Cervical Cancer Screening Rates Declining in Younger Women
|
By HospiMedica International staff writers Posted on 21 Jan 2019 |

Image: A disparity exists between self-reported and actual CVC screening (Photo courtesy of Corbis).
A new study concludes that the percentage of women screened for cervical cancer (CVC) may be far lower than self-reported data suggests.
Researchers at the Mayo Clinic (Rochester, MN, USA) conducted an epidemiologic study in order to assess implementation of the 2012 U.S. CVC screening recommendations, which recommend Papanicolaou (PAP) testing every three years for women 21–65 years of age, or PAP–human papillomavirus (PAP-HPV) co-testing at five year intervals for women 30–65 years of age. A secondary aim was to assess sociodemographic factors correlating with screening. The study population included women 16 years and older living in Olmsted County (MN, USA).
Rochester Epidemiology Project data was collected yearly from 2005 (47,203 participants) through 2016 (49,510 participants). Significant declines in PAP completion rates over time were observed in all age groups, including an unexpected decline in women 21- 29 year of age. Coincident with decreasing PAP screening rates, PAP-HPV co-testing significantly increased among women age 30–65 years, from 10% in 2007 to 60.8% in 2016, with 64.6% of women 30-65-years of age up to date with CVC screening. The study was published on January 9, 2018, in the Journal of Women's Health.
“These cervical cancer rates are unacceptably low. Routine screening every three years with a PAP test or every five years with a PAP-HPV co-test ensures precancerous changes are caught early and may be followed more closely or treated,” said lead author Kathy MacLaughlin, MD. “African-American women were 50% less likely to be up-to-date on CVC screening than white woman, and Asian women were nearly 30% less likely to be current on screening. These racial disparities are especially concerning.”
PAP-HPV co-testing has a lower false negative rate than PAP testing alone, as it reduces the likelihood of missing an anomaly and improves detection of glandular cell abnormalities. Despite these benefits, the study results indicate that actual screening rates are substantially lower than the 81% compliance rate self-reported in the 2015 National Health Interview Survey.
Related Links:
Mayo Clinic
Researchers at the Mayo Clinic (Rochester, MN, USA) conducted an epidemiologic study in order to assess implementation of the 2012 U.S. CVC screening recommendations, which recommend Papanicolaou (PAP) testing every three years for women 21–65 years of age, or PAP–human papillomavirus (PAP-HPV) co-testing at five year intervals for women 30–65 years of age. A secondary aim was to assess sociodemographic factors correlating with screening. The study population included women 16 years and older living in Olmsted County (MN, USA).
Rochester Epidemiology Project data was collected yearly from 2005 (47,203 participants) through 2016 (49,510 participants). Significant declines in PAP completion rates over time were observed in all age groups, including an unexpected decline in women 21- 29 year of age. Coincident with decreasing PAP screening rates, PAP-HPV co-testing significantly increased among women age 30–65 years, from 10% in 2007 to 60.8% in 2016, with 64.6% of women 30-65-years of age up to date with CVC screening. The study was published on January 9, 2018, in the Journal of Women's Health.
“These cervical cancer rates are unacceptably low. Routine screening every three years with a PAP test or every five years with a PAP-HPV co-test ensures precancerous changes are caught early and may be followed more closely or treated,” said lead author Kathy MacLaughlin, MD. “African-American women were 50% less likely to be up-to-date on CVC screening than white woman, and Asian women were nearly 30% less likely to be current on screening. These racial disparities are especially concerning.”
PAP-HPV co-testing has a lower false negative rate than PAP testing alone, as it reduces the likelihood of missing an anomaly and improves detection of glandular cell abnormalities. Despite these benefits, the study results indicate that actual screening rates are substantially lower than the 81% compliance rate self-reported in the 2015 National Health Interview Survey.
Related Links:
Mayo Clinic
Latest Patient Care News
- First-Of-Its-Kind Portable Germicidal Light Technology Disinfects High-Touch Clinical Surfaces in Seconds
- Surgical Capacity Optimization Solution Helps Hospitals Boost OR Utilization

- Game-Changing Innovation in Surgical Instrument Sterilization Significantly Improves OR Throughput
- Next Gen ICU Bed to Help Address Complex Critical Care Needs
- Groundbreaking AI-Powered UV-C Disinfection Technology Redefines Infection Control Landscape
- Clean Hospitals Can Reduce Antibiotic Resistance, Save Lives
- Smart Hospital Beds Improve Accuracy of Medical Diagnosis
- New Fast Endoscope Drying System Improves Productivity and Traceability
- World’s First Automated Endoscope Cleaner Fights Antimicrobial Resistance
- Portable High-Capacity Digital Stretcher Scales Provide Precision Weighing for Patients in ER
- Portable Clinical Scale with Remote Indicator Allows for Flexible Patient Weighing Use
- Innovative and Highly Customizable Medical Carts Offer Unlimited Configuration Possibilities
- Biomolecular Wound Healing Film Adheres to Sensitive Tissue and Releases Active Ingredients
- Wearable Health Tech Could Measure Gases Released From Skin to Monitor Metabolic Diseases
- Wearable Cardioverter Defibrillator System Protects Patients at Risk of Sudden Cardiac Arrest
- World's First AI-Ready Infrasound Stethoscope Listens to Bodily Sounds Not Audible to Human Ear
Channels
Artificial Intelligence
view channel
AI-Powered Algorithm to Revolutionize Detection of Atrial Fibrillation
Atrial fibrillation (AFib), a condition characterized by an irregular and often rapid heart rate, is linked to increased risks of stroke and heart failure. This is because the irregular heartbeat in AFib... Read more
AI Diagnostic Tool Accurately Detects Valvular Disorders Often Missed by Doctors
Doctors generally use stethoscopes to listen for the characteristic lub-dub sounds made by heart valves opening and closing. They also listen for less prominent sounds that indicate problems with these valves.... Read moreCritical Care
view channel
Machine Learning Tool Identifies Rare, Undiagnosed Immune Disorders from Patient EHRs
Patients suffering from rare diseases often endure extensive delays in receiving accurate diagnoses and treatments, which can lead to unnecessary tests, worsening health, psychological strain, and significant... Read more
On-Skin Wearable Bioelectronic Device Paves Way for Intelligent Implants
A team of researchers at the University of Missouri (Columbia, MO, USA) has achieved a milestone in developing a state-of-the-art on-skin wearable bioelectronic device. This development comes from a lab... Read more
First-Of-Its-Kind Dissolvable Stent to Improve Outcomes for Patients with Severe PAD
Peripheral artery disease (PAD) affects millions and presents serious health risks, particularly its severe form, chronic limb-threatening ischemia (CLTI). CLTI develops when arteries are blocked by plaque,... Read more
AI Brain-Age Estimation Technology Uses EEG Scans to Screen for Degenerative Diseases
As individuals age, so do their brains. Premature aging of the brain can lead to age-related conditions such as mild cognitive impairment, dementia, or Parkinson's disease. The ability to determine "brain... Read moreSurgical Techniques
view channel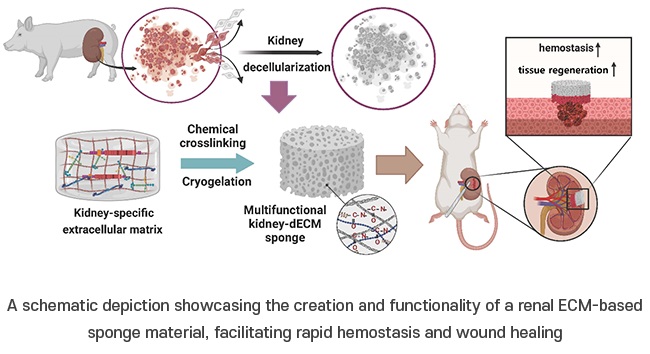
Porous Gel Sponge Facilitates Rapid Hemostasis and Wound Healing
The kidneys are essential organs that handle critical bodily functions, including waste elimination and blood pressure regulation. Often referred to as the silent organ because they typically do not manifest... Read more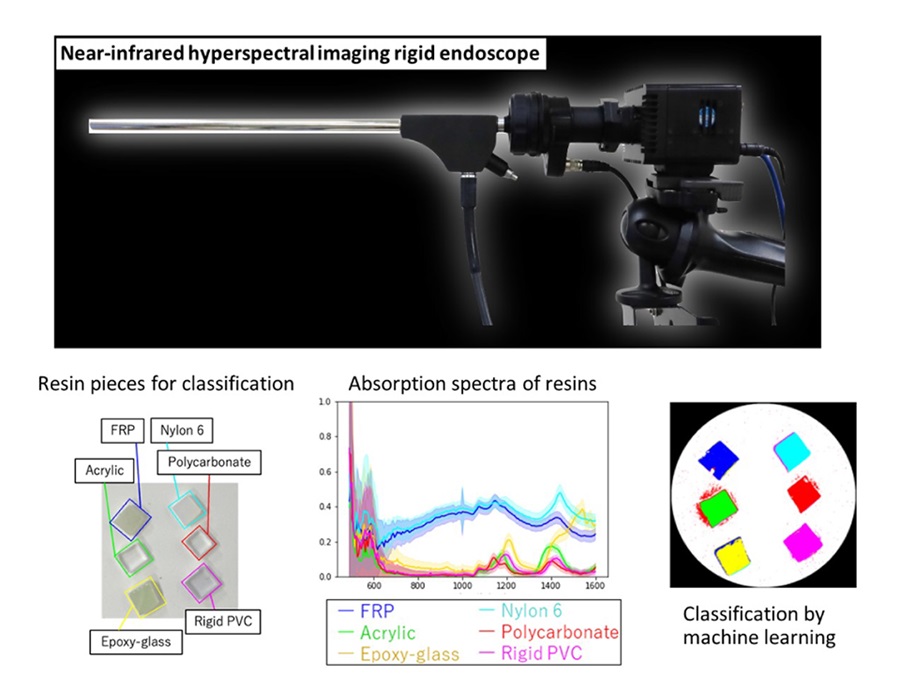
Novel Rigid Endoscope System Enables Deep Tissue Imaging During Surgery
Hyperspectral imaging (HSI) is an advanced technique that captures and processes information across a given electromagnetic spectrum. Near-infrared hyperspectral imaging (NIR-HSI) has particularly gained... Read more
Robotic Nerve ‘Cuffs’ Could Treat Various Neurological Conditions
Electric nerve implants serve dual functions: they can either stimulate or block signals in specific nerves. For example, they may alleviate pain by inhibiting pain signals or restore movement in paralyzed... Read more
Flexible Microdisplay Visualizes Brain Activity in Real-Time To Guide Neurosurgeons
During brain surgery, neurosurgeons need to identify and preserve regions responsible for critical functions while removing harmful tissue. Traditionally, neurosurgeons rely on a team of electrophysiologists,... Read moreHealth IT
view channel
Machine Learning Model Improves Mortality Risk Prediction for Cardiac Surgery Patients
Machine learning algorithms have been deployed to create predictive models in various medical fields, with some demonstrating improved outcomes compared to their standard-of-care counterparts.... Read more
Strategic Collaboration to Develop and Integrate Generative AI into Healthcare
Top industry experts have underscored the immediate requirement for healthcare systems and hospitals to respond to severe cost and margin pressures. Close to half of U.S. hospitals ended 2022 in the red... Read more
AI-Enabled Operating Rooms Solution Helps Hospitals Maximize Utilization and Unlock Capacity
For healthcare organizations, optimizing operating room (OR) utilization during prime time hours is a complex challenge. Surgeons and clinics face difficulties in finding available slots for booking cases,... Read more
AI Predicts Pancreatic Cancer Three Years before Diagnosis from Patients’ Medical Records
Screening for common cancers like breast, cervix, and prostate cancer relies on relatively simple and highly effective techniques, such as mammograms, Pap smears, and blood tests. These methods have revolutionized... Read morePoint of Care
view channel
Critical Bleeding Management System to Help Hospitals Further Standardize Viscoelastic Testing
Surgical procedures are often accompanied by significant blood loss and the subsequent high likelihood of the need for allogeneic blood transfusions. These transfusions, while critical, are linked to various... Read more
Point of Care HIV Test Enables Early Infection Diagnosis for Infants
Early diagnosis and initiation of treatment are crucial for the survival of infants infected with HIV (human immunodeficiency virus). Without treatment, approximately 50% of infants who acquire HIV during... Read more
Whole Blood Rapid Test Aids Assessment of Concussion at Patient's Bedside
In the United States annually, approximately five million individuals seek emergency department care for traumatic brain injuries (TBIs), yet over half of those suspecting a concussion may never get it checked.... Read more
New Generation Glucose Hospital Meter System Ensures Accurate, Interference-Free and Safe Use
A new generation glucose hospital meter system now comes with several features that make hospital glucose testing easier and more secure while continuing to offer accuracy, freedom from interference, and... Read moreBusiness
view channel
Johnson & Johnson Acquires Cardiovascular Medical Device Company Shockwave Medical
Johnson & Johnson (New Brunswick, N.J., USA) and Shockwave Medical (Santa Clara, CA, USA) have entered into a definitive agreement under which Johnson & Johnson will acquire all of Shockwave’s... Read more














