Handheld Scanner Accurately Diagnoses Pressure Ulcers
|
By HospiMedica International staff writers Posted on 27 Nov 2013 |
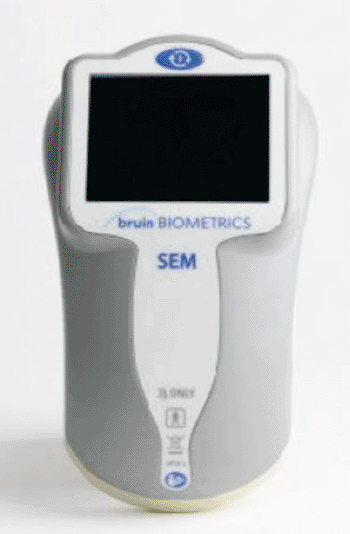
Image: The subepidermal moisture (SEM) scanner (Photo courtesy of Bruin Biometrics).
A novel scanner detects localized tissue edema and pressure induced tissue damage that may lead to the formation of decubitus ulcers, before the damage becomes visible.
The subepidermal moisture (SEM) scanner is a point of care diagnostic tool that uses electrical surface capacitance to assess increased fluid content in skin and underlying tissue, a biophysical marker of inflammation that indicates impending skin damage and pressure ulcer formation. The early detection capability can lead to targeted interventions, reduced ulceration severity, and early recovery, as well as decreased hospital days. Additional advantages of the device include objective, evidence-based measurements; noninvasive, rapid results; clear risk assessment documentation; and standardized patient assessment.
An enterprise version of the device will also include wireless syncing to a databank for rapid analytics and reporting to healthcare providers. In clinical studies, the SEM scanner has been shown to identify pressure-induced tissue damage up to 10 days before it becomes visible skin surface, and can help with the more difficult diagnosis of pressure ulcers in patients with a darker skin tone. The SEM scanner, which was developed by Bruin Biometrics (BBI; Los Angeles, CA, USA) in conjunction research colleagues at the University of California, Los Angeles (UCLA; USA), has received the European Community CE marking of approval.
Related Links:
Bruin Biometrics
University of California, Los Angeles
The subepidermal moisture (SEM) scanner is a point of care diagnostic tool that uses electrical surface capacitance to assess increased fluid content in skin and underlying tissue, a biophysical marker of inflammation that indicates impending skin damage and pressure ulcer formation. The early detection capability can lead to targeted interventions, reduced ulceration severity, and early recovery, as well as decreased hospital days. Additional advantages of the device include objective, evidence-based measurements; noninvasive, rapid results; clear risk assessment documentation; and standardized patient assessment.
An enterprise version of the device will also include wireless syncing to a databank for rapid analytics and reporting to healthcare providers. In clinical studies, the SEM scanner has been shown to identify pressure-induced tissue damage up to 10 days before it becomes visible skin surface, and can help with the more difficult diagnosis of pressure ulcers in patients with a darker skin tone. The SEM scanner, which was developed by Bruin Biometrics (BBI; Los Angeles, CA, USA) in conjunction research colleagues at the University of California, Los Angeles (UCLA; USA), has received the European Community CE marking of approval.
Related Links:
Bruin Biometrics
University of California, Los Angeles
Latest Patient Care News
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization

- Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
- Next Gen ICU Bed to Help Address Complex Critical Care Needs
- Groundbreaking AI-Powered UV-C Disinfection Technology Redefines Infection Control Landscape
- Clean Hospitals Can Reduce Antibiotic Resistance, Save Lives
- Smart Hospital Beds Improve Accuracy of Medical Diagnosis
- New Fast Endoscope Drying System Improves Productivity and Traceability
- World’s First Automated Endoscope Cleaner Fights Antimicrobial Resistance
- Portable High-Capacity Digital Stretcher Scales Provide Precision Weighing for Patients in ER
- Portable Clinical Scale with Remote Indicator Allows for Flexible Patient Weighing Use
- Innovative and Highly Customizable Medical Carts Offer Unlimited Configuration Possibilities
- Biomolecular Wound Healing Film Adheres to Sensitive Tissue and Releases Active Ingredients
- Wearable Health Tech Could Measure Gases Released From Skin to Monitor Metabolic Diseases
- Wearable Cardioverter Defibrillator System Protects Patients at Risk of Sudden Cardiac Arrest
- World's First AI-Ready Infrasound Stethoscope Listens to Bodily Sounds Not Audible to Human Ear
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreSurgical Techniques
view channel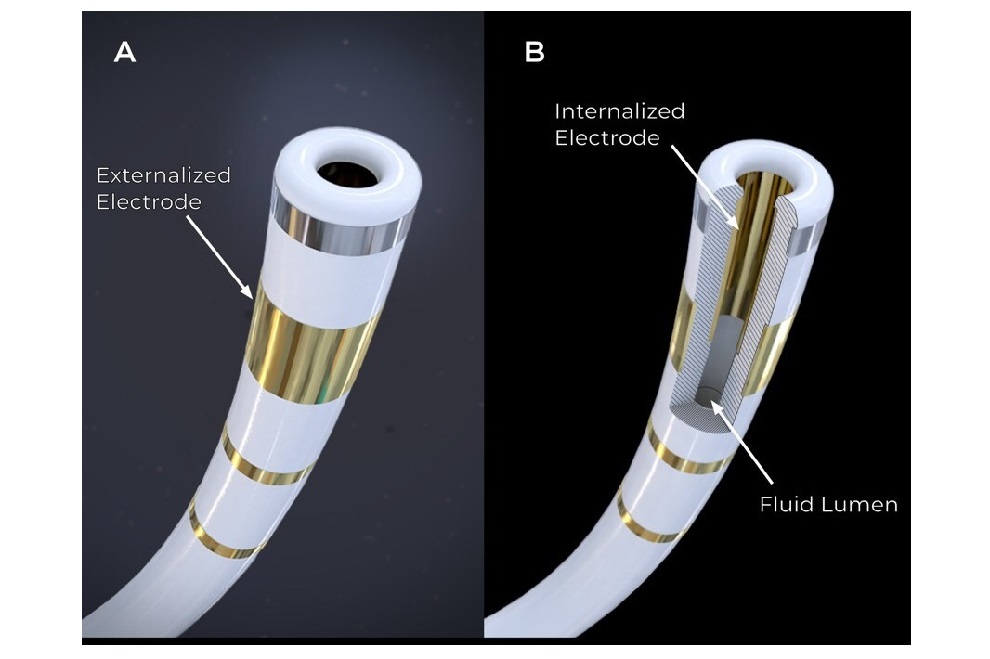
First-Ever Contact Force Pulsed Field Ablation System to Transform Treatment of Ventricular Arrhythmias
It is estimated that over 6 million patients in the US and Europe are affected by ventricular arrhythmias, which include conditions such as ventricular tachycardia (VT) and premature ventricular contractions (PVCs).... Read more
Caterpillar Robot with Built-In Steering System Crawls Easily Through Loops and Bends
Soft robots often face challenges in being guided effectively because adding steering mechanisms typically reduces their flexibility by increasing rigidity. Now, a team of engineers has combined ancient... Read more
Tiny Wraparound Electronic Implants to Revolutionize Treatment of Spinal Cord Injuries
The spinal cord functions as a vital conduit, transmitting nerve impulses to and from the brain, much like a highway. When the spinal cord is damaged, this flow of information is disrupted, leading to... Read morePatient Care
view channelFirst-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
Reducing healthcare-acquired infections (HAIs) remains a pressing issue within global healthcare systems. In the United States alone, 1.7 million patients contract HAIs annually, leading to approximately... Read more
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel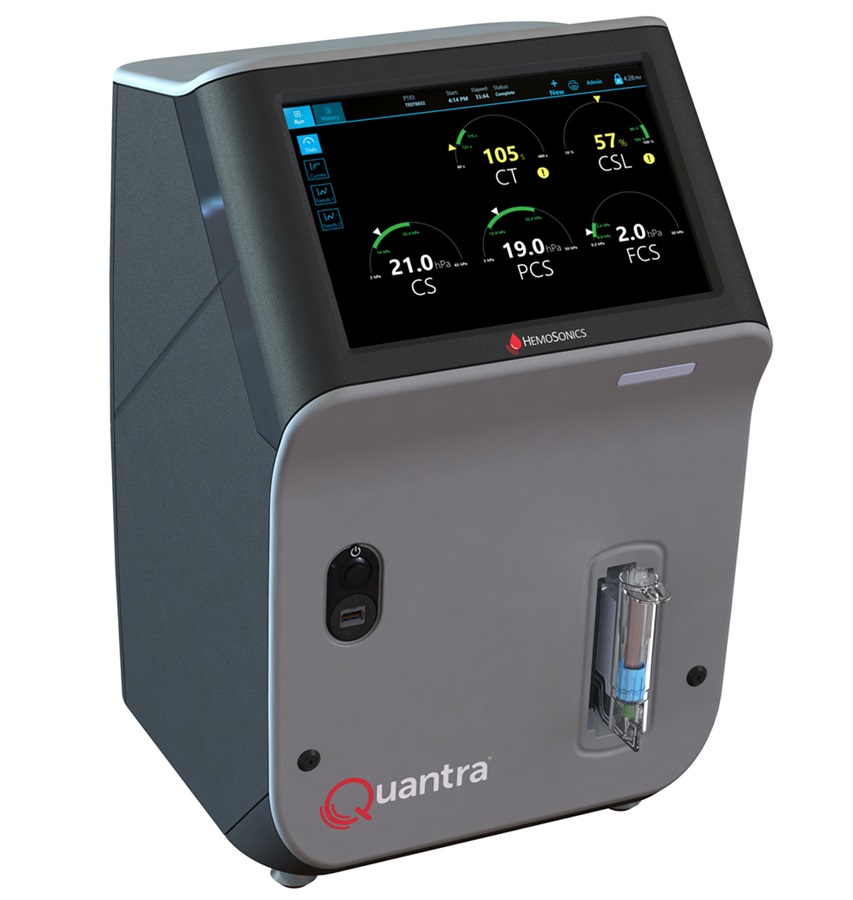
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more
New Generation Glucose Hospital Meter System Ensures Accurate, Interference-Free and Safe Use
A new generation glucose hospital meter system now comes with several features that make hospital glucose testing easier and more secure while continuing to offer accuracy, freedom from interference, and... Read moreBusiness
view channel
Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
Johnson & Johnson (New Brunswick, N.J., USA) and Shockwave Medical (Santa Clara, CA, USA) have entered into a definitive agreement under which Johnson & Johnson will acquire all of Shockwave’s... Read more