Contraceptive Implant Hails New Era of Birth Control
|
By HospiMedica International staff writers Posted on 22 Jul 2014 |

Image: A prototype contraceptive implant (Photo courtesy of MicroCHIPS).
A fingernail-sized implant holds enough 30-microgram daily doses of levonorgestrel to provide contraception for 16 years.
Developed by MicroCHIPS (Lexington, MA, USA), a MIT (Cambridge, MA, USA) startup, the device, which measures just 20 x 20 x 7 millimeters, is designed to be implanted under the skin of the buttocks, upper arm, or abdomen. It then dispenses a daily dose levonorgestrel from a reservoir sealed with a titanium and platinum hermetic stopper. Passing an electric current through the stopper melts it temporarily, allowing a 30-microgram dose of the hormone to diffuse out each day.
Women who receive the implant also get a remote control that allows them to halt or restart the implant on demand; to conceive, women turn off the implant with a remote control; another click of the remote restarts it. Doctors could also adjust dosages remotely. MicroCHIPS have previously demonstrated how the microchip technology could release daily doses of an osteoporosis drug. The new application could revolutionize the level of control women have over their birth control technologies.
The technology also includes a secure encryption protocol to prevent outsiders from blocking or reprogramming the implants wirelessly. Another safety factor is that the device is triggered remotely by radio communication over a special frequency called Medical Implant Communication Service (MICS), which only works over a very short distance, almost requiring skin contact to function. The device will begin preclinical testing in the US in 2015, with the goal of market approval by 2018. The study describing the technology was published on July 4, 2014, in MIT Technology Review.
“The idea of using a thin membrane like an electric fuse was the most challenging and the most creative problem we had to solve,” said Robert Farra, president of MicroCHIPS. “We have heard a lot of positive feedback. Women are excited about the capability to change the state of an implant without having to go through another procedure to have it removed. They like the long-term medication, and not having to remember to take medication each day.”
Related Links:
MicroCHIPS
MIT
Developed by MicroCHIPS (Lexington, MA, USA), a MIT (Cambridge, MA, USA) startup, the device, which measures just 20 x 20 x 7 millimeters, is designed to be implanted under the skin of the buttocks, upper arm, or abdomen. It then dispenses a daily dose levonorgestrel from a reservoir sealed with a titanium and platinum hermetic stopper. Passing an electric current through the stopper melts it temporarily, allowing a 30-microgram dose of the hormone to diffuse out each day.
Women who receive the implant also get a remote control that allows them to halt or restart the implant on demand; to conceive, women turn off the implant with a remote control; another click of the remote restarts it. Doctors could also adjust dosages remotely. MicroCHIPS have previously demonstrated how the microchip technology could release daily doses of an osteoporosis drug. The new application could revolutionize the level of control women have over their birth control technologies.
The technology also includes a secure encryption protocol to prevent outsiders from blocking or reprogramming the implants wirelessly. Another safety factor is that the device is triggered remotely by radio communication over a special frequency called Medical Implant Communication Service (MICS), which only works over a very short distance, almost requiring skin contact to function. The device will begin preclinical testing in the US in 2015, with the goal of market approval by 2018. The study describing the technology was published on July 4, 2014, in MIT Technology Review.
“The idea of using a thin membrane like an electric fuse was the most challenging and the most creative problem we had to solve,” said Robert Farra, president of MicroCHIPS. “We have heard a lot of positive feedback. Women are excited about the capability to change the state of an implant without having to go through another procedure to have it removed. They like the long-term medication, and not having to remember to take medication each day.”
Related Links:
MicroCHIPS
MIT
Latest Patient Care News
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization

- Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
- Next Gen ICU Bed to Help Address Complex Critical Care Needs
- Groundbreaking AI-Powered UV-C Disinfection Technology Redefines Infection Control Landscape
- Clean Hospitals Can Reduce Antibiotic Resistance, Save Lives
- Smart Hospital Beds Improve Accuracy of Medical Diagnosis
- New Fast Endoscope Drying System Improves Productivity and Traceability
- World’s First Automated Endoscope Cleaner Fights Antimicrobial Resistance
- Portable High-Capacity Digital Stretcher Scales Provide Precision Weighing for Patients in ER
- Portable Clinical Scale with Remote Indicator Allows for Flexible Patient Weighing Use
- Innovative and Highly Customizable Medical Carts Offer Unlimited Configuration Possibilities
- Biomolecular Wound Healing Film Adheres to Sensitive Tissue and Releases Active Ingredients
- Wearable Health Tech Could Measure Gases Released From Skin to Monitor Metabolic Diseases
- Wearable Cardioverter Defibrillator System Protects Patients at Risk of Sudden Cardiac Arrest
- World's First AI-Ready Infrasound Stethoscope Listens to Bodily Sounds Not Audible to Human Ear
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreCritical Care
view channel
One-Hour Endoscopic Procedure Could Eliminate Need for Insulin for Type 2 Diabetes
Over 37 million Americans are diagnosed with diabetes, and more than 90% of these cases are Type 2 diabetes. This form of diabetes is most commonly seen in individuals over 45, though an increasing number... Read moreAI Can Prioritize Emergecny Department Patients Requiring Urgent Treatment
Emergency departments across the world are facing severe overcrowding and excessive demands, but a new study indicates that artificial intelligence (AI) might soon assist in prioritizing patients who require... Read more
AI to Improve Diagnosis of Atrial Fibrillation
Abnormal heart rhythms frequently arise from—and contribute to—structural abnormalities in the heart. Atrial fibrillation is a specific type of abnormal rhythm that may not be consistently present, often... Read more
Stretchable Microneedles to Help In Accurate Tracking of Abnormalities and Identifying Rapid Treatment
The field of personalized medicine is transforming rapidly, with advancements like wearable devices and home testing kits making it increasingly easy to monitor a wide range of health metrics, from heart... Read moreSurgical Techniques
view channel
First-Ever Contact Force Pulsed Field Ablation System to Transform Treatment of Ventricular Arrhythmias
It is estimated that over 6 million patients in the US and Europe are affected by ventricular arrhythmias, which include conditions such as ventricular tachycardia (VT) and premature ventricular contractions (PVCs).... Read more
Caterpillar Robot with Built-In Steering System Crawls Easily Through Loops and Bends
Soft robots often face challenges in being guided effectively because adding steering mechanisms typically reduces their flexibility by increasing rigidity. Now, a team of engineers has combined ancient... Read more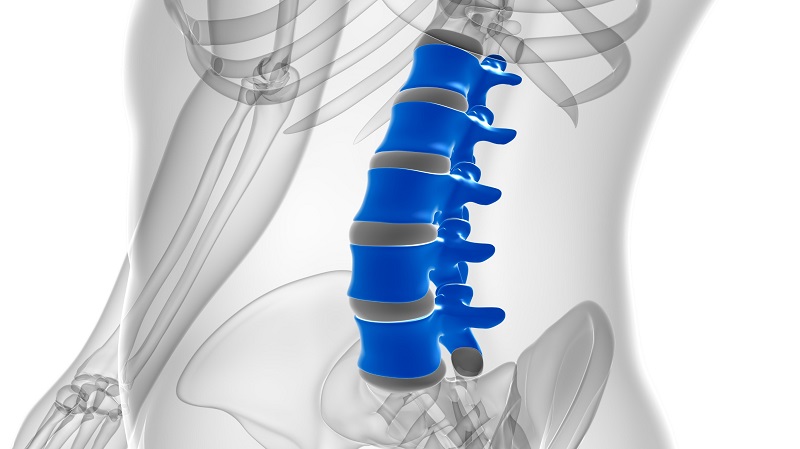
Tiny Wraparound Electronic Implants to Revolutionize Treatment of Spinal Cord Injuries
The spinal cord functions as a vital conduit, transmitting nerve impulses to and from the brain, much like a highway. When the spinal cord is damaged, this flow of information is disrupted, leading to... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel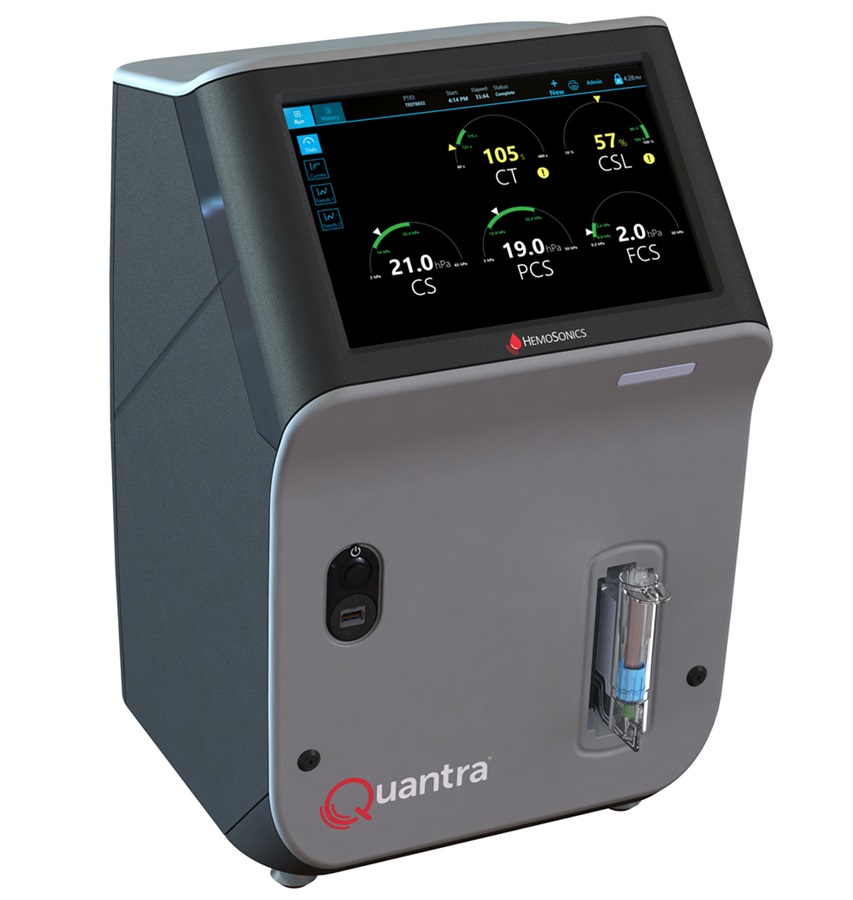
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more
New Generation Glucose Hospital Meter System Ensures Accurate, Interference-Free and Safe Use
A new generation glucose hospital meter system now comes with several features that make hospital glucose testing easier and more secure while continuing to offer accuracy, freedom from interference, and... Read moreBusiness
view channel
Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
Johnson & Johnson (New Brunswick, N.J., USA) and Shockwave Medical (Santa Clara, CA, USA) have entered into a definitive agreement under which Johnson & Johnson will acquire all of Shockwave’s... Read more