Nova's Stat Profile Prime Plus Blood Gas Analyzer Receives FDA Clearance for POC Use
|
By HospiMedica International staff writers Posted on 20 Apr 2020 |
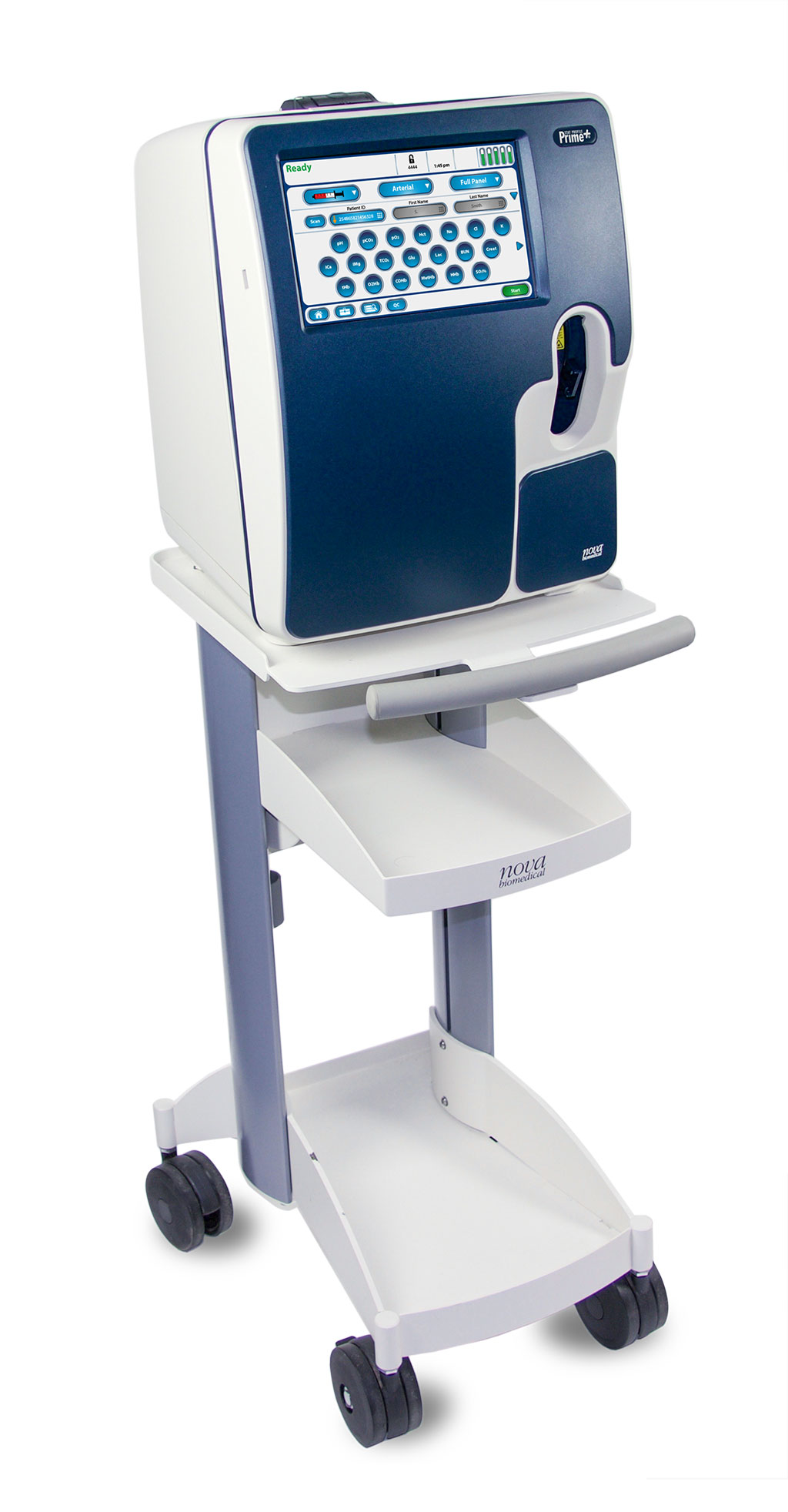
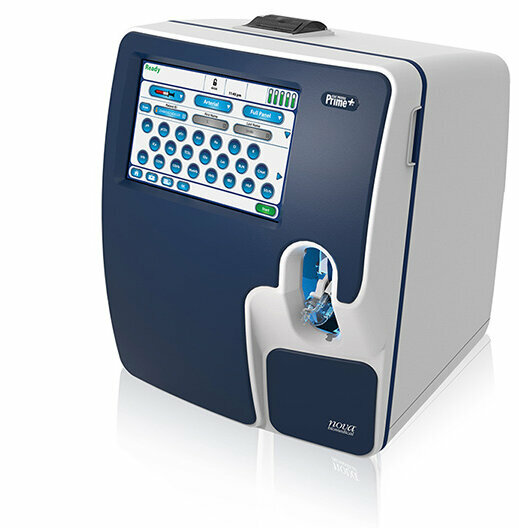
Image: Nova\'s Stat Profile Prime Plus critical care blood gas analyzer (Photo courtesy of Nova Biomedical)
Nova Biomedical (Waltham, MA, USA) has secured clearance from the US Food and Drug Administration (FDA) for its Stat Profile Prime Plus critical care blood gas analyzer for point-of-care (POC) use. The clearance allows POC personnel to perform bedside critical care testing with lab-quality results in as little as one minute. Prime Plus incorporates Nova’s maintenance-free sensor technology with individual MicroSensor Cards, calibrator cartridges, and quality control cartridges. This design eliminates sensor and co-oximeter maintenance, improves analyzer uptime, and reduces testing costs.
Prime Plus features maintenance-free sensor technology to provide 19 essential critical care tests including blood gases, electrolytes, metabolites, hematology, and co-oximetry, providing clinicians with a rapid, detailed diagnostic profile, including a comprehensive metabolic panel. The Prime Plus MicroSensor Card™ provides a critical care panel, which includes pH, PCO2, PO2, SO2%, Na, K, Cl, iCa, iMg, glucose, lactate, urea, creatinine, Hct, Hb, O2Hb, COHb, MetHb, and HHb and requires a small 60μl minimum sample volume. Prime Plus features patented, non-lysing whole blood co-oximetry technology, which eliminates corrosive lysing reagents and mechanical complexity while improving instrument reliability and reducing costs.
A unique safety sample port allows for easy docking and hands-free sampling, and the Clot Block™ sample flow path protects sensors from blood clot blockages and related downtime. Prime Plus provides real-time supplemental quality monitoring with automated QC that verifies analyzer accuracy and precision. The on-board assayed liquid controls meet or exceed all current regulatory guidelines and do not require the development of an Individualized Quality Control Plan (IQCP). Supplemental quality monitoring electronically tracks and verifies analytical performance between QC intervals and for each analysis and calibration.
Prime Plus features maintenance-free sensor technology to provide 19 essential critical care tests including blood gases, electrolytes, metabolites, hematology, and co-oximetry, providing clinicians with a rapid, detailed diagnostic profile, including a comprehensive metabolic panel. The Prime Plus MicroSensor Card™ provides a critical care panel, which includes pH, PCO2, PO2, SO2%, Na, K, Cl, iCa, iMg, glucose, lactate, urea, creatinine, Hct, Hb, O2Hb, COHb, MetHb, and HHb and requires a small 60μl minimum sample volume. Prime Plus features patented, non-lysing whole blood co-oximetry technology, which eliminates corrosive lysing reagents and mechanical complexity while improving instrument reliability and reducing costs.
A unique safety sample port allows for easy docking and hands-free sampling, and the Clot Block™ sample flow path protects sensors from blood clot blockages and related downtime. Prime Plus provides real-time supplemental quality monitoring with automated QC that verifies analyzer accuracy and precision. The on-board assayed liquid controls meet or exceed all current regulatory guidelines and do not require the development of an Individualized Quality Control Plan (IQCP). Supplemental quality monitoring electronically tracks and verifies analytical performance between QC intervals and for each analysis and calibration.
Latest Business News
- Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
- Mindray to Acquire Chinese Medical Device Company APT Medical
- Olympus Acquires Korean GI Stent Maker Taewoong Medical
- Karl Storz Acquires British AI Specialist Innersight Labs
- Stryker to Acquire French Joint Replacement Company SERF SAS
- Medical Illumination Acquires Surgical Lighting Specialist Isolux
- 5G Remote-Controlled Robots to Enable Even Cross-Border Surgeries

- International Hospital Federation Announces 2023 IHF Award Winners
- Unprecedented AI Integration Transforming Surgery Landscape, Say Experts

- New WHO Guidelines to Revolutionize AI in Healthcare
- Getinge Acquires US-Based Medical Equipment Provider Healthmark Industries
- Global Surgical Lights Market Driven by Increasing Number of Procedures
- Global Capsule Endoscopy Market Driven by Demand for Accurate Diagnosis of Gastrointestinal Conditions
- Global OR Integration Market Driven by Need for Improved Workflow Efficiency and Productivity
- Global Endoscopy Devices Market Driven by Increasing Adoption of Endoscopes in Surgical Procedures
- Global Minimally Invasive Medical Devices Market Driven by Benefits of MIS Procedures
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreCritical Care
view channel
Machine Learning Tool Identifies Rare, Undiagnosed Immune Disorders from Patient EHRs
Patients suffering from rare diseases often endure extensive delays in receiving accurate diagnoses and treatments, which can lead to unnecessary tests, worsening health, psychological strain, and significant... Read more
On-Skin Wearable Bioelectronic Device Paves Way for Intelligent Implants
A team of researchers at the University of Missouri (Columbia, MO, USA) has achieved a milestone in developing a state-of-the-art on-skin wearable bioelectronic device. This development comes from a lab... Read more
First-Of-Its-Kind Dissolvable Stent to Improve Outcomes for Patients with Severe PAD
Peripheral artery disease (PAD) affects millions and presents serious health risks, particularly its severe form, chronic limb-threatening ischemia (CLTI). CLTI develops when arteries are blocked by plaque,... Read more
AI Brain-Age Estimation Technology Uses EEG Scans to Screen for Degenerative Diseases
As individuals age, so do their brains. Premature aging of the brain can lead to age-related conditions such as mild cognitive impairment, dementia, or Parkinson's disease. The ability to determine "brain... Read moreSurgical Techniques
view channel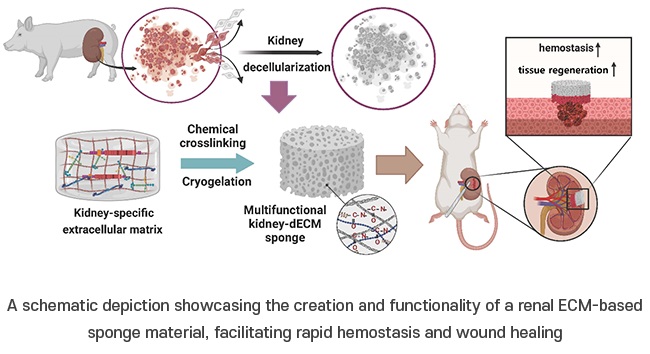
Porous Gel Sponge Facilitates Rapid Hemostasis and Wound Healing
The kidneys are essential organs that handle critical bodily functions, including waste elimination and blood pressure regulation. Often referred to as the silent organ because they typically do not manifest... Read more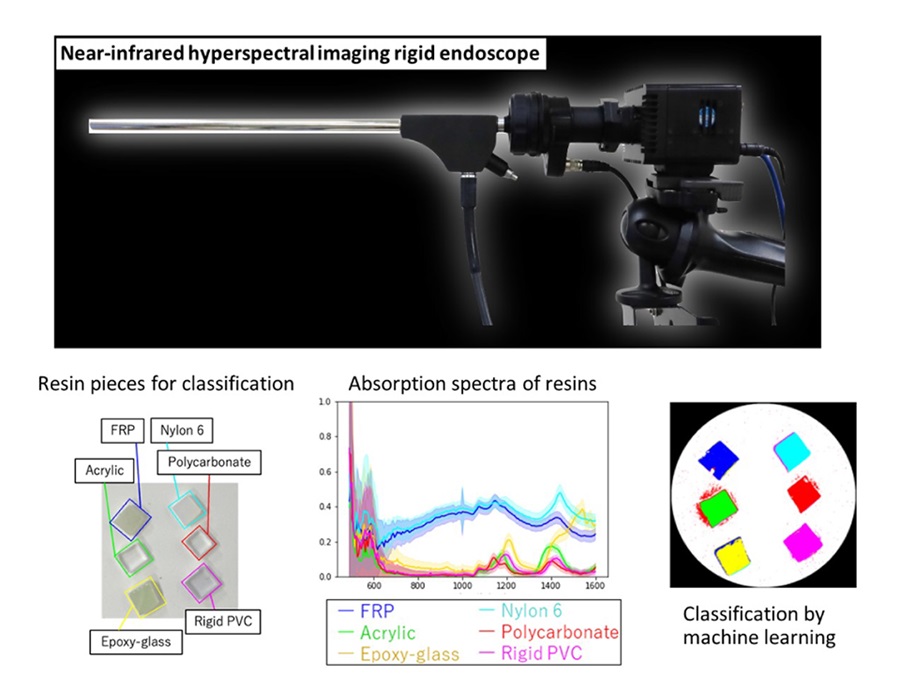
Novel Rigid Endoscope System Enables Deep Tissue Imaging During Surgery
Hyperspectral imaging (HSI) is an advanced technique that captures and processes information across a given electromagnetic spectrum. Near-infrared hyperspectral imaging (NIR-HSI) has particularly gained... Read more
Robotic Nerve ‘Cuffs’ Could Treat Various Neurological Conditions
Electric nerve implants serve dual functions: they can either stimulate or block signals in specific nerves. For example, they may alleviate pain by inhibiting pain signals or restore movement in paralyzed... Read more
Flexible Microdisplay Visualizes Brain Activity in Real-Time To Guide Neurosurgeons
During brain surgery, neurosurgeons need to identify and preserve regions responsible for critical functions while removing harmful tissue. Traditionally, neurosurgeons rely on a team of electrophysiologists,... Read morePatient Care
view channelFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more