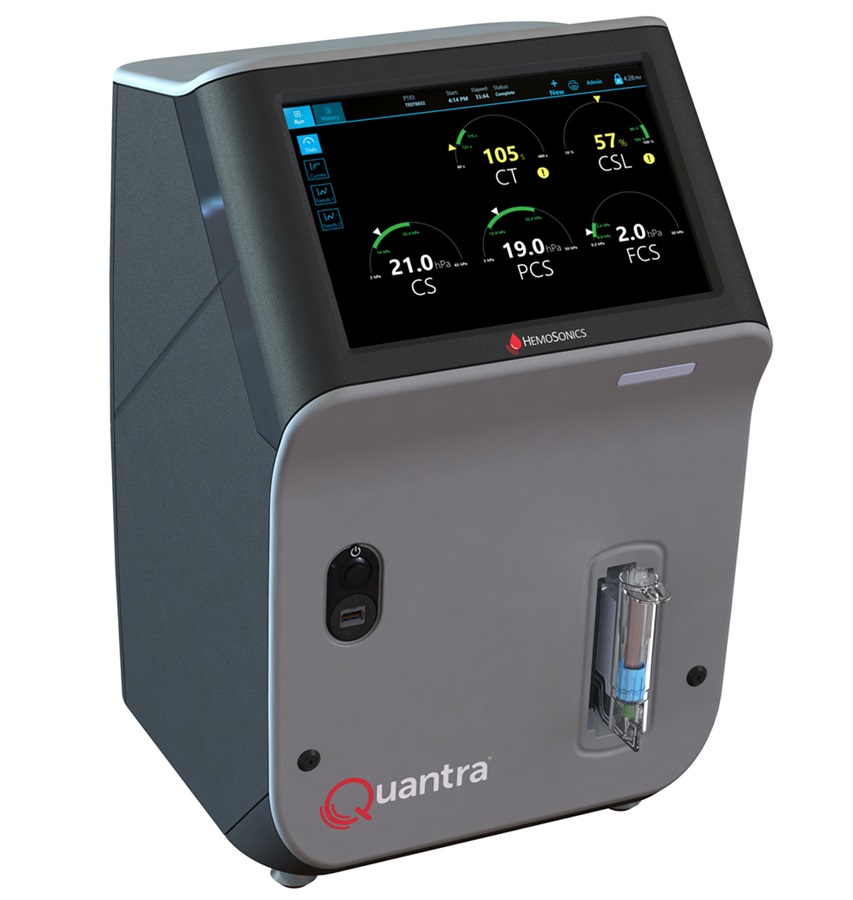
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more
New Generation Glucose Hospital Meter System Ensures Accurate, Interference-Free and Safe Use
A new generation glucose hospital meter system now comes with several features that make hospital glucose testing easier and more secure while continuing to offer accuracy, freedom from interference, and... Read moreBusiness
view channel
Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
Johnson & Johnson (New Brunswick, N.J., USA) and Shockwave Medical (Santa Clara, CA, USA) have entered into a definitive agreement under which Johnson & Johnson will acquire all of Shockwave’s... Read more