Medtronic Buys Tyrx Antibacterial Envelope Manufacturer
|
By HospiMedica International staff writers Posted on 13 Jan 2014 |
Startup company Tyrx (Monmouth Junction, NJ, USA), which manufactures bioresorbable envelopes for implanted medical devices, is now a part of Medtronic (Minneapolis, MN, USA), the world's largest medical device company.
The acquisition supports Medtronic's expansion of its medical device product offerings to include broader healthcare services and solutions, providing meaningful clinical outcomes and economic value for hospitals, physicians, patients, and payers. TYRX brings to Medtronic the AIGISRx R fully resorbable antibacterial envelope, designed to reduce surgical site infections associated with cardiac implantable electronic devices (CIEDs), and the AIGISRx N antibacterial envelope, used with spinal cord neurostimulators.
The bioresorbable envelopes use a combination of two antibiotic drugs: Minocycline and Rifampin. Minocycline is a tetracycline-class antibiotic commonly used to treat skin acne, among other things; Rifampin is commonly used to treat tuberculosis. Both drugs have different modes of action, and working together target roughly 80% of the germs, which can be introduced during surgery, including Methicillin Resistant Staphylococcus aureus (MRSA) and a number of other pathogens.
“While the risk of infection from an implanted pacemaker or defibrillator is low for most patients, repeated operative procedures after the initial device implant are associated with a substantial incremental risk of infection. This is estimated to cost the US healthcare system more than one billion dollars per year,” said Pat Mackin, president of the cardiac rhythm disease management business and senior vice president at Medtronic. “TYRX has developed an innovative, proven technology to reduce infection risk, making the procedure safer for patients and removing significant costs from the healthcare system.”
“We look forward to joining Medtronic as part of a combined portfolio that can positively impact outcomes for patients by reducing implant-related infections, and bring value to our customers,” said Robert White, president and CEO of Tyrx.
The all-cash transaction includes an initial payment of USD 160 million plus potential earn out and performance based milestone payments.
Related Links:
Tyrx
Medtronic
Antibacterial Envelope Protects Cardiac Implants
The acquisition supports Medtronic's expansion of its medical device product offerings to include broader healthcare services and solutions, providing meaningful clinical outcomes and economic value for hospitals, physicians, patients, and payers. TYRX brings to Medtronic the AIGISRx R fully resorbable antibacterial envelope, designed to reduce surgical site infections associated with cardiac implantable electronic devices (CIEDs), and the AIGISRx N antibacterial envelope, used with spinal cord neurostimulators.
The bioresorbable envelopes use a combination of two antibiotic drugs: Minocycline and Rifampin. Minocycline is a tetracycline-class antibiotic commonly used to treat skin acne, among other things; Rifampin is commonly used to treat tuberculosis. Both drugs have different modes of action, and working together target roughly 80% of the germs, which can be introduced during surgery, including Methicillin Resistant Staphylococcus aureus (MRSA) and a number of other pathogens.
“While the risk of infection from an implanted pacemaker or defibrillator is low for most patients, repeated operative procedures after the initial device implant are associated with a substantial incremental risk of infection. This is estimated to cost the US healthcare system more than one billion dollars per year,” said Pat Mackin, president of the cardiac rhythm disease management business and senior vice president at Medtronic. “TYRX has developed an innovative, proven technology to reduce infection risk, making the procedure safer for patients and removing significant costs from the healthcare system.”
“We look forward to joining Medtronic as part of a combined portfolio that can positively impact outcomes for patients by reducing implant-related infections, and bring value to our customers,” said Robert White, president and CEO of Tyrx.
The all-cash transaction includes an initial payment of USD 160 million plus potential earn out and performance based milestone payments.
Related Links:
Tyrx
Medtronic
Antibacterial Envelope Protects Cardiac Implants
Latest Business News
- Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
- Mindray to Acquire Chinese Medical Device Company APT Medical
- Olympus Acquires Korean GI Stent Maker Taewoong Medical
- Karl Storz Acquires British AI Specialist Innersight Labs
- Stryker to Acquire French Joint Replacement Company SERF SAS
- Medical Illumination Acquires Surgical Lighting Specialist Isolux
- 5G Remote-Controlled Robots to Enable Even Cross-Border Surgeries

- International Hospital Federation Announces 2023 IHF Award Winners
- Unprecedented AI Integration Transforming Surgery Landscape, Say Experts

- New WHO Guidelines to Revolutionize AI in Healthcare
- Getinge Acquires US-Based Medical Equipment Provider Healthmark Industries
- Global Surgical Lights Market Driven by Increasing Number of Procedures
- Global Capsule Endoscopy Market Driven by Demand for Accurate Diagnosis of Gastrointestinal Conditions
- Global OR Integration Market Driven by Need for Improved Workflow Efficiency and Productivity
- Global Endoscopy Devices Market Driven by Increasing Adoption of Endoscopes in Surgical Procedures
- Global Minimally Invasive Medical Devices Market Driven by Benefits of MIS Procedures
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreCritical Care
view channel
AI Brain-Age Estimation Technology Uses EEG Scans to Screen for Degenerative Diseases
As individuals age, so do their brains. Premature aging of the brain can lead to age-related conditions such as mild cognitive impairment, dementia, or Parkinson's disease. The ability to determine "brain... Read more
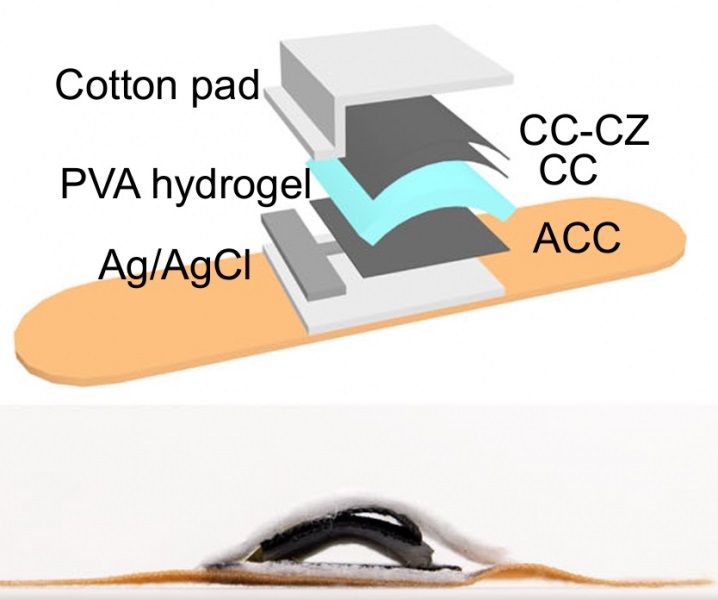
Wheeze-Counting Wearable Device Monitors Patient's Breathing In Real Time
Lung diseases like asthma, chronic obstructive pulmonary disease (COPD), lung cancer, bronchitis, and infections such as pneumonia, rank among the leading causes of death worldwide. Traditionally, medical... Read moreSurgical Techniques
view channel
Robotic Nerve ‘Cuffs’ Could Treat Various Neurological Conditions
Electric nerve implants serve dual functions: they can either stimulate or block signals in specific nerves. For example, they may alleviate pain by inhibiting pain signals or restore movement in paralyzed... Read more
Flexible Microdisplay Visualizes Brain Activity in Real-Time To Guide Neurosurgeons
During brain surgery, neurosurgeons need to identify and preserve regions responsible for critical functions while removing harmful tissue. Traditionally, neurosurgeons rely on a team of electrophysiologists,... Read more.jpg)
Next-Gen Computer Assisted Vacuum Thrombectomy Technology Rapidly Removes Blood Clots
Pulmonary embolism (PE) occurs when a blood clot blocks one of the arteries in the lungs. Often, these clots originate from the leg or another part of the body, a condition known as deep vein thrombosis,... Read morePatient Care
view channel
Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization
An innovative solution has the capability to transform surgical capacity utilization by targeting the root cause of surgical block time inefficiencies. Fujitsu Limited’s (Tokyo, Japan) Surgical Capacity... Read more
Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
A groundbreaking innovation enables hospitals to significantly improve instrument processing time and throughput in operating rooms (ORs) and sterile processing departments. Turbett Surgical, Inc.... Read more
Next Gen ICU Bed to Help Address Complex Critical Care Needs
As the critical care environment becomes increasingly demanding and complex due to evolving hospital needs, there is a pressing requirement for innovations that can facilitate patient recovery.... Read moreGroundbreaking AI-Powered UV-C Disinfection Technology Redefines Infection Control Landscape
Healthcare-associated infection (HCAI) is a widespread complication in healthcare management, posing a significant health risk due to its potential to increase patient morbidity and mortality, prolong... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more